
Capsule packaging refers to how pharmaceutical capsules (hard-shell or softgel) are enclosed for distribution and use. It includes primary packaging (the immediate container in contact with the capsules, e.g. blister cavities or bottles) and secondary packaging (outer cartons, boxes, and labels that further protect and identify the product). Good capsule packaging must protect the drug from moisture, light, and contamination, ensure stability through shelf-life, and meet regulatory requirements. For example, WHO notes that “packaging components surround the product from production until use,” and must satisfy functions like protection, stability, and identification. FDA’s GMP regulations (21 CFR 211) require written procedures for inspecting and testing packaging materials upon receipt, and mandate secure labeling and tamper controls. In practice, we must match the capsule product (dosage form, sensitivity, dosage, etc.) to the appropriate packaging format and material.

The primary packaging of pharmaceutical capsules – is critical for product protection, compliance, and patient safety: It keeps capsules dry, uncontaminated, and clearly labeled. For instance, blister packs and amber bottles are known to be ideal for moisture-sensitive medicines. The right packaging also supports patient compliance (unit-dose blister dosing, child-resistant bottles, clear labeling) and supply-chain needs (serialization, logistics). Below we review each option in detail.
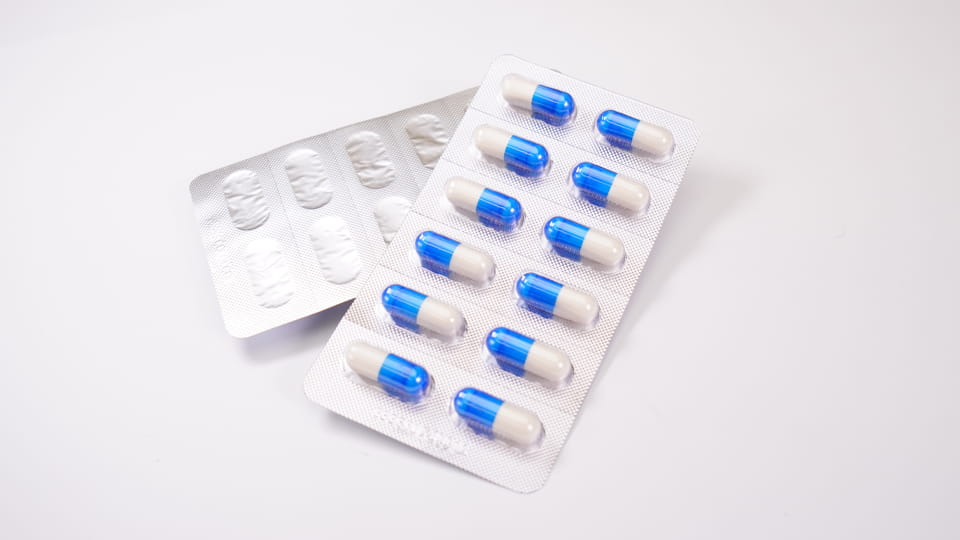
Blister packs consist of pre-formed plastic cavities (“blisters”) that hold individual capsules, sealed with a backing (foil or plastic). This primary packaging offers unit-dose protection. Blisters provide excellent barrier properties: they can be made with materials like PVC/PVDC or foil (Alu-PVC, Alu-Alu) to block moisture, oxygen and light. For example, Jinlu’s DPP-180Pro blister machine can form and seal up to 4,800 capsule blisters per hour in Alu-PVC or Alu-Alu laminates. Key features of capsule blister packaging include:
Figure:Jinlu JL-180Pro blister packaging machine forming capsule blisters (Alu-PVC), 4,800 cavities/hour.
Jinlu’s blister machines (e.g. the DPP-180Pro) can automatically form cavities, feed capsules, seal, cut plates, and even print lot codes. They typically run at medium-high speed (thousands of cavities per hour), ideal for mass production. Different blister formats (Alu-PVC vs Alu-Alu) are selected by product: Alu-Alu (foil-foil) offers the highest barrier (best for very moisture-sensitive drugs), whereas PVC/PVDC is common for standard capsules. Blister packaging also simplifies counting and inspection: most lines easily integrate camera inspection or tablet-counting machines to verify capsule fill.
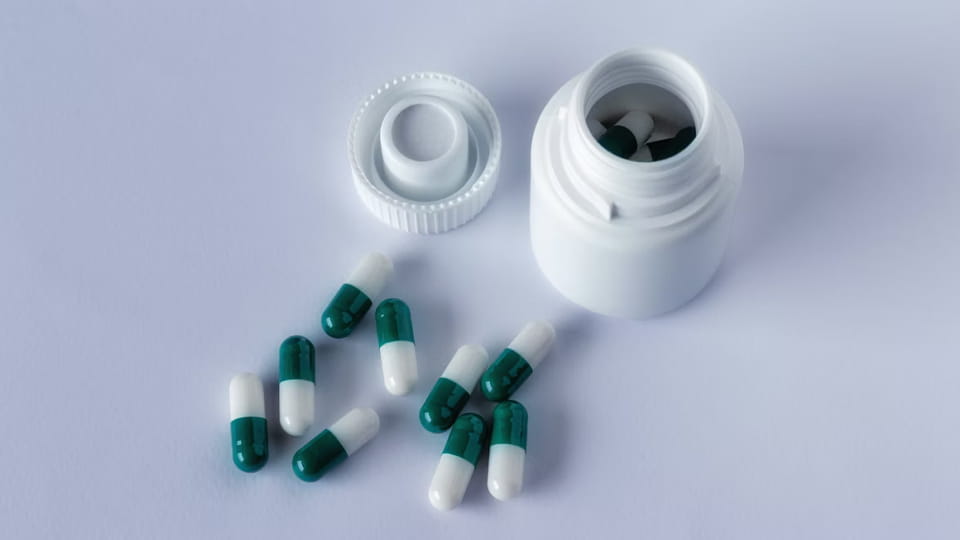
Bottles are the classic multi-dose container. Capsules are filled into bottles (plastic or glass) which are then capped. This format is well known for vitamins, supplements, and many prescription pills. Key features:
Figure: Jinlu JL-16H capsule/tablet bottling line (100 bottles/min) with unscrambler, filler, capper, and labeler.
Capsule bottling lines integrate multiple machines: a bottle unscrambler (or feed hopper), a counting-filling machine (that drops X capsules into each bottle), a capping/sealing machine (apply and tighten caps), and a labeling machine. For example, Jinlu’s JL-16H line can handle 3–40 mm capsules, achieving ~100 bottles/min with >99.8% counting accuracy. Bottles can be filled in bulk very rapidly. To meet regulations, bottle lines often include a station for inserting desiccants (e.g. silica packs) and a tamper-evident seal station. Child-resistant caps are another common feature for pediatric meds.
In choosing bottle packaging, consider product volume and stability: high-volume products (e.g. daily vitamins) suit bottles, whereas low-volume or highly sensitive capsules may prefer blisters. Note bottles generally offer less inherent moisture/oxygen protection than high-barrier blisters. Amber glass or opaque containers can add light protection, and inner seals (aluminum foil liners) can improve barrier.

Sachets (and stick packs) are flexible packets that hold one dose of powder, granules or liquid. While not as common for capsules, they are relevant for related dosage forms (e.g. nutraceutical capsules broken into powder, or powder filler itself). Nonetheless, sachets and stick packs deserve mention in a capsule packaging guide:
Both sachets and stick packs are produced by vertical form-fill-seal (VFFS) or horizontal FFS machines. Jinlu’s JL-VP series, for instance, is a multi-lane stick/sachet FFS machine (40 bags/min/lane, ±0.02g accuracy) for powders, granules, liquids or gels. If capsules themselves were to be packaged in sachets, one would open the sachet to retrieve the dose – but more commonly, capsules are packaged intact in blister or bottle, while sachets/stick packs handle powdered contents.
Regulatory note: sachets and stick packs are allowed for solid and semisolid drugs (per WHO definitions). They are considered primary packaging if they directly contain the product. Like blisters, sachets can be designed child-resistant or with tear notches.
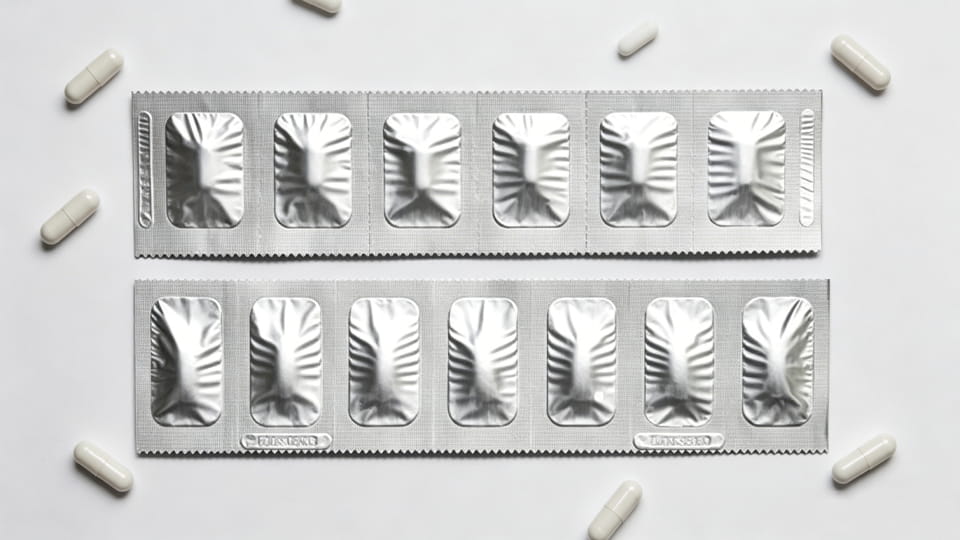
Strip packs (also called foil strips or “dose bands”) are another form of unit-dose primary pack. A strip is typically a narrow web of material (often aluminum or multi-layer laminate) folded over and sealed, with perforations between doses. Unlike blisters, strip packs don’t form deep cavities – instead, the capsule is sandwiched between two layers of film. Strip packs are widely used in Asia and for products where high moisture protection is needed but blisters are too bulky. Key points:

From a selection standpoint, strip packs occupy a middle ground between bottle (bulk) and blister (rigidity). They are often chosen when very high moisture barrier is needed but in a thin, lightweight form. (Note: strip packs may not provide tamper evidence as clearly as blisters unless additional seals are used.)
After choosing a primary pack, capsules are usually placed into secondary packaging like carton boxes or shrink-wrapped cases. Cartons provide extra protection during shipping, allow additional labeling (drug info, branding, tamper-evident seals), and help organize multi-pack products. Key considerations for secondary packaging:
In summary, secondary packaging wraps up the product for final distribution and ensures all labeling and compliance features are in place. Good design here enhances brand image and user convenience (easy-open cartons, clear labeling).
Choosing the right packaging material is critical for protecting capsules. Materials must be compatible with the drug, meet barrier needs, and comply with regulations (ISO 15378 GMP for packaging materials, etc.). Common materials include:
Generally, choose the highest barrier that makes sense for your capsule. For example, a moisture-sensitive gelatin capsule may warrant Alu-Alu blister or an amber glass bottle with desiccant. In contrast, non-sensitive capsules might use standard PVC blister or HDPE bottle. Consider also shelf life: longer-term products need more robust barrier.
Safety/recycle notes: Sustainable trends favor mono-material packs (e.g. PP blisters, recyclable PET bottles). Testing materials for extractables, compatibility, and meeting FDA/EU limits (e.g. USP <661> for plasticizers) is part of packaging compliance.
Capsules come in various types and their formulation dictates packaging needs:
In short, the capsule shell material and fill type will affect packaging choice. Highly moisture-sensitive formulations always favor the tightest barrier (Alu-Alu blister, foil sachet, or sealed bottle with desiccant). Less sensitive formulations have more options. Always test stability of the final packaged capsule (ICH Q1A requires stability in the proposed container).
Selecting an optimal capsule packaging involves evaluating multiple factors: dosage form, patient compliance, product sensitivity, shelf-life, regulatory requirements, and cost. Below is a simplified decision framework:
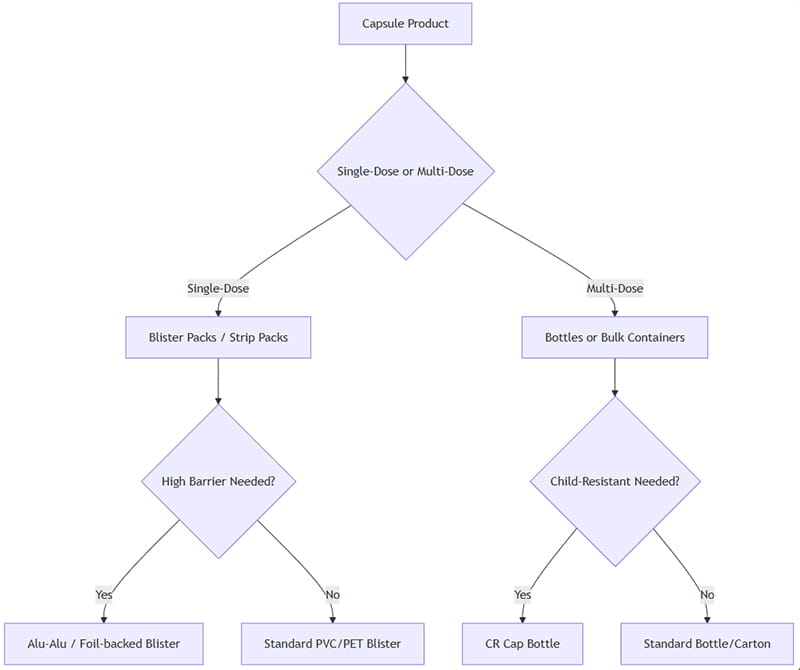
How to apply this: If the capsules are intended as single units (e.g. tight dosing or retail blister cards), blisters/strips are preferred. For multi-dose (e.g. supplements or high-use drugs), bottles are more efficient. If moisture/light protection is critical, select high-barrier options (Alu-Alu, amber bottle). If pediatric safety is needed, use child-resistant bottle closures or tamper-evident blister designs.
Other criteria from the industry include:
Ultimately, pilot testing (stability studies, consumer studies) and total-cost analysis will guide the final decision. The flowchart above is a starting point for typical scenarios.
Packaging capsules requires specialized machinery. After capsules are filled (by capsule fillers), these are transferred to packaging lines:
Equipment compliance: All packaging machines for pharmaceuticals must meet GMP design (stainless steel, cleanable surfaces, validated performance). Jinlu’s machines are CE/cGMP certified, often with configurable guardings and reject systems for out-of-spec pills. Integration is key: for example, a blister machine can feed directly into a cartoner that erects boxes and packs each blister card. Comprehensive systems (from filling to final box) allow end-to-end automation of capsule packaging.
Capsule packaging for pharmaceuticals must adhere to strict regulations. Key points include:
Overall, meeting FDA, EMA and WHO guidelines is essential. Many packagers work with regulated suppliers (like Jinlu) who can provide validated machines and documentation (IQ/OQ/PQ).
Choosing the right capsule packaging requires balancing protection, usability, and cost. Blister packs, bottles, sachets, and strips each have their place in pharmaceutical packaging. Factors like moisture sensitivity, dose form, and patient needs guide the decision. Alongside, stringent regulatory and GMP requirements must be met.
At Jinlu Packing, we specialize in end-to-end pharmaceutical packaging solutions. Our equipment – from high-speed blister machines to fully automated counting & bottling lines – helps you package capsules efficiently and compliantly. We also support sustainable choices (e.g. machines that run new recyclable films) and offer one-stop service (customization, validation). Our team can help you evaluate the best capsule packaging system for your product.
Ready to optimize your capsule packaging? Contact Jinlu Packing today for expert guidance and quotes. Our solutions are backed by global quality standards and over 3,000 satisfied customers worldwide. Let’s package your capsules for success!
Capsule packaging refers to the process of enclosing pharmaceutical capsules in protective materials to maintain stability, safety, and compliance. Common formats include blister packs, bottles, and strip packaging.
In pharmaceutical practice, packaging is not just containment—it protects capsules from moisture, oxygen, and contamination, which directly affects drug quality and shelf life.
There is no single “best” option—it depends on your product and market.
• Blister packaging → best for moisture-sensitive or high-value drugs
• Bottle packaging → best for bulk storage and supplements
• Strip packaging → common in cost-sensitive markets
Blister packaging is often preferred because it provides unit-dose protection and better environmental isolation.
Use materials that block the key threats (moisture, oxygen, light). For example, Alu-Alu foil blisters or amber glass bottles offer excellent barrier for moisture-sensitive capsules. PVC or PET blisters with PVDC coating are common and cheaper for stable products. HDPE bottles are standard but may need a desiccant pouch if the capsules are sensitive. Evaluate the water vapor transmission rates (WVTR) of films. In sustainability-conscious production, consider recyclable monomaterials (e.g. PP blisters). Always test packaging compatibility via stability studies.
Capsules are sensitive to environmental factors such as humidity, oxygen, and light.
Proper packaging:
• Prevents moisture absorption
• Reduces oxidation
• Maintains chemical stability
Blister packaging, in particular, creates sealed micro-environments for each capsule, significantly extending shelf life.
It depends on format. For blisters, you need a blister forming/sealing machine (with cavity molds to fit your capsule) plus possibly a cartoner. For bottles, you need a bottle filling/capping line (unscrambler, counter-filler, capper, labeler). For stick packs, use a vertical form-fill-seal sachet machine. Jinlu offers all these: e.g. our DPP series blisters, JL-16H counting line, and JL-VP sachet packer. Selecting the right machine involves capacity, capsule size, and required integrations (vision systems, reject stations, etc.).
Consider factors like dosage (single vs. multiple), stability needs, and patient use. For single-dose convenience and high protection (especially for moisture/light sensitive capsules), blisters are often preferred. For large-volume or economic reasons, bottles are usually chosen. Also, if child-resistant or elderly-friendly features are needed, bottles may offer more options. As a rule, match your capsule’s properties to packaging: e.g. very hygroscopic capsules → Alu-Alu blister; high-demand supplements → bottles.
References:
1.Annex 9 Guidelines on packaging for pharmaceutical products —— WHO
2.Technical Requirements for Medicines and Health Products —— World Health Organization
3.Pharmaceutical Packaging Materials and Medication Safety: A Mini-Review —— MDPI
4.Pharmacy Packaging and Inserts —— National Library of Medicine
5.A review on characterization and recyclability of pharmaceutical blisters —— ScienceDirect

Petty Fu, Founder of Jinlupacking, brings over 20 years of expertise to the pharmaceutical machinery sector. Under his leadership, Jinlu has grown into a trusted supplier integrating design, production, and sales. Petty is passionate about sharing his deep industry knowledge to help clients navigate the complexities of pharma packaging, ensuring they receive not just equipment, but a true one-stop service partnership tailored to their production goals.