
Pharmaceutical excipients are the “inactive” ingredients that make medicines work. In simple terms, a pharmaceutical excipient is any substance other than the active pharmaceutical ingredient (API) that is intentionally included in a drug formulation. These excipients help shape the final medication – they give tablets their size and form, suspend ingredients in syrups, thicken creams, improve stability, and make pills safe to swallow. While excipients themselves have no therapeutic effect, they play vital supporting roles. In fact, nearly every commercial drug contains excipients, often more by weight than the API. Think of them as essential helpers behind the scenes: without the right excipients, a medication wouldn’t hold together, dissolve properly, or meet safety standards.
Excipients come from many sources – common examples include sugars (like lactose, sucrose), starches, cellulose derivatives, polymers (e.g. polyvinylpyrrolidone or polyethylene glycol), and even water or oils. By definition, excipients must be carefully tested and pharmacopeial-grade. They are evaluated for safety and purity just like APIs. In practice, excipients should be inert and non-reactive, but they can have active effects (for example, some help drugs dissolve or stabilize them). Broadly, excipients are chosen to aid manufacturing and improve the drug’s performance without acting as medicine themselves.
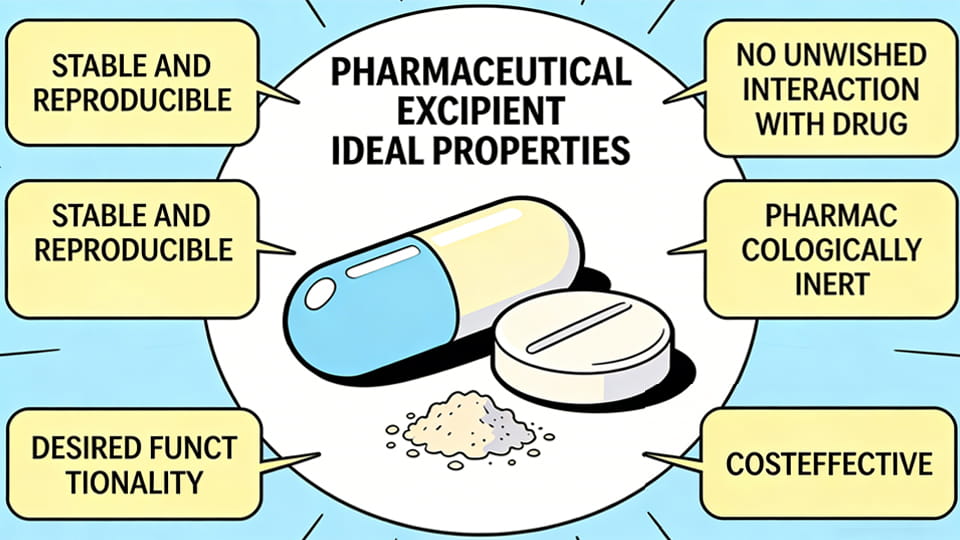
Figure: Ideal properties of pharmaceutical excipients. An ideal excipient is stable, safe, inert, functional (performs its role well), and cost-effective. In reality, excipients vary from very simple natural substances to engineered polymers. They must be compatible with the API and dosage form. For example, some excipients improve a drug’s solubility or absorption, control viscosity, or protect the active ingredient from moisture or degradation. Modern excipient design balances these qualities: they need to be pharma-grade (meeting USP, Ph. Eur., JP standards) and produced under GMP conditions.
Excipients perform multiple critical functions in a drug product:
In plain language, excipients are the “stagehands” of a medication. You don’t see their effect as you take a pill, but the medicine wouldn’t work without them. For example, an excipient may act as a binder to hold tablet ingredients together or as a glidant to improve powder flow into the tablet press. These functions are crucial. Without disintegrants, a tablet might not dissolve properly; without lubricants, tablets could stick to the press machinery; without stabilizers, a medicine could spoil before reaching patients.
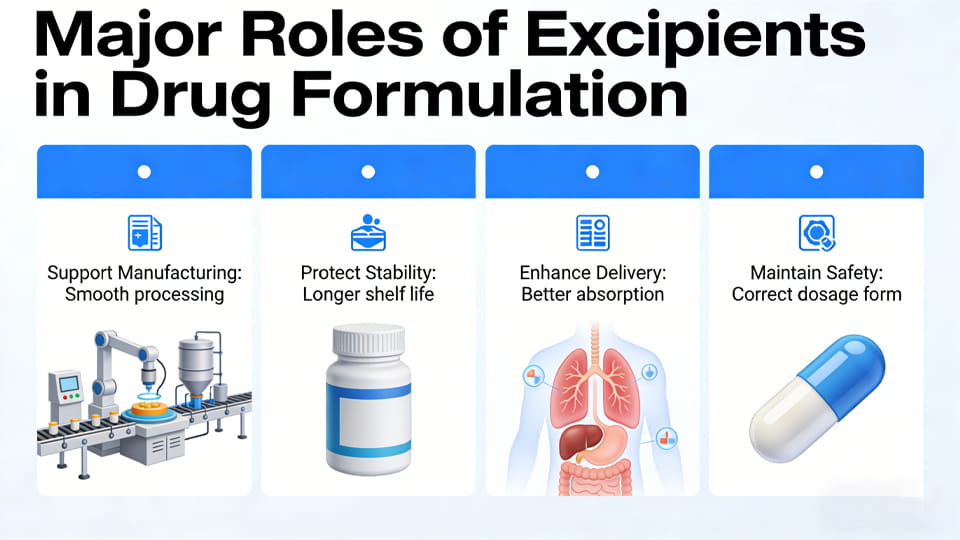
Figure: Major roles of excipients in a drug formulation. Excipients support manufacturing (smooth processing), protect stability (longer shelf life), enhance delivery (better absorption), and maintain safety (correct dosage form). Each excipient is chosen for one or more of these roles.
Pharmaceutical excipients are often categorized by their function or by the dosage form they support. Below is a quick reference table of common excipient categories, what they do, and examples of each:
| Category | Primary Function | Examples |
|---|---|---|
| Fillers / Diluents | Add bulk to formulations, especially when the API dose is small. | Lactose, microcrystalline cellulose, starch |
| Binders | Hold the tablet or granules together. | Polyvinylpyrrolidone (PVP), hydroxypropyl cellulose, starch |
| Disintegrants | Help tablets/capsules break apart (disperse) when in contact with liquid. | Croscarmellose sodium, sodium starch glycolate (SSG), corn starch |
| Lubricants | Reduce friction in tablet presses or capsule machines to prevent sticking. | Magnesium stearate, stearic acid, talc |
| Glidants | Improve powder flow properties during manufacturing. | Colloidal silicon dioxide, talc |
| Coating agents | Form a protective or functional film on tablets. | Hydroxypropyl methylcellulose (HPMC), ethylcellulose, shellac |
| Preservatives | Prevent microbial growth in liquids/creams. | Sodium benzoate, parabens (methylparaben), sorbic acid |
| Solvents (liquids) | Dissolve or suspend ingredients in liquid forms. | Purified water, ethanol, glycerin |
| Thickeners (liquids/semisolids) | Increase viscosity, stabilize suspensions or gels. | Methylcellulose, carbomer, xanthan gum |
| Emulsifiers | Stabilize oil/water mixtures in creams/ointments. | Lecithin, polysorbates, cetostearyl alcohol |
| Humectants | Retain moisture in formulations (e.g., creams). | Glycerin, propylene glycol, sorbitol |
| Colorants & Flavors | Improve appearance and taste for better compliance. | Titanium dioxide (white colorant), food dyes, sucrose, fruit flavors |
Each drug formulation uses a subset of these depending on its form. For example, a tablet may contain fillers, binders, disintegrants, lubricants, and a coating. A liquid syrup will use solvents (water), sweeteners, preservatives, and viscosity modifiers. A cream or gel includes emulsifiers, thickeners, and humectants.

Different dosage forms require specific excipients:
This breakdown shows how excipients are tailored to the form. For every pill, syrup, or cream, the right “inactive” ingredients make it possible to manufacture, store, and use that product effectively.
In manufacturing, excipients and equipment go hand-in-hand. For example, once the powder blend of API plus excipients is ready, a tablet press machine (sometimes called a pill maker) compresses it into tablets. Modern pill maker machines, from small tabletop presses to large rotary tablet presses, are engineered for precision: they apply adjustable pressure to form tablets of uniform weight and hardness. High-quality machines are made of stainless steel (easy to clean) and comply with GMP standards. The right combination of excipients (fillers, binders, lubricants, etc.) ensures these machines run smoothly. For instance, without sufficient lubricant (like magnesium stearate), powder may stick to the punch faces and jam the tablet press.

Figure: Automated tablet press compressing powders into tablets. Tablet presses (pill maker machines) combine the API and excipients into firm tablets. The design of these machines (often with computer controls) and use of proper excipients ensures each tablet has consistent weight and dissolution.
Similarly, capsule filling machines are used when the dosage form is a capsule. These machines automatically fill empty gelatin or HPMC capsules with precise doses of powdered ingredients. In industrial production, capsule fillers work at high speed and accuracy. A capsule filler is literally “a type of machine used to fill empty capsules with pharmaceutical ingredients,” combining APIs with excipients to create each dose. Manufacturers may use manual, semi-automatic, or fully automatic capsule-filling equipment depending on their output needs. Across the board, excipients like fillers and lubricants are carefully selected so that the powder flows and fills the capsule uniformly. The synergy between formulation and machinery is critical – good excipients make production efficient, while good equipment ensures product quality.
Because excipients comprise most of a drug’s mass (often 50–90%), their quality is vital. Regulatory bodies and pharmacopeias (e.g. USP, Ph. Eur., JP) require that every ingredient in a medication be identified and tested for safety. This means excipients must meet strict standards: they are designated pharmaceutical grade and manufactured under Good Manufacturing Practice (GMP) conditions. In practice, this involves detailed specifications for purity, microbial limits, and absence of contaminants. The sourcing of excipients is also closely monitored – global companies often audit suppliers to ensure consistent quality across batches. As one industry expert notes, “Quality assurance in sourcing and using excipients is paramount. Companies need to ensure that these substances meet all regulatory standards for safety and efficacy.”
Excipients can also have regulatory impacts on the final product. For example, if an excipient causes an unexpected interaction or allergy, that could trigger additional safety reviews. Therefore, formulators prefer well-characterized, proven excipients. Many regulatory filings now include detailed profiles of each excipient, including its functional role and any relevant testing.
The field of excipients is evolving. Traditional excipients (sugars, cellulose, mineral salts) are still ubiquitous, but new “functional excipients” are emerging. These advanced ingredients might carry the API (like certain lipids or cyclodextrins for improved delivery), or respond to triggers (pH, temperature) for controlled release. Personalized medicine also demands novel excipients: for instance, tailor-made polymers for 3D-printed pills or biocompatible materials for injectables. Nanotechnology is another frontier; nanoscale carriers often blur the line between “excipient” and “drug delivery system,” but still rely on excipients to stabilize nanoparticles. The overall goal remains the same: enable better, safer, more effective medications. As one review puts it, excipients are indispensable in ensuring “the safety, effectiveness, and quality of medications,” and their importance will only grow as drug technology advances.
In summary, a pharmaceutical excipient is any non-active ingredient in a drug that helps make it into an effective, safe product. Excipients are carefully chosen for each formulation – whether a tablet, capsule, liquid, or cream – to improve manufacturability, stability, and patient experience. They enable pills and capsules to be formed by machines like tablet presses and capsule fillers. For pharma professionals (engineers, buyers, manufacturers), understanding excipients means knowing what makes a good formulation and how to meet global standards.
If you’re planning a new formulation or upgrading your production line, remember to consider excipient selection alongside equipment. Need help with equipment or formulations? Our team is ready to assist. Contact Jinlu Packaging today for a quote on high-quality tablet presses, capsule filling machines, and complete pharmaceutical production solutions.
A pharmaceutical excipient is any substance in a drug product that is not the active pharmaceutical ingredient (API). These ingredients are added to support the formulation, stability, manufacturing process, and usability of the medicine. Excipients help create tablets, capsules, liquids, or creams that are safe, stable, and easy for patients to take.
Pharmaceutical excipients play several critical roles in pharma manufacturing. They improve powder flow, help tablets maintain their shape, enhance drug stability, and ensure consistent dosing. Without excipients, many APIs could not be processed into practical dosage forms like tablets or capsules.
Some common examples of excipients include:
• Lactose (filler or diluent)
• Microcrystalline cellulose (binder)
• Magnesium stearate (lubricant)
• Croscarmellose sodium (disintegrant)
• Hydroxypropyl methylcellulose (tablet coating)
• Sodium benzoate (preservative)
These ingredients support formulation stability, manufacturability, and patient acceptability.
Typical excipients used in tablet formulation include:
• Fillers or diluents
• Binders
• Disintegrants
• Lubricants
• Glidants
• Coating agents
These ingredients ensure tablets compress properly, break down correctly in the body, and remain stable during storage and transport.
Excipients used in liquid dosage forms help maintain stability and improve taste. Common examples include:
• Water or glycerin (solvents)
• Sweeteners and flavoring agents
• Preservatives
• Buffering agents
• Viscosity modifiers
These excipients ensure the drug remains stable, palatable, and safe throughout its shelf life.
Semi-solid dosage forms like creams, gels, and ointments typically use:
• Emulsifiers
• Thickening agents
• Humectants
• Preservatives
• Base materials such as petroleum jelly or waxes
These excipients provide the proper texture, stability, and drug release characteristics for topical medications.
Yes. Pharmaceutical excipients must meet strict regulatory standards before being used in medicines. They are carefully evaluated for safety, compatibility, and quality by regulatory authorities such as the FDA and EMA. Only approved excipients that meet pharmacopeial standards are used in pharmaceutical products.
Excipients directly influence the performance of pharmaceutical production equipment such as tablet presses and capsule filling machines. Lubricants reduce friction in tablet compression, while glidants improve powder flow to ensure accurate dosing and consistent tablet weight during large-scale production.
The Active Pharmaceutical Ingredient (API) is the component that produces the therapeutic effect of a drug. In contrast, a drug excipient is an inactive ingredient that supports the API by improving stability, manufacturing efficiency, and delivery to the body.
Formulation scientists choose excipients based on several factors:
• Compatibility with the API
• Dosage form requirements
• Manufacturing process
• Stability and shelf life
• Regulatory compliance
The goal is to create a formulation that is safe, effective, and easy to manufacture at scale.
References:
1. Glossary:Definition of pharmaceutical excipient — World Health Organization (WHO).
2.Get Involved in development of new and revised standards for Excipients — United States Pharmacopeia (USP).
3.General Overview of Pharmaceutical Excipients — Wikipedia.
4.A comprehensive review on pharmaceutical excipients — National Library of Medicine.

Petty Fu, Founder of Jinlupacking, brings over 30 years of expertise to the pharmaceutical machinery sector. Under his leadership, Jinlu has grown into a trusted supplier integrating design, production, and sales. Petty is passionate about sharing his deep industry knowledge to help clients navigate the complexities of pharma packaging, ensuring they receive not just equipment, but a true one-stop service partnership tailored to their production goals.
Copyright © 2026 JinLuPacking.All rights reserved. Terms & Conditions and Privacy Policy
Friendly Links: Rich Packing | Capsule Filling Machine Manufacturers