
Bao bì viên nang đề cập đến cách viên nang dược phẩm (vỏ cứng hoặc viên mềm) được kèm theo để phân phối và sử dụng. Nó bao gồm bao bì sơ cấp (thùng chứa ngay lập tức tiếp xúc với viên nang, ví dụ. lỗ phồng rộp hoặc chai) Và bao bì thứ cấp (thùng carton bên ngoài, Hộp, và nhãn nhằm bảo vệ và nhận dạng sản phẩm hơn nữa). Bao bì viên nang tốt phải bảo vệ thuốc khỏi độ ẩm, ánh sáng, và ô nhiễm, đảm bảo sự ổn định thông qua thời hạn sử dụng, và đáp ứng các yêu cầu quy định. Ví dụ, WHO lưu ý rằng “Các thành phần bao bì bao quanh sản phẩm từ khi sản xuất cho đến khi sử dụng,” và phải đáp ứng các chức năng như bảo vệ, sự ổn định, và nhận dạng. Quy định GMP của FDA (21 CFR 211) yêu cầu có quy trình bằng văn bản để kiểm tra và thử nghiệm vật liệu đóng gói khi nhận, và bắt buộc phải ghi nhãn an toàn và kiểm soát giả mạo. Trong thực tế, chúng ta phải phù hợp với sản phẩm viên nang (dạng bào chế, sự nhạy cảm, Liều dùng, vân vân.) với hình thức và chất liệu đóng gói phù hợp.

Bao bì chính của viên nang dược phẩm – rất quan trọng đối với bảo vệ sản phẩm, Tuân thủ, và an toàn bệnh nhân: Nó giữ viên nang khô, không bị ô nhiễm, và được dán nhãn rõ ràng. Ví dụ, vỉ và chai màu hổ phách được biết đến là lý tưởng cho các loại thuốc nhạy cảm với độ ẩm. Bao bì phù hợp cũng hỗ trợ sự tuân thủ của bệnh nhân (liều lượng đơn vị vỉ, chai chống trẻ em, ghi nhãn rõ ràng) và nhu cầu của chuỗi cung ứng (tuần tự hóa, hậu cần). Dưới đây chúng tôi xem xét chi tiết từng tùy chọn.
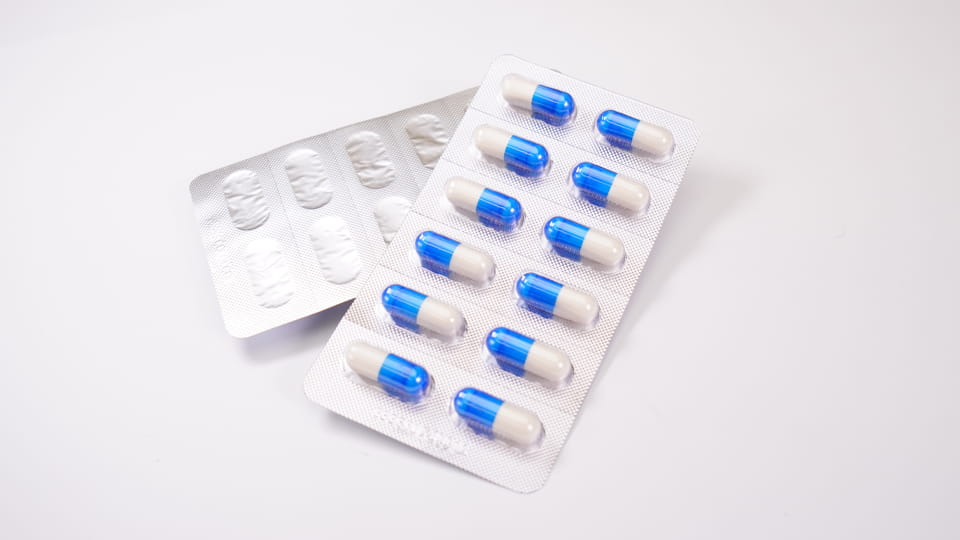
Gói vỉ bao gồm các khoang nhựa được hình thành trước (“mụn nước”) chứa các viên nang riêng lẻ, niêm phong với một sự ủng hộ (giấy bạc hoặc nhựa). Cái này bao bì sơ cấp ưu đãi bảo vệ liều đơn vị. Vỉ cung cấp rào cản tuyệt vời của cải: chúng có thể được thực hiện với vật liệu như PVC/PVDC hoặc giấy bạc (ALU-PVC, Mọi thời đại) để chặn độ ẩm, oxy và ánh sáng. Ví dụ, Máy đóng vỉ DPP-180Pro của Jinlu có thể tạo hình và hàn kín tối đa 4,800 vỉ nang mỗi giờ trong tấm Alu-PVC hoặc Alu-Alu. Các tính năng chính của bao bì vỉ viên nang bao gồm:
Nhân vật:Máy đóng gói vỉ Jinlu JL-180Pro tạo thành vỉ nang (ALU-PVC), 4,800 sâu răng/giờ.
Máy đóng vỉ của Jinlu (ví dụ. DPP-180Pro) có thể tự động hình thành sâu răng, viên nang thức ăn, niêm phong, tấm cắt, và thậm chí in mã lô. Họ thường chạy ở tốc độ trung bình cao (hàng ngàn sâu răng mỗi giờ), lý tưởng cho sản xuất hàng loạt. Các dạng vỉ khác nhau (Alu-PVC và Alu-Alu) được lựa chọn theo sản phẩm: Mọi thời đại (giấy bạc) đưa ra rào cản cao nhất (tốt nhất cho các loại thuốc rất nhạy cảm với độ ẩm), trong khi PVC/PVDC thường dùng cho viên nang tiêu chuẩn. Đóng gói vỉ cũng đơn giản hóa việc đếm và kiểm tra: hầu hết các dây chuyền đều dễ dàng tích hợp máy kiểm tra camera hoặc máy đếm viên để xác minh việc nạp viên nang.
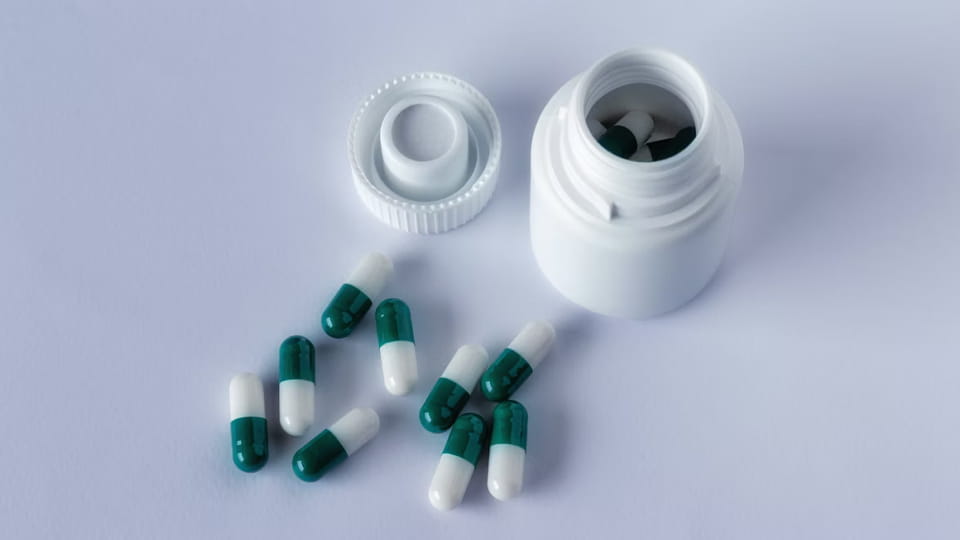
Chai là loại hộp đựng đa liều cổ điển. Viên nang được đổ vào chai (nhựa hoặc thủy tinh) sau đó được giới hạn. Định dạng này nổi tiếng với vitamin, chất bổ sung, và nhiều loại thuốc theo toa. Các tính năng chính:
Nhân vật: Dây chuyền đóng chai viên nang/viên nén Jinlu JL-16H (100 chai/phút) với trình giải mã, que, capper, và người dán nhãn.
Dây chuyền đóng chai viên nang tích hợp nhiều máy: Một Chai Unscrambler (hoặc phễu thức ăn), Một máy đếm điền (thả viên X vào mỗi chai), Một máy đóng nắp/niêm phong (áp dụng và thắt chặt nắp), và một máy dán nhãn. Ví dụ, Dòng JL-16H của Jinlu có thể xử lý viên nang 3–40 mm, đạt được ~100 chai/phút với >99.8% Đếm độ chính xác. Chai có thể được đổ đầy với số lượng lớn rất nhanh. Để đáp ứng quy định, Các dòng chai thường có một trạm để nạp chất hút ẩm (ví dụ. gói silica) và một trạm niêm phong chống giả mạo. Mũ chống trẻ em là một tính năng phổ biến khác của thuốc dành cho trẻ em.
Trong việc lựa chọn bao bì chai, xem xét khối lượng sản phẩm và độ ổn định: sản phẩm có số lượng lớn (ví dụ. vitamin hàng ngày) chai phù hợp với, trong khi viên nang có thể tích thấp hoặc có độ nhạy cao có thể thích dạng phồng rộp hơn. Lưu ý chai thường cung cấp khả năng bảo vệ độ ẩm/oxy vốn có ít hơn so với các vết phồng rộp có rào cản cao. Thủy tinh màu hổ phách hoặc hộp đựng mờ đục có thể bảo vệ ánh sáng, và con dấu bên trong (lót lá nhôm) có thể cải thiện rào cản.

Gói (và gói dính) là gói linh hoạt chứa một liều bột, hạt hoặc chất lỏng. Mặc dù không phổ biến đối với viên nang, chúng có liên quan đến các dạng bào chế liên quan (ví dụ. viên nang dinh dưỡng vỡ thành bột, hoặc bột độn). Tuy nhiên, gói và gói dính xứng đáng được đề cập trong hướng dẫn đóng gói viên nang:
Cả gói và gói dạng que đều được sản xuất bởi dấu điền theo chiều dọc (VFF) hoặc máy FFS ngang. Dòng JL-VP của Jinlu, ví dụ, là máy FFS dạng thanh/gói nhiều làn (40 túi/phút/làn, Độ chính xác ± 0,02g) cho bột, hạt, chất lỏng hoặc gel. Nếu viên nang được đóng gói trong túi, người ta sẽ mở gói để lấy liều thuốc – nhưng thông thường hơn, viên nang được đóng gói nguyên vẹn trong vỉ hoặc chai, trong khi gói/gói thanh xử lý nội dung dạng bột.
Lưu ý quy định: Được phép dùng túi và gói dạng que đối với thuốc dạng rắn và dạng bán rắn (theo định nghĩa của WHO). Chúng được coi là bao bì chính nếu chúng chứa trực tiếp sản phẩm. Giống như mụn nước, gói có thể được thiết kế để chống trẻ em hoặc có vết rách.
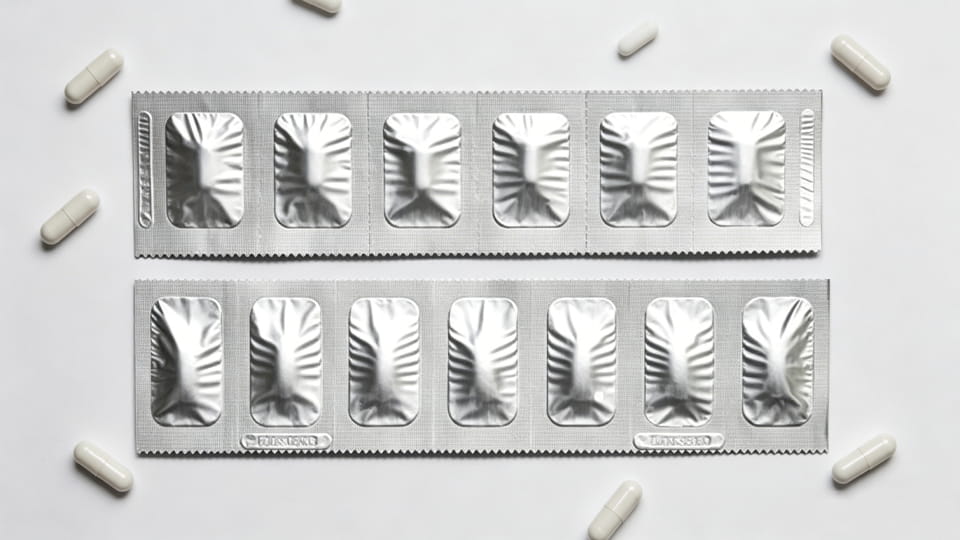
Gói dải (còn được gọi là dải giấy bạc hoặc “dải liều”) là một dạng khác của gói chính theo đơn vị liều. Một dải thường là một mạng lưới vật liệu hẹp (thường là nhôm hoặc nhiều lớp) gấp lại và niêm phong, có thủng giữa các liều. Khác với mụn nước, gói dải không tạo thành khoang sâu – thay vào đó, viên nang được kẹp giữa hai lớp phim. Gói dải được sử dụng rộng rãi ở châu Á và dành cho các sản phẩm cần bảo vệ độ ẩm cao nhưng vỉ quá cồng kềnh. Điểm mấu chốt:

Từ góc độ lựa chọn, gói dải chiếm một khoảng trống giữa chai (số lượng lớn) và phồng rộp (độ cứng). Chúng thường được chọn khi rào cản độ ẩm rất cao là cần thiết nhưng trong một mỏng, hình thức nhẹ. (Ghi chú: gói dải có thể không cung cấp bằng chứng giả mạo rõ ràng như vỉ trừ khi sử dụng con dấu bổ sung.)
Sau khi chọn gói chính, viên nang thường được đặt vào bao bì thứ cấp như hộp carton hoặc bọc co lại trường hợp. Thùng carton cung cấp thêm sự bảo vệ trong quá trình vận chuyển, cho phép ghi nhãn bổ sung (thông tin thuốc, Thương hiệu, con dấu rõ ràng giả mạo), và giúp tổ chức các sản phẩm nhiều gói. Những cân nhắc chính cho bao bì thứ cấp:
Tóm lại, bao bì thứ cấp bao bọc sản phẩm để phân phối lần cuối và đảm bảo có sẵn tất cả các tính năng ghi nhãn và tuân thủ. Thiết kế tốt ở đây nâng cao hình ảnh thương hiệu và sự thuận tiện cho người dùng (thùng carton dễ mở, ghi nhãn rõ ràng).
Việc lựa chọn vật liệu đóng gói phù hợp là rất quan trọng để bảo vệ viên nang. Chất liệu phải tương thích với thuốc, đáp ứng nhu cầu rào cản, và tuân thủ các quy định (ISO 15378 GMP cho vật liệu đóng gói, vân vân.). Vật liệu phổ biến bao gồm:
Nói chung là, chọn rào cản cao nhất có ý nghĩa cho viên nang của bạn. Ví dụ, viên nang gelatin nhạy cảm với độ ẩm có thể cần có vỉ Alu-Alu hoặc chai thủy tinh màu hổ phách có chất hút ẩm. Ngược lại, viên nang không nhạy cảm có thể sử dụng vỉ PVC hoặc chai HDPE tiêu chuẩn. Cũng xem xét thời hạn sử dụng: sản phẩm dài hạn cần rào cản mạnh mẽ hơn.
Ghi chú về an toàn/tái chế: Xu hướng bền vững ủng hộ các gói vật liệu đơn sắc (ví dụ. vỉ PP, chai PET tái chế). Vật liệu thử nghiệm cho chất chiết xuất, khả năng tương thích, và đáp ứng các giới hạn của FDA/EU (ví dụ. USP <661> cho chất dẻo) là một phần của việc tuân thủ bao bì.
Viên nang có nhiều loại khác nhau và công thức của chúng quyết định nhu cầu đóng gói:
Tóm lại, các vật liệu vỏ viên nang và loại điền sẽ ảnh hưởng đến việc lựa chọn bao bì. Công thức nhạy cảm với độ ẩm cao luôn ưu tiên hàng rào chặt chẽ nhất (Vỉ Alu-Alu, gói giấy bạc, hoặc chai kín có hút ẩm). Công thức ít nhạy cảm hơn có nhiều lựa chọn hơn. Luôn kiểm tra độ ổn định của viên nang đóng gói cuối cùng (ICH Q1A yêu cầu sự ổn định trong thùng chứa được đề xuất).
Việc lựa chọn bao bì viên nang tối ưu liên quan đến việc đánh giá nhiều yếu tố: dạng bào chế, Tuân thủ bệnh nhân, độ nhạy sản phẩm, hạn sử dụng, yêu cầu quy định, và chi phí. Dưới đây là một khung quyết định đơn giản hóa:
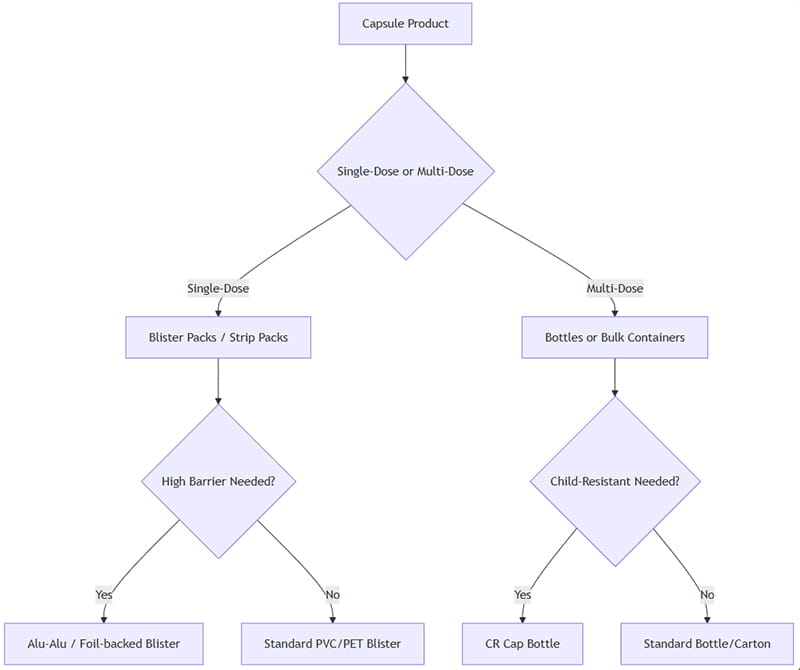
Làm thế nào để áp dụng điều này: Nếu viên nang được dự định là đơn vị riêng lẻ (ví dụ. liều lượng chặt chẽ hoặc thẻ vỉ bán lẻ), vỉ/dải được ưa thích. Đối với đa liều (ví dụ. thuốc bổ sung hoặc thuốc có công dụng cao), chai hiệu quả hơn. Nếu việc bảo vệ độ ẩm/ánh sáng là rất quan trọng, chọn các tùy chọn có rào cản cao (Mọi thời đại, chai hổ phách). Nếu cần sự an toàn cho trẻ em, sử dụng kiểu đóng chai chống trẻ em hoặc thiết kế vỉ chống giả mạo.
Các tiêu chí khác của ngành bao gồm:
Cuối cùng, thử nghiệm thí điểm (nghiên cứu độ ổn định, nghiên cứu người tiêu dùng) và phân tích tổng chi phí sẽ hướng dẫn quyết định cuối cùng. Sơ đồ trên là điểm khởi đầu cho các kịch bản điển hình.
Đóng gói viên nang cần có máy móc chuyên dụng. Sau khi viên nang được lấp đầy (bằng chất độn viên nang), chúng được chuyển đến dây chuyền đóng gói:
Tuân thủ thiết bị: Tất cả các máy đóng gói dược phẩm phải đạt tiêu chuẩn thiết kế GMP (Thép không gỉ, bề mặt có thể làm sạch, hiệu suất được xác nhận). Máy của Jinlu được chứng nhận CE/cGMP, thường có các hệ thống bảo vệ và loại bỏ có thể cấu hình được đối với những viên thuốc không đạt tiêu chuẩn kỹ thuật. Tích hợp là chìa khóa: Ví dụ, một máy ép vỉ có thể nạp trực tiếp vào một thùng carton để dựng các hộp và đóng gói từng thẻ vỉ. Hệ thống toàn diện (từ điền đến hộp cuối cùng) cho phép tự động hóa từ đầu đến cuối việc đóng gói viên nang.
Bao bì viên nang cho dược phẩm phải tuân thủ quy định nghiêm ngặt. Các điểm chính bao gồm:
Tổng thể, cuộc họp FDA, Hướng dẫn của EMA và WHO là cần thiết. Nhiều nhà đóng gói làm việc với các nhà cung cấp được quản lý (như Jinlu) người có thể cung cấp máy móc và tài liệu đã được xác nhận (IR/WH/PQ).
Lựa chọn bao bì viên nang phù hợp đòi hỏi phải có sự bảo vệ cân bằng, khả năng sử dụng, và chi phí. Gói vỉ, chai, gói, và mỗi dải đều có vị trí của chúng bao bì dược phẩm. Các yếu tố như độ nhạy ẩm, dạng liều, và nhu cầu của bệnh nhân hướng dẫn quyết định. Bên cạnh, phải đáp ứng các yêu cầu nghiêm ngặt về quy định và GMP.
Tại Đóng gói Jinlu, chúng tôi chuyên về các giải pháp đóng gói dược phẩm trọn gói. Thiết bị của chúng tôi – từ máy ép vỉ tốc độ cao ĐẾN đếm hoàn toàn tự động & dây chuyền đóng chai – giúp bạn đóng gói viên nang hiệu quả và tuân thủ. Chúng tôi cũng ủng hộ những lựa chọn bền vững (ví dụ. máy chạy màng tái chế mới) và cung cấp dịch vụ một cửa (tùy biến, xác nhận). Nhóm của chúng tôi có thể giúp bạn đánh giá hệ thống đóng gói viên nang tốt nhất cho sản phẩm của bạn.
Sẵn sàng tối ưu hóa bao bì viên nang của bạn? Liên hệ với Jinlu Đóng gói ngay hôm nay để được hướng dẫn và báo giá từ chuyên gia. Các giải pháp của chúng tôi được hỗ trợ bởi các tiêu chuẩn chất lượng toàn cầu và hơn thế nữa 3,000 khách hàng hài lòng trên toàn thế giới. Hãy đóng gói viên nang của bạn để thành công!
Bao bì viên nang đề cập đến quá trình bao bọc viên nang dược phẩm trong vật liệu bảo vệ để duy trì sự ổn định, sự an toàn, và tuân thủ. Các định dạng phổ biến bao gồm gói vỉ, chai, và đóng gói dạng dải.
Trong thực hành dược phẩm, Bao bì không chỉ có tác dụng ngăn chặn—nó bảo vệ viên nang khỏi bị ẩm, ôxy, và ô nhiễm, ảnh hưởng trực tiếp đến chất lượng và thời hạn sử dụng của thuốc.
Không có lựa chọn “tốt nhất” duy nhất - nó phụ thuộc vào sản phẩm và thị trường của bạn.
• Đóng gói theo vỉ → tốt nhất cho các loại thuốc nhạy cảm với độ ẩm hoặc có giá trị cao
• Đóng gói dạng chai → tốt nhất để bảo quản số lượng lớn và bổ sung
• Bao bì dạng dải → phổ biến ở các thị trường nhạy cảm về chi phí
Đóng gói theo vỉ thường được ưa chuộng hơn vì nó mang lại khả năng bảo vệ theo liều lượng và cách ly môi trường tốt hơn..
Sử dụng các tài liệu ngăn chặn các mối đe dọa chính (độ ẩm, ôxy, ánh sáng). Ví dụ, Vỉ nhôm Alu-Alu hoặc chai thủy tinh màu hổ phách mang lại rào cản tuyệt vời cho viên nang nhạy cảm với độ ẩm. Vỉ PVC hoặc PET có lớp phủ PVDC là phổ biến và rẻ hơn cho các sản phẩm ổn định. Chai HDPE là loại tiêu chuẩn nhưng có thể cần túi hút ẩm nếu viên nang nhạy cảm. Đánh giá tốc độ truyền hơi nước (WVTR) của bộ phim. Trong sản xuất có ý thức bền vững, xem xét các vật liệu đơn có thể tái chế (ví dụ. vỉ PP). Luôn kiểm tra khả năng tương thích của bao bì thông qua các nghiên cứu về độ ổn định.
Viên nang rất nhạy cảm với các yếu tố môi trường như độ ẩm, ôxy, và ánh sáng.
Bao bì phù hợp:
• Ngăn chặn sự hấp thụ độ ẩm
• Giảm quá trình oxy hóa
• Duy trì sự ổn định hóa học
Bao bì vỉ, đặc biệt, tạo ra môi trường vi mô kín cho mỗi viên nang, kéo dài đáng kể thời hạn sử dụng.
Nó phụ thuộc vào định dạng. Đối với mụn nước, bạn cần một máy tạo hình/hàn kín vỉ (với khuôn khoang để phù hợp với viên nang của bạn) cộng thêm có thể là một thùng carton. Đối với chai, bạn cần dây chuyền chiết rót/đóng nắp chai (người giải mã, chất phản ứng, capper, Labolder). Đối với gói dính, sử dụng máy đóng gói dạng dọc. Jinlu cung cấp tất cả những thứ này: ví dụ. vỉ dòng DPP của chúng tôi, Dây đếm JL-16H, và máy đóng gói gói JL-VP. Lựa chọn máy phù hợp liên quan đến công suất, Kích thước viên nang, và tích hợp cần thiết (hệ thống tầm nhìn, trạm từ chối, vân vân.).
Xem xét các yếu tố như liều lượng (độc thân vs. nhiều), nhu cầu ổn định, và bệnh nhân sử dụng. Để thuận tiện khi sử dụng liều đơn và mang lại hiệu quả bảo vệ cao (đặc biệt đối với viên nang nhạy cảm với độ ẩm/ánh sáng), mụn nước thường được ưa thích. Vì lý do số lượng lớn hoặc kinh tế, chai thường được chọn. Cũng, nếu cần các tính năng thân thiện với trẻ em hoặc người già, chai có thể cung cấp nhiều lựa chọn hơn. Như một quy luật, kết hợp các đặc tính của viên nang với bao bì: ví dụ. viên nang hút ẩm → vỉ Alu-Alu; nhu cầu bổ sung cao → chai.
Tài liệu tham khảo:
1.Phụ lục 9 Hướng dẫn về bao bì đối với dược phẩm -- AI
2.Yêu cầu kỹ thuật đối với thuốc và sản phẩm y tế -- Tổ chức Y tế Thế giới
3.Vật liệu đóng gói dược phẩm và an toàn thuốc: Một đánh giá nhỏ —— MDPI
4.Bao bì và phụ kiện dược phẩm --- Thư viện Y khoa Quốc gia
5.Đánh giá về đặc tính và khả năng tái chế của vỉ dược phẩm —— Khoa học trực tiếp

Petty fu, Người sáng lập Jinlupacking, mang lại 20 năm kinh nghiệm trong lĩnh vực máy móc dược phẩm. Dưới sự lãnh đạo của ông, Jinlu đã phát triển thành một nhà cung cấp thiết kế tích hợp đáng tin cậy, sản xuất, và bán hàng. Petty đam mê chia sẻ kiến thức chuyên sâu về ngành của mình để giúp khách hàng giải quyết sự phức tạp của bao bì dược phẩm, đảm bảo họ không chỉ nhận được thiết bị, mà là mối quan hệ đối tác dịch vụ một cửa thực sự phù hợp với mục tiêu sản xuất của họ.