
In pharmaceutical manufacturing, a tablet defect is any flaw in a pill that makes it fall short of quality standards. This could be a chip on the edge, a crack through the surface, an uneven coating, or even a misprint. In simple terms, a defective tablet isn’t quite “right” – it might break easily, look odd, or have the wrong dose. Tablet defects can happen for many reasons (improper ingredients, machine issues, human error, etc.). For example, even with modern equipment and strict GMP rules, problems like cracks, chips, poor coating, or printing errors still occur.
Millions of tablets roll off production lines every day, and each one must meet tight specs. A single flaw can have big consequences: an entire batch may be scrapped, or worse, recalled. In fact, any tablet with a physical flaw usually cannot be shipped to patients. FDA data shows about 24% of all drug recalls involve manufacturing quality issues. Recalls are expensive, too – industry reports estimate a single pharma recall can cost $10–100 million or more. Most importantly, defects risk patient safety and trust. Imagine a patient getting a cracked pill that splits in two (with no dose guarantee) or a tablet with unapproved specks of material on it. Clearly, manufacturers must catch these defects early.

Here are some of the most common tablet defects that crop up during production. Each defect has typical causes, which we’ll cover in the next section. Understanding them helps everyone spot and prevent problems on the line:
These defects are the tip of the iceberg of what can go wrong. The key takeaway is that any visual flaw – cracks, chips, layers, discoloration, unevenness – should be investigated. Tablet defects often indicate a problem in production.
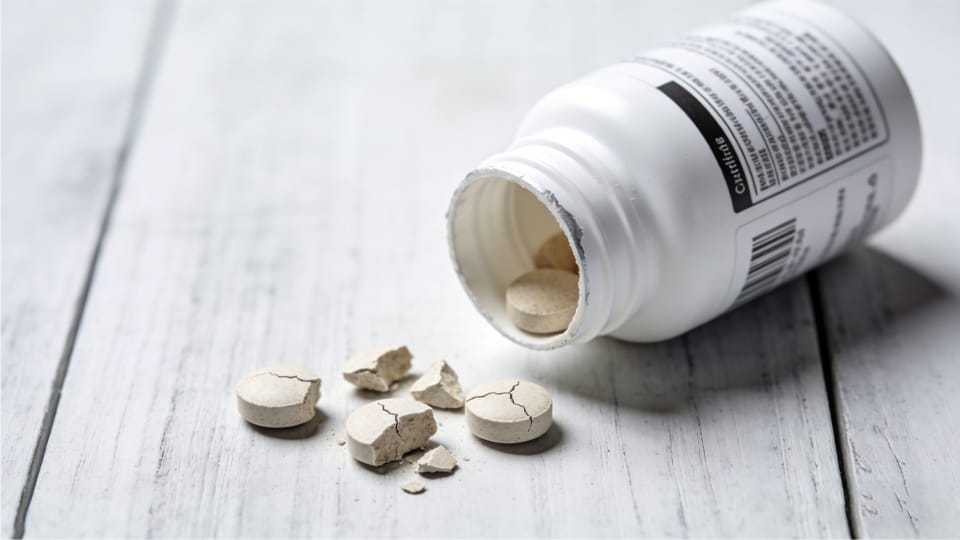
Tablet defects aren’t just cosmetic – they signal bigger risks. A defect means the manufacturing process didn’t go exactly as planned, which can affect dose accuracy, drug release, or stability. For patients, a flawed tablet might deliver too little (or too much) medicine, or have altered efficacy. In worst cases, quality issues can harm patients. For manufacturers, defects mean waste and cost. Defective tablets often can’t be sold, forcing costly rework or scrap. For example, one industry report notes that one recall can cost a drug maker $10–100 million. That same report said a single warranty/recall event in life sciences could run as high as $600 million when litigation and indirect costs are included.
Regulators take defects seriously. The FDA’s quality reports show that almost a quarter of all drug recalls are tied to manufacturing or CGMP failures. In other words, manufacturing quality (like avoiding tablet defects) is directly linked to recalls and fines. Patient safety, company reputation, and the bottom line are all at stake.
Put simply: defective tablets waste money and put patients at risk. As one industry expert explains, skipping proper maintenance or quality checks “will result in defective tablets and ultimately lead to a complete loss of the batch”. In practice, that means a bad batch must be destroyed before it ever ships to patients. No company wants that.
Behind every defect is one or more root causes. The tableting process has many steps, so issues can arise from materials, formulation, machines, or human factors. Common causes include:
In summary, most defects come from either the materials/formulation side or the process/equipment side. For instance, capping often results from trapped air or elastic recovery in the granules, whereas chipping often points to tooling or lubrication problems. Identifying the root cause usually involves looking at both the recipe and the machine setup.

The good news is that defects can be greatly reduced with proper practices. Here are key strategies industry pros use:
In practice, combining these measures is most effective. For example, using an AI vision system or automated inspection ensures every tablet is checked (instead of just a small sample). Likewise, following a Quality by Design (QbD) approach at the development stage means the formulation itself is optimized to be robust, which prevents defects down the line. The bottom line is to build quality in at each step, rather than try to inspect it in later.
To put it in perspective, tablets are the most common drug form worldwide – they’re cheap, stable, and convenient. A single large pharma facility might run hundreds of thousands of tablets an hour. With such volume, even a tiny defect rate can mean thousands of bad pills. That’s why big companies invest heavily in automation and inspection.
All these factors mean that preventing tablet defects is critical. Not only does it protect patients, it also saves companies from massive losses.
Tablet defects – whether it’s a tiny chip, a split layer, an off-color spot, or an imprint error – are a headache for any pharma manufacturer. But understanding what they look like and why they happen is the first step to eliminating them. In summary: identify common defects early, find and fix the root cause (ingredient or process), and build strong quality checks at every step.
If your factory is struggling with tablet quality, you don’t have to go it alone. At Jinlu Packing, we specialize in pharmaceutical production and packaging equipment designed for precision. Our high-performance tablet presses, coating machines, and inspection systems are engineered to minimize defects (through uniform compression, gentle handling, and vision inspection).
Ready to ensure defect-free tablets? Contact us for a quote tailored to your needs. Our team can recommend equipment or process upgrades to keep your tablets consistent and your lines running smoothly. Don’t let tablet flaws cost you time and money – let’s work together to produce perfect pills.
A tablet defect refers to any visible or physical flaw in a tablet that prevents it from meeting quality standards. These defects can include cracks, chipped edges, uneven color, sticking surfaces, or incorrect weight. Such defects usually occur during compression, coating, or handling processes in pharmaceutical manufacturing and may affect the tablet’s appearance, stability, or dosage accuracy.
Some of the most common defects in tablet manufacturing include:
• Capping
• Lamination
• Sticking
• Picking
• Chipping
• Cracking
• Weight variation
• Mottling (uneven color)
These defects often occur due to formulation issues, improper machine settings, or environmental factors during tablet production.
Capping happens when the top or bottom layer of a tablet separates from the body, while lamination refers to the tablet splitting into multiple layers.
The main causes include:
• Air trapped in the powder during compression
• Insufficient compression force
• Poor granule binding
• Excessive press speed
Proper compression settings and optimized granulation can significantly reduce these defects.
Sticking occurs when the tablet material adheres to the punch face or die wall during compression. This defect often results in rough surfaces or damaged tablet logos.
Common causes include:
• Excess moisture in the formulation
• Inadequate lubrication
• Rough or worn punches
• Sticky active ingredients
Maintaining proper lubrication and using polished tooling can help prevent sticking issues.
Both defects involve tablet material adhering to tooling surfaces, but they occur in slightly different ways:
• Sticking: Powder sticks to punch faces or die walls.
• Picking: Material is pulled away from the tablet surface, usually around embossed logos or lettering.
Picking is generally considered a specific type of sticking that occurs on engraved punch designs.
Tablet formulation plays a major role in defect formation. Poorly designed formulas may contain:
• Too little binder
• Too much moisture
• Excess fine particles
• Poorly flowing granules
These issues can lead to problems like capping, lamination, sticking, or weight variation during compression.
• Optimize formulation and granulation quality
• Maintain correct compression force and press speed
• Ensure proper lubrication of punches and dies
• Control humidity and temperature in the production area
• Conduct regular equipment maintenance and inspection
Advanced tablet press machines and automated inspection systems can also help detect defects early in the process.
Preventing tablet defects requires controlling several factors during production:
Tablet weight variation occurs when individual tablets in a batch have inconsistent mass. This can lead to inaccurate dosage and regulatory compliance issues.
Common causes include:
• Poor powder flow
• Uneven granule distribution
• Incorrect feeder settings
• Improper mixing
Maintaining consistent material flow into the die cavity is essential to avoid this defect.
Yes. Tablet defects can impact drug stability, dose accuracy, and patient safety. For example, cracked or chipped tablets may break apart during packaging or transport, while weight variation can lead to incorrect dosing. Because of this, pharmaceutical manufacturers must follow strict quality control standards and inspection procedures.
Modern tablet press machines are designed to reduce defects through:
• Precise compression force control
• Stable turret speed
• Improved granule feeding systems
• Automated monitoring of tablet weight and hardness
High-quality equipment ensures consistent compression and better process control, which helps pharmaceutical manufacturers minimize tablet defects and improve production efficiency.
References:
1. MANUFACTURING DEFECTS OF TABLETS – A REVIEW — Journal of Drug Delivery and Therapeutics
2.Tablet’s Common Defects and Solution — Journal of Chemical and Pharmaceutical Research
3.Understanding tablet defects in commercial manufacture and transfer – ScienceDirect
4.Exploring the Impact of Formulation and Tablet Shape on Tablet Integrit – Journal of Pharmaceutical Sciences (2025)

Petty Fu, Founder of Jinlupacking, brings over 30 years of expertise to the pharmaceutical machinery sector. Under his leadership, Jinlu has grown into a trusted supplier integrating design, production, and sales. Petty is passionate about sharing his deep industry knowledge to help clients navigate the complexities of pharma packaging, ensuring they receive not just equipment, but a true one-stop service partnership tailored to their production goals.
Copyright © 2026 JinLuPacking.All rights reserved. Terms & Conditions and Privacy Policy
Friendly Links: Rich Packing | Capsule Filling Machine Manufacturers