
What is Unit Dose Packaging? In simple terms, unit dose packaging (also called single-dose packaging) means each individual dose of a medication is pre-packaged and sealed by itself. Imagine one pill or tablet placed in its own little pouch or blister cavity, with all the drug information printed right on that package. Each package contains exactly one dose, so you don’t have to count pills from a bottle. This approach is very common in hospitals and pharmacies: for example, over 75% of oral medications in U.S. hospitals are dispensed in unit dose form. In other words, a “medication unit dose” is one dose of medicine ready for immediate use. This packing style is about safety and convenience – each dose is sealed and labeled, ready for a single use.
Unit dose packaging has grown in popularity because it helps healthcare providers give the right dose at the right time. It prevents mix-ups, since each package is clearly marked and contains only one dose. Think of it like the difference between bulk packaging (a bottle of 100 tablets) and a unit dose pack (a small blister with one tablet). With unit dose packs, nurses and patients don’t have to count out pills – they just grab one package and open it. This also reduces handling errors. In fact, studies show that properly labeled single-dose packs greatly cut down medication errors.

Unit dose formats may sound simple, but they offer big benefits for pharmaceutical producers and healthcare users. Here are some of the key advantages:
These perks make unit-dose packaging a must-have in many settings – hospitals, long-term care, and even consumer health products. According to one source, unit-dose systems were originally created for hospitals to ensure safe, efficient drug distribution. Over time they’ve “vastly expanded” into other industries (like cosmetics or retail supplements) because of their safety features. Today, unit dose packs are prized by pharmacists, nursing staff, and procurement managers alike for their ability to simplify inventory control and reduce errors.
There are several popular ways to pack a single dose, depending on the product’s form (pill, powder, liquid, etc.). Below are the most common unit-dose formats used in pharmaceuticals and related fields:
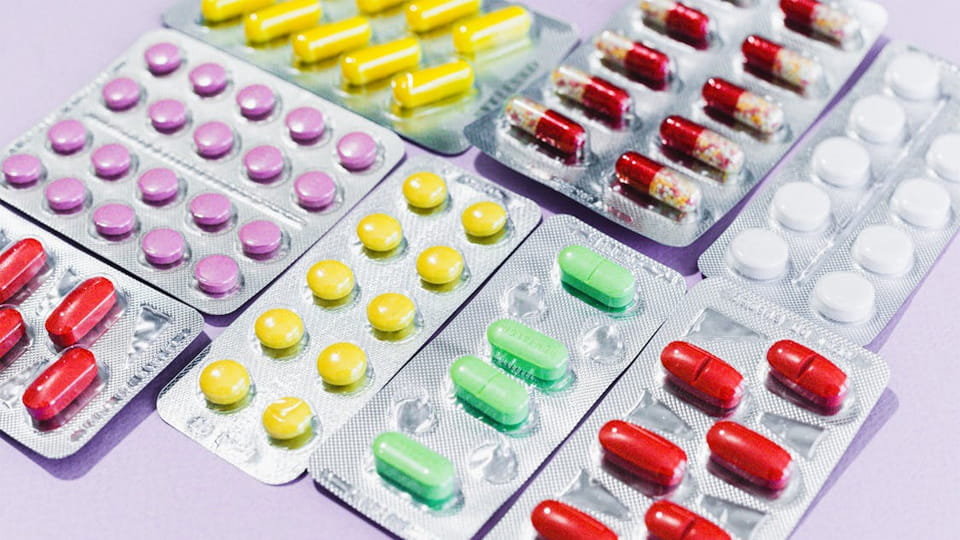


Each of these formats keeps one dose isolated. For example, calendar packs are special blister cards arranged by day/time, still a unit-dose concept – each blister is one dose arranged in a schedule. In all cases, the core idea is the same: individual, tamper-evident packaging for each dose.
Quick Comparison: In general, tablets/capsules use blister packs or strips, powders/liquids use sachets or small bottles, and injectables use single-dose vials/ampoules. The choice depends on the drug’s physical form and stability needs. Often, the unit dose packaging is the primary packaging (the container that touches the drug) and is complemented by a secondary carton or box with labels and instructions.
Units of Use: Some products combine multiple single doses into a strip or card, but each unit is still single-use. For instance, unit-dose blister cards might hold 7 daily doses, but each day’s dose is sealed separately. Likewise, a unit-dose pouch can contain a ready-to-mix medicine, which is still one dose. The key is that at dispensation time, each dose is separable and used one at a time.
Behind every unit-dose package there is specialized machinery that forms, fills, and seals the dose. Here’s an overview of the types of machines used to make unit dose packs:
Manufacturers often refer to these machines collectively as unit-dose packaging machines or blister machines, sachet machines, vial filling lines, etc. For instance, a pharmaceutical OEM might advertise a “blister packaging line for tablets/capsules” or a “stick pack machine for unit-dose powder”. The key is that each of these machines is set up to ensure one dose per package.
Choosing the right machine: The choice depends on the product:
For example, JinLu Packing notes that blister packs are made on blister packing machines, and stick or sachet packs are made on sachet packaging machines. In practice, a factory producing unit-dose packs will have a packaging line that may combine multiple machines (forming/sealing machine + labeling + cartoning, etc.) to go from raw dose to finished packaged unit.
When selecting a unit dose format and machine, consider these factors (bullet points for key tips):
In summary, match the type of unit pack to your product, and choose a machine that can reliably handle it. Many machine vendors (like Jinlu Packing, Bosch, etc.) offer unit-dose solutions – just be sure to describe your dose form (tablet, vial, sachet, etc.) and required output.
Unit dose packaging is all about one-at-a-time medication safety. By pre-packaging each single dose, manufacturers and pharmacies can improve accuracy, hygiene, and convenience. The most common unit-dose formats are blister packs, strip packs, sachets, and single-use bottles/vials. Each format requires specialized equipment – for example, blister packaging machines, form-fill-seal (sachet) machines, and vial fillers.
If you’re in the pharmaceutical or supplement industry, understanding unit-dose packaging is crucial. It can help your customers (hospitals, clinics, patients) use medications correctly. And if you manufacture packaging machines, offering the right unit-dose solutions can give you a competitive edge.
Key Takeaway: Unit Dose Packaging (also called single-dose packaging) means packaging each pill, tablet, or dose individually. It comes in several types (blisters, strips, sachets, vials, etc.), and requires matching packaging machines. The result is safer, error-proof dosing that modern buyers and regulators increasingly expect.
Unit dose packaging refers to the method of packaging each individual medication dose in a separate sealed unit, like a blister pocket, sachet, vial, or strip. Each package contains exactly one pre-measured dose that can be administered directly, which helps protect the drug’s integrity and reduce errors.
Unit dose packaging enhances medication safety and accuracy, protects doses from contamination, makes dosing convenient, and supports efficient inventory management in hospitals, clinics, pharmacies, and home care.
The most common types include:
• Blister packs – plastic cavities sealed with foil for tablets and capsules.
• Strips – laminated strip packs containing single pills.
• Sachets/Stick packs – for powders or granules.
• Single-use vials or ampoules – for liquids and injectables.
• Pre-filled syringes – ready for injection.
A blister unit dose pack is a type of packaging where each tablet or capsule sits in its own cavity (blister), sealed with foil or film. This keeps each dose separate, clean, and easy to identify and dispense.
Because each dose is individually labeled and sealed, there’s less risk of miscounting or mixing up medications. Clear labeling and dose separation help ensure patients and caregivers give the correct dose at the correct time.
Different machines are used depending on the packaging type:
• Blister packaging machines for tablets and capsules.
• Vertical form-fill-seal machines for sachets/stick packs.
• Vial/ampoule filling and capping machines for liquid doses.
• Pre-filled syringe assembly lines for injection doses.
These machines form, fill, seal, and prepare unit doses efficiently.
No. While it’s most common in pharmaceuticals and hospitals, unit dose concepts are also used in other industries such as nutraceuticals, cosmetics, personal care, food sampling, and industrial liquids where precise single-use dosing adds value.
Patients benefit from accuracy, convenience, portability, reduced contamination, easier compliance tracking, and clearer labeling of each dose – making medicine regimens simpler to follow.
Yes. Because each dose is separated and labeled, hospitals and pharmacies can better track medication usage, control stocks, and reduce waste or leftover unused doses.
Yes. Sealed individual units can protect medicines from moisture, oxygen, and light, helping maintain potency and extend shelf life until the moment of use.
References:
1. Unit dose packaging definition — Law Insider
2.Single Unit and Unit Dose Packages of Drugs — ASHP Technical Assistance Bulletin
3.Unit Dose Packaging System – Definitive Healthcare
4.Recent trends and future of pharmaceutical packaging technology – National Library of Medicine

Petty Fu, Founder of Jinlupacking, brings over 30 years of expertise to the pharmaceutical machinery sector. Under his leadership, Jinlu has grown into a trusted supplier integrating design, production, and sales. Petty is passionate about sharing his deep industry knowledge to help clients navigate the complexities of pharma packaging, ensuring they receive not just equipment, but a true one-stop service partnership tailored to their production goals.
Copyright © 2026 JinLuPacking.All rights reserved. Terms & Conditions and Privacy Policy
Friendly Links: Rich Packing | Capsule Filling Machine Manufacturers