
The evolution of the pharmaceutical tablet from a rudimentary compressed powder to a sophisticated delivery vehicle represents one of the most significant engineering achievements in modern medicine. In the globalized world of pharmaceutical manufacturing, a tablet is no longer just a carrier for an active pharmaceutical ingredient (API); it is a complex intersection of material science, mechanical engineering, and patient psychology. For industry buyers and procurement engineers, understanding the multifaceted nature of pharmaceutical tablet design is essential not only for therapeutic efficacy but also for ensuring the long-term viability and profitability of a production line.
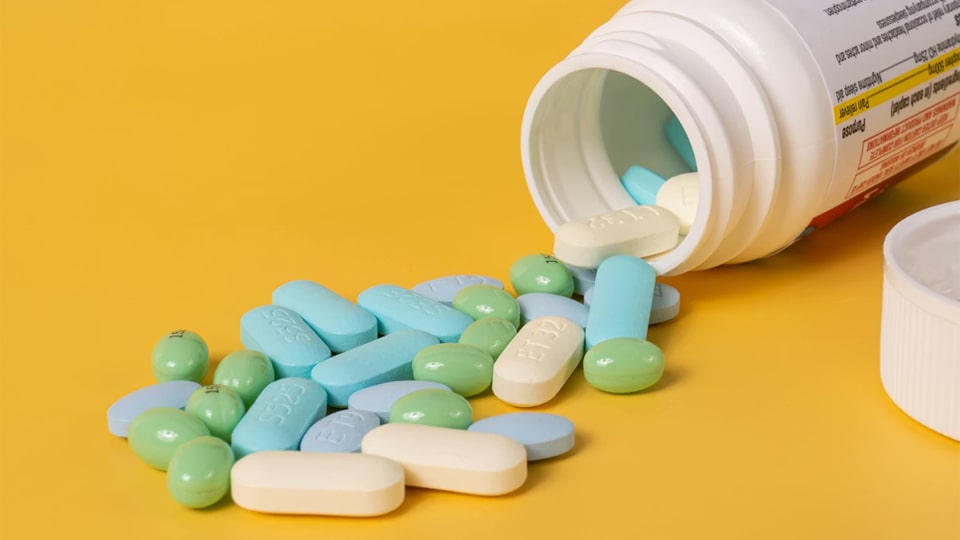
The design of a pharmaceutical tablet serves as the primary interface between the drug and the patient. It is a critical component of the “tablet pharmacy” ecosystem, where the physical form of the medication dictates everything from patient compliance to the mechanical limits of a high-speed rotary press. Successful design requires a holistic approach that balances the clinical needs of the patient with the industrial realities of high-volume manufacturing.
In the contemporary landscape, manufacturers are increasingly adopting Quality by Design (QbD) principles. This methodology emphasizes that quality must be built into the product during the design phase rather than tested into the final product after production. By identifying Critical Quality Attributes (CQAs) such as hardness, disintegration, and dissolution early in the process, engineers can create robust formulations that withstand the rigors of the compression cycle.
A critical first question in tablet design is size and shape. Tablets must be easy to swallow and safe for patients. In general, smaller is better: minimizing tablet diameter (e.g. under 22 mm, and ideally close to 8 mm) makes swallowing easier and improves patient compliance. Large, square, or irregularly shaped tablets can be hard to swallow and even pose a choking hazard. Rounded or oval shapes with smooth edges are preferred. If your drug dose requires a big pill, consider splitting the dose into two smaller tablets or using a special effervescent form.
Finding the right balance between dose and size often depends on the drug’s compressibility. If your active ingredient is low-dose or highly compressible, you may form a compact pill even in a small tablet. For high-dose or poorly compressible drugs, granulation or special binders might be needed to avoid making the tablet too large.
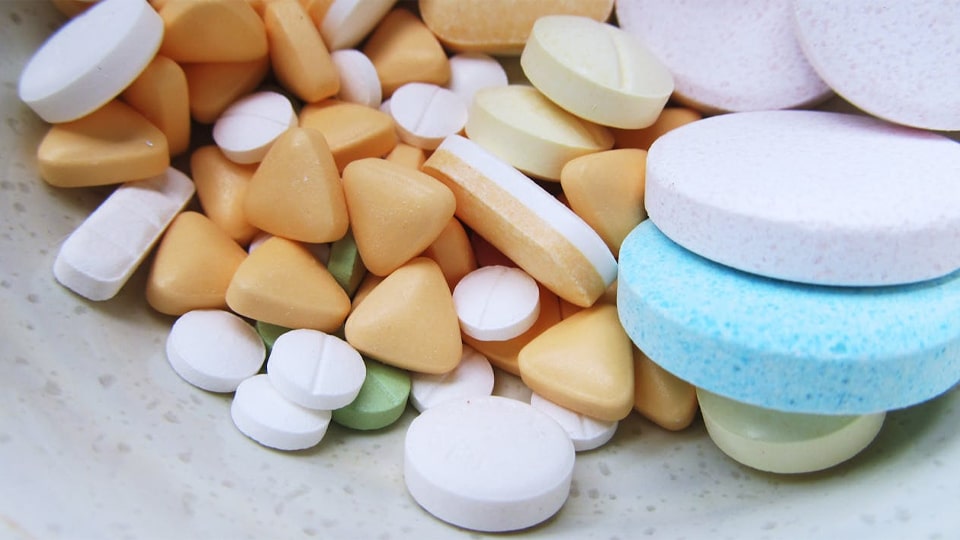
Figure: Film-coated pharmaceutical capsules. Color and coating make tablets easy to identify, improve swallowability, and strengthen branding. Color plays a big role in tablet design. A distinctive tablet color (or color pattern) enhances brand identity and helps patients distinguish between different medications and dosages. For example, one might use different shades for 100 mg vs 200 mg tablets of the same drug. Plain white tablets are cheap to make but often forgettable and can lead to mix-ups. By contrast, a brightly colored or two-toned tablet stands out on pharmacy shelves and reduces dosing errors.
In short, use color and coating strategically: they should reinforce safety and marketing goals, not just be decorative. Distinct colors per dosage and a user-friendly coating system make a big difference in patient compliance.

Behind every tablet is a carefully formulated blend of active drug and excipients (inactive ingredients). The formulation impacts manufacturing and performance:
The raw material properties have big effects: particle size, shape, and moisture content all matter. For instance, very fine or very moist granules can clog the press or lead to “capping” (tablet splitting). Too dry, and tablets may end up too brittle. The Friability Test (an industry-standard test) checks that tablets aren’t too fragile. A well-formulated tablet loses less than 1% of its weight in a friability test. Factors like adding a bit of moisture or binder can improve durability.
In practice, design your formulation to meet these goals:
To summarize, ingredient selection and ratios determine the tablet’s mechanical properties and performance. A good rule is to prototype your formulation and test tablet hardness, friability, dissolution, and uniformity early. Adjust binder/lubricant levels and granulation methods until you hit quality targets.

The tablet press machine you use must match your design. Key factors are the tablet’s size, shape complexity, and required production volume. Tablet presses range from small R&D benchtop units to huge high-speed rotary machines.
Cost Considerations: Tablet press prices vary widely with size and capacity. As a rough guide:
The table below summarizes typical press categories:
| Tablet Press Machine | Output (tablets/hour) | Max Tablet Size | Approx. Price (USD) |
| Single-punch / Benchtop R&D | ~1,000–10,000 | ~10 mm | $5,000–15,000 |
| Medium Rotary (12–19 stations) | ~30,000–100,000 | ~25 mm | $10,000–50,000 |
| High-speed Rotary (30+ stations) | 100,000–950,000+ | up to 60 mm | $200,000–800,000 |
Table: Example tablet press machines, output and price ranges. Notice how both capacity and size jump for larger machines. If your design calls for very large tablets or dual-layer tablets, ensure the press can handle those demands.
Lead Time & Support: Finally, consider lead time for ordering equipment. Jinlu Packing advertises ready stock for standard presses and a 7-day shipping promise, but custom or large models may take longer. Also factor in service support – a global service center or on-site setup (some suppliers charge a fee per day) can be crucial for smooth start-up.
A tablet’s visual identity extends beyond color. Think of it as a mini billboard. An attractive, consistent look helps both marketing and safety.
One example is apt: everyone recognizes the blue, diamond-shaped Viagra pill just by sight. You want your tablet to have a similarly clear visual identity. Work with designers to pick a color scheme and shape that fit your brand image.

Finally, any tablet design must meet strict quality and regulatory standards. Think of these as non-negotiable checkpoints:
Regulatory Compliance: Follow cGMP and pharmacopeia (USP, EP, etc.) guidelines. Maintain records of formulation trials, stability data, and process controls. A robust design includes built-in quality checks at each step.
By addressing these factors from the start—ingredient properties, machine parameters, environment, and tooling—you can avoid common pitfalls. In particular, maintain strict control of tablet press parameters (compression force, turret speed, fill depth) as even slight changes affect hardness and weight. Keep an eye on the production environment (temperature, humidity) since these can cause powder clumping or moisture uptake.
Good pharmaceutical tablet design is a balance of art and science. You must consider physical properties (size, shape, color), chemical formulation (ingredients, release mechanisms), manufacturing logistics (machines and cost), and quality requirements (testing and compliance). Each choice—from choosing an oval shape to picking a film coating—has ripple effects on patient experience and production efficiency.
By keeping tablets small enough for easy swallowing, using distinctive colors/coatings for branding and safety, formulating with the right excipients, and selecting appropriate tablet presses (with the necessary capacity and precision), you set your product up for success. Don’t forget to build quality tests into your process to catch any issues early.
Whether you’re a pharma manufacturer or an equipment buyer, understanding these factors ensures you make informed decisions. For tablet compression, consider how machine capabilities and price align with your design: even a straightforward-looking tablet can challenge a press if it’s too large or your output goals are high. Conversely, a powerful press is wasted on a tiny tablet with low volume.
Invest time in prototyping and consultation. Work with formulation experts and machinery vendors (like Jinlu Packing) early to match your tablet design to the best production solution. With the right design choices, your tablet will not only be effective therapeutically but also safe, appealing, and cost-efficient to make.
A pharmaceutical tablet is a solid dose form created by compressing a blend of active pharmaceutical ingredient (API) and excipients into a defined shape and size. Tablets are designed for oral administration, and their physical design affects factors like swallowability, dissolution, and drug release profile in the body.
Size and shape influence patient compliance, swallowability, and manufacturing efficiency. Smaller tablets (ideally under ~8–10 mm in diameter) are easier for patients to swallow, while shapes like oval or caplet can further enhance comfort and ease of use. Larger or irregular shapes may complicate coating and production processes.
Color and coating serve both functional and branding purposes. Distinctive colors help patients distinguish between dosages and reduce dosing errors, while film coatings make tablets smoother to swallow and can protect APIs from moisture or enhance release profiles.
A tablet formulation typically includes the active drug (API), fillers/diluents (to add bulk), binders (to improve tablet integrity), disintegrants (to aid dissolution), and lubricants (to prevent sticking during compression). The right balance ensures tablet strength, uniformity, and proper drug release.
Match the tablet design with appropriate equipment based on expected production volume, tablet size, shape, and complexity. Rotary presses are ideal for high-volume production, while single-punch presses work better for R&D or smaller batches. Ensure the machine can handle your specific tablet design and tooling requirements.
Tooling defines the final physical shape of the tablet. High-quality materials and precise design ensure consistent dimensions, reduce wear and tear, and prevent defects like rough edges. Poor tooling can lead to manufacturing issues or compromised tablet quality.
Excipients assist in achieving reliable manufacturing and drug performance. They contribute to flowability, compressibility, tablet strength, and dissolution rate. The choice and proportion of excipients can determine whether a tablet meets quality and functional benchmarks.
Common tests include hardness (ensuring physical durability), friability (tablet shouldn’t crumble under stress), dissolution (how the drug releases in the body), and content uniformity (each tablet contains the correct API amount). These help ensure safety and performance.
Patient factors such as ease of swallowing, taste masking, and tablet appearance impact adherence. A well-designed tablet increases the likelihood that patients will take medication as prescribed, thereby enhancing therapeutic outcomes.
Yes. Simple, symmetrical shapes (like round or oval) generally flow better through presses and coat evenly, improving manufacturing efficiency. Complex shapes may need custom tooling and careful coating parameter controls to avoid inconsistencies.
References:
1.Five factors to consider when designing a tablet – www.manufacturingchemist.com
2.FORMULATION DESIGN, MANUFACTURE CRITERIA AND REQUIREMENT OF VARIOUS TYPES OF TABLET – PharmaTutor Edu Labs
3.Tablet (pharmacy) – Wikipedia

Petty Fu, Founder of Jinlupacking, brings over 30 years of expertise to the pharmaceutical machinery sector. Under his leadership, Jinlu has grown into a trusted supplier integrating design, production, and sales. Petty is passionate about sharing his deep industry knowledge to help clients navigate the complexities of pharma packaging, ensuring they receive not just equipment, but a true one-stop service partnership tailored to their production goals.
Copyright © 2026 JinLuPacking.All rights reserved. Terms & Conditions and Privacy Policy
Friendly Links: Rich Packing | Capsule Filling Machine Manufacturers