
Pharmaceutical manufacturing refers to the industrial-scale process of turning raw ingredients into finished medicines. In other words, it’s how drug companies produce tablets, capsules, injectables and other dosage forms under strictly controlled conditions. This process involves a series of precise steps – from blending and mixing chemicals to forming pills and packaging them – all designed to ensure the final medicines are safe, effective, and high-quality. As Petty Fu notes, pharmaceutical manufacturing “is the industrial-scale process of synthesizing, preparing, and packaging pharmaceutical drugs and medicinal products”. In practice, it means transforming active pharmaceutical ingredients (APIs) and excipients into dosage forms (like tablets, capsules, liquids, or injectables) through a variety of unit operations (milling, granulation, compression, coating, aseptic filling, etc.) under tight regulations. The end goal is to produce reliable medicines on a large scale while meeting all health and safety standards.
Pharmaceutical manufacturing is not just casual mixing of chemicals – it’s a highly engineered process. At its core, it involves: identifying or synthesizing an active pharmaceutical ingredient (API), developing a stable formulation by mixing the API with other materials (binders, fillers, solvents, etc.), and then processing this blend into a final dosage form. Each operation (mixing, granulation, drying, compressing, coating, filling, etc.) must be done with precision. The FDA and other agencies treat this entire process very seriously – every batch must meet strict criteria for identity, strength, purity, and quality. In short, pharmaceutical manufacturing is the backbone of the drug industry, using specialized machinery and controlled environments to safely and efficiently turn chemical ingredients into medicines.

Pharmaceutical manufacturing is one of the most regulated industries in the world. Why? Because we’re making products people will inject or swallow, it’s critical that each dose is exactly right and free from contamination. Regulations (especially GMP – Good Manufacturing Practices) exist to ensure patient safety and consistent product quality. For example, FDA regulations require that drug makers follow Current Good Manufacturing Practices (cGMP), which are guidelines to make sure products “are consistently produced and controlled according to quality standards”. The goal is to guarantee that every medicine:

In short, because people’s lives are on the line, drug manufacturing is governed by rigorous rules from agencies like the FDA, EMA, and WHO. These rules cover every aspect – facility design, equipment, personnel training, raw material testing, record-keeping, cleaning, validation, and more. This way, regulators help ensure that every pill or vial that reaches patients is made in a way that “ensures a product is safe for use”.
The path from raw chemistry to final medicine involves multiple key stages. While specific processes vary by product, a typical pharmaceutical manufacturing workflow includes these main steps:
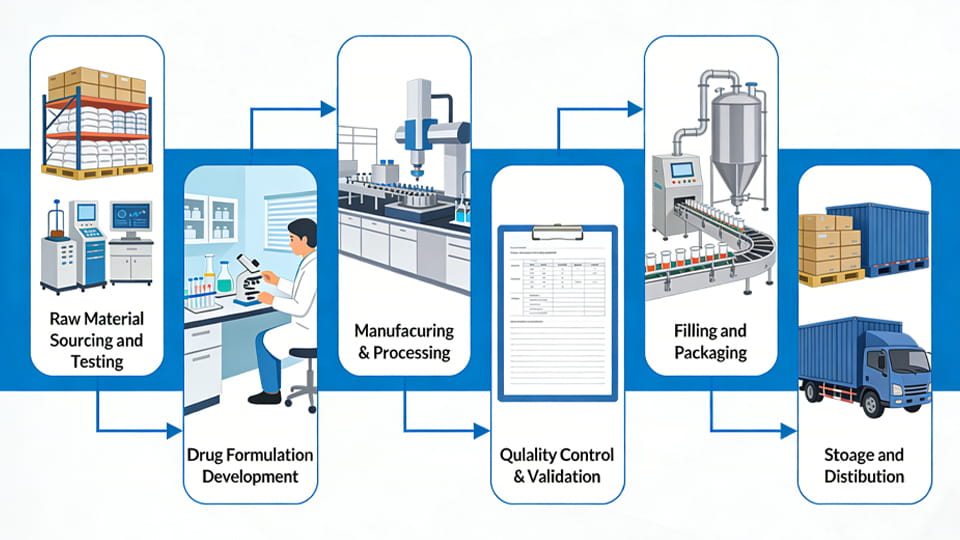
Throughout all these stages, strict adherence to GMP is enforced. From supplier audits in stage 1 to sterility checks in stage 6, quality is built in at every step. This multi-stage process – from raw materials to final delivery – defines modern pharmaceutical manufacturing.
Pharmaceutical plants can be organized in different ways depending on the product and scale. The main types of manufacturing approaches are:
Traditionally, most drug products have been made in batches. In a batch process, a fixed quantity (batch) of product is manufactured from start to finish. Each stage is executed separately on one batch before moving on. For example, a formula is mixed in a big tank, granulated, dried, compressed into tablets, and then that batch of tablets is packaged and tested. The advantage of batch manufacturing is flexibility: it’s relatively easy to switch formulations or modify a step for a new product. Early pharmaceutical plants favored this because it allowed simple process changes and straightforward documentation. Batch production involves every operation executed separately using a defined quantity of materials, with intermediate checks between phases. In practice, this means after one phase is done, the material is sampled and tested before proceeding.
Batch Production Pros: Easy to change products or batch sizes, simple process control and traceability, ideal for smaller runs or when frequent changeovers are needed.
Batch Production Cons: Generally slower and less efficient for large volume – after each batch, equipment must be cleaned and set up for the next one. More manual handling increases chances of human error. There are “numerous operations needed to handle, store, and monitor intermediate products” in batch mode. Also, if a mistake is found at the end, the entire batch may be scrapped (for example, a label error or a failed QC on tablets could force discarding all tablets in that batch).
An emerging approach is continuous manufacturing. Instead of discrete batches, continuous manufacturing keeps materials flowing through every step non-stop. Raw materials are fed in at one end of the production line and finished product constantly comes out the other. All unit operations (mixing, granulation, compression, etc.) are integrated in one line under real-time control. Continuous processes are highly automated, with in-line sensors measuring parameters (like mix uniformity or tablet hardness) on the fly. The benefits include much higher throughput, shorter production times, and more consistent quality. Continuous manufacturing optimizes the supply chain, enhances the robustness of the manufacturing process and thereby reduces product failures. In fact, research shows continuous processing can halve many costs – cutting product variability, quality-control time, and energy consumption by large margins (estimates range from 40% to 70% reductions). For example, because materials keep moving, problems affect only a small portion of product: any off-spec portion can be diverted while the rest proceeds. Regulatory agencies (FDA, EMA, etc.) are increasingly supportive of continuous manufacturing – in 2022 the ICH even published guidelines (Q13) to encourage its adoption.
Continuous Manufacturing Pros: Greater efficiency and throughput, fewer manual interventions, more uniform final product, and often smaller production footprint. Real-time quality monitoring means issues are caught immediately. It also allows quick scale-up by simply running the line longer or adding parallel lines.
Continuous Manufacturing Cons: Requires heavy capital investment in new, integrated equipment and control systems. Changing products or recipes is harder because the line is optimized for a specific process. Extensive process validation and regulatory approval is needed to switch from a proven batch to a continuous setup (companies must prove the new process yields the same result). As noted, adoption has been slow partly due to these challenges.
| Aspect | Batch Manufacturing | Continuous Manufacturing |
|---|---|---|
| Production Style | Discrete lot-by-lot runs | Ongoing, nonstop flow |
| Flexibility | High – easy to change products or batch sizes | Lower – optimized for one product, fewer changeovers |
| Throughput | Lower – downtime needed between batches | Higher – steady output, shorter lead times |
| Quality Control | Stage-by-stage sampling and testing | Inline monitoring and feedback control |
| Risk of Variability | Higher – each batch can vary from the last | Lower – built-in consistency (only small portion scrapped if issue) |
| Example Products | Many older drugs, small-volume batches, clinical supplies | High-volume solid dosage forms (tablets, capsules), APIs by flow chemistry |
Another key distinction is whether the product must be sterile. Sterile manufacturing applies to injectables (vaccines, IV drugs, eye drops) and implantables. These products cannot tolerate any microbial contamination. As a result, sterile pharma lines run under extremely strict conditions (high-grade cleanrooms, air filtration, sterilized equipment). For example, injectables often require ISO-5 cleanroom environments (class A/B in EU GMP terms) and aseptic filling processes. Every vial or syringe is filled under sterile laminar flow hoods or in pre-sterilized containers, and components (needles, filters) are sanitized. By contrast, non-sterile manufacturing includes products like tablets, capsules, ointments, and oral liquids that have acceptable limits for microbial content (governed by pharmacopeial standards). Non-sterile lines are still very clean, but they don’t require full aseptic conditions. Quality controls focus more on uniformity and stability rather than absolute sterility. In summary, sterile processes are more complex and costly (due to extra sterilization and cleanroom needs), whereas non-sterile processes, while still regulated, are somewhat simpler.
Pharmaceutical manufacturing can also be categorized by dosage form:
Each type (solid, liquid, injectable) has its own process nuances, but all must comply with GMP. The choice of manufacturing type depends on the medicine’s form and use. For instance, vaccines (injectable) go through more elaborate sterile processes than a tablet for oral use.
Modern pharma manufacturing relies on a wide array of specialized machinery. Equipment falls broadly into these categories:
Each piece of equipment is designed to meet pharmaceutical standards (stainless steel contact parts, ability to clean easily, and so on). Leading pharmaceutical equipment manufacturers (like Jinlupacking) produce machines such as tablet presses, capsule fillers, blister packers, counting machines, cartoners, and more. These machines are the backbone that drives efficiency and consistency in the pharma production line.
Good Manufacturing Practice (GMP) plays a central role in pharmaceutical manufacturing by ensuring that medicines are consistently produced and controlled according to strict quality standards, covering everything from raw materials and equipment to personnel, procedures, and documentation. According to the World Health Organization, GMP is designed to minimize risks such as contamination, incorrect labeling, and dosage errors that cannot be eliminated through final product testing alone . Similarly, the U.S. Food and Drug Administration emphasizes that GMP systems ensure drug products meet required standards for identity, strength, quality, and purity through controlled processes and continuous monitoring . By embedding quality into every stage of the pharmaceutical manufacturing process, GMP not only safeguards patient safety but also enables manufacturers to maintain consistency, compliance, and global market access.
Pharmaceutical manufacturing faces multiple complex challenges, primarily driven by strict regulatory requirements, high GMP compliance costs, and the need for consistent product quality across global markets. Manufacturers must continuously adapt to evolving regulations from agencies like FDA and WHO while ensuring data integrity, process validation, and contamination control, which significantly increases operational complexity . In addition, supply chain instability—such as raw material shortages, API quality risks, and supplier dependency—can directly disrupt pharmaceutical production and impact delivery timelines . Many companies also struggle with outdated equipment, limited automation, and the need for skilled personnel, all of which affect efficiency and compliance . As the pharmaceutical manufacturing industry continues to globalize, balancing cost control, quality assurance, and regulatory compliance remains one of the biggest ongoing challenges.
The pharmaceutical manufacturing industry is rapidly evolving with the rise of Pharma 4.0, where technologies such as AI, automation, and IoT are enabling smarter, data-driven production systems that improve efficiency and product quality . At the same time, more manufacturers are adopting continuous manufacturing to achieve faster production, real-time quality control, and more consistent output . These advancements, combined with increasing demand for personalized medicine and sustainable production, are pushing the industry toward more flexible, automated, and efficient pharmaceutical production models.
The sophisticated equipment used in pharma production isn’t just for show – it directly boosts efficiency and reliability. Here’s how modern machinery makes a difference:

In summary, advanced pharmaceutical machinery dramatically streamlines production. It turns what used to be labor-intensive, slow processes into high-speed, reliable workflows. As our sources show, leveraging continuous lines and automation leads to significantly better output and quality. The end result is that manufacturers can meet demand more quickly and consistently, while maintaining the highest quality standards.
Pharmaceutical manufacturing is the vital bridge between drug discovery and patient care. It’s the art and science of making medicines – a high-tech endeavor that requires precision, cleanliness, and strict compliance at every turn. We’ve seen that pharma manufacturing follows well-defined stages (from raw materials to packaging) and uses specialized equipment to make large quantities of safe, effective drugs. Because people’s health depends on it, the industry is tightly regulated under GMP to uphold quality. Looking forward, advances like continuous manufacturing, automation, and digitalization promise to make the process faster and even more reliable.
By understanding these processes and trends, industry professionals can appreciate how pharmaceutical equipment – tablet presses, blister machines, mixers, and more – contributes to efficiency and quality. Whether you are an engineer, buyer, or manufacturer in the pharmaceutical field, keeping up with these principles is essential.
If you’re looking to upgrade or scale your pharma production line, consider how the right machinery and expertise can make a difference. Contact us for a quote – our team at Jinlupacking specializes in providing high-quality pharmaceutical manufacturing and packaging equipment solutions to optimize your processes.
Pharmaceutical manufacturing is the industrial process of producing medicines from raw materials into finished dosage forms such as tablets, capsules, liquids, or injectables. It involves formulation, processing, quality control, and packaging under strict regulatory standards to ensure safety and effectiveness.
Pharmaceutical manufacturing is essential because it ensures that medicines are produced consistently, safely, and at scale. Without it, drugs developed in laboratories could not reach patients in reliable, usable forms.
The pharmaceutical manufacturing process typically includes:
• Raw material sourcing and testing
• Formulation development
• Manufacturing and processing
• Quality control and validation
• Filling and packaging
• Storage and distribution
Each stage ensures product quality and compliance with regulatory standards.
GMP (Good Manufacturing Practice) refers to a set of regulations that ensure medicines are consistently produced and controlled according to quality standards. It helps minimize risks such as contamination, incorrect dosage, and labeling errors.
Common pharmaceutical machinery includes:
• Tablet press machines
• Capsule filling machines
• Mixers and granulators
• Blister packaging machines
• Bottle filling and labeling machines
These machines ensure efficiency, precision, and compliance in pharmaceutical production.
• Batch manufacturing produces medicines in fixed quantities step-by-step.
• Continuous manufacturing runs non-stop with materials flowing through all stages.
Continuous manufacturing offers higher efficiency and consistency, while batch manufacturing provides flexibility.
API (Active Pharmaceutical Ingredient) is the main substance in a drug that produces the intended therapeutic effect. It is combined with excipients to create the final dosage form.
It is highly regulated to ensure patient safety, product quality, and consistency. Regulatory agencies require strict controls to prevent contamination, ensure correct dosage, and maintain product integrity throughout production.
Pharmaceutical packaging equipment is used to protect medicines, ensure accurate dosing, and provide essential information. Machines like blister packers and bottle fillers help maintain product stability and improve patient compliance.
Key challenges include:
• Strict regulatory compliance
• Supply chain disruptions
• High production costs
• Complex quality control requirements
• Risk of contamination
These factors make pharmaceutical manufacturing one of the most demanding industrial sectors.
References:
1. Pharmaceutical manufacturing — Wikipedia.
2.Medicines: Good manufacturing practices — WHO.
3.Current Good Manufacturing Practice (CGMP) Regulations — FDA.
4.Pharmaceutical Manufacturing & Quality Guidelines — European Medicines Agency.

Petty Fu, Founder of Jinlupacking, brings over 30 years of expertise to the pharmaceutical machinery sector. Under his leadership, Jinlu has grown into a trusted supplier integrating design, production, and sales. Petty is passionate about sharing his deep industry knowledge to help clients navigate the complexities of pharma packaging, ensuring they receive not just equipment, but a true one-stop service partnership tailored to their production goals.
Copyright © 2026 JinLuPacking.All rights reserved. Terms & Conditions and Privacy Policy
Friendly Links: Rich Packing | Capsule Filling Machine Manufacturers