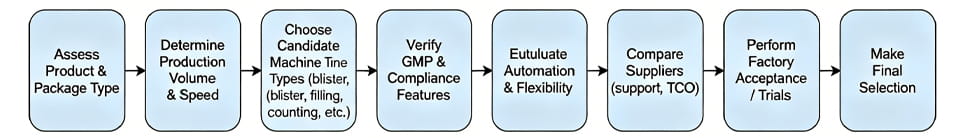
Lựa chọn quyền máy đóng gói dược phẩm là rất quan trọng để đảm bảo an toàn sản phẩm, tuân thủ quy định, và sản xuất hiệu quả. Thị trường bao bì dược phẩm toàn cầu đang bùng nổ - một dự báo dự báo nó sẽ vượt quá $160 tỷ bởi 2026. Đồng thời, cơ quan quản lý yêu cầu kiểm soát chất lượng nghiêm ngặt: ví dụ. CGMP của FDA (21 Bộ phận CFR 210–211) đặt ra các yêu cầu nghiêm ngặt về sản xuất, xử lý, và đóng gói thuốc. Trong thực tế, gần như một nửa thu hồi thuốc liên quan đến lỗi đóng gói hoặc ghi nhãn, làm nổi bật cổ phần. Trong hướng dẫn này, chúng ta sẽ xem xét tất cả những điều cần cân nhắc chính – định dạng sản phẩm, dung tích, Tuân thủ, tự động hóa, hỗ trợ nhà cung cấp, và hơn thế nữa – để bạn có thể tự tin lựa chọn thiết bị đóng gói dược phẩm phù hợp với nhu cầu của mình. Chúng tôi bao gồm các loại máy (máy vỉ, chất độn viên nang, thùng giấy, vân vân.), so sánh hệ thống tự động và bán tự động, và bao gồm lời khuyên của chuyên gia, một bảng so sánh, sơ đồ các bước lựa chọn, một câu hỏi thường gặp, và những khuyến nghị khả thi.
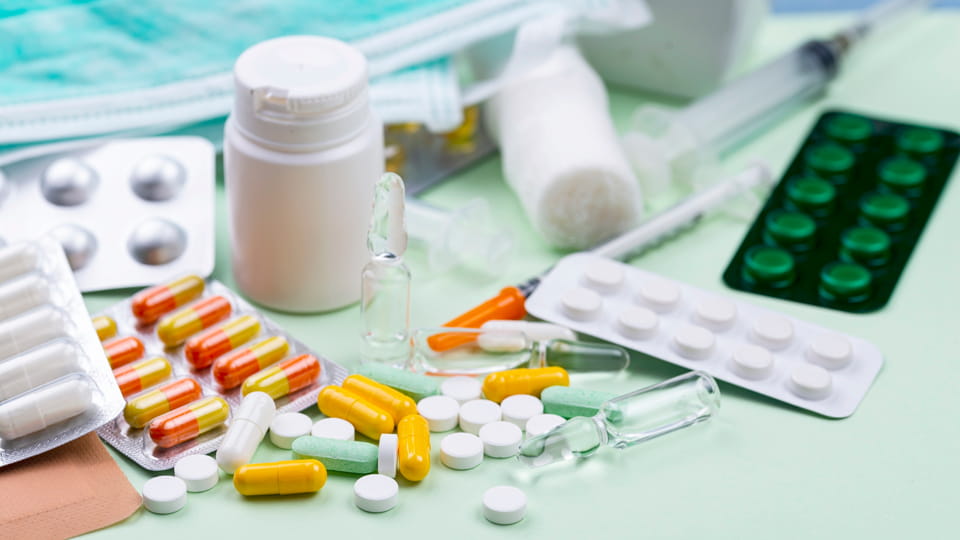
Tại sao việc chọn đúng máy lại quan trọng
Bao bì dược phẩm quan trọng hơn nhiều so với bao bì tiêu dùng thông thường. Nó bảo vệ chất lượng thuốc, đảm bảo độ chính xác về liều lượng, và bảo vệ sự an toàn của bệnh nhân. Ví dụ, Hướng dẫn của WHO nhấn mạnh rằng bao bì phải bảo vệ thuốc khỏi bị ẩm, ánh sáng, ôxy, và các ảnh hưởng bất lợi khác trong suốt thời hạn sử dụng của chúng. Tương tự như vậy, Các quy tắc CGMP của FDA quy định rằng “các phương pháp, cơ sở, và kiểm soát” trong chế biến, đóng gói phải bảo đảm thuốc an toàn và đúng thành phần. Về mặt thực tế, bao bì kém có thể dẫn đến sự ô nhiễm, ghi nhãn sai, lỗi liều lượng, hoặc trục trặc của thiết bị – thậm chí gây hại cho bệnh nhân. Thực vậy, phân tích dữ liệu thu hồi chỉ ra lên đến 50% thu hồi thuốc là do lỗi đóng gói hoặc ghi nhãn. Điều đó có nghĩa là một sự giám sát dường như nhỏ (mã vạch sai, vỉ bị hư hỏng, đếm nhầm viên thuốc) có thể gây ra việc thu hồi tốn kém hoặc cảnh báo theo quy định.
Ngoài sự an toàn, máy phù hợp sẽ tăng năng suất và giảm chi phí. Dòng dược phẩm hiện đại tích hợp chiết rót, niêm phong, đếm và ghi nhãn vào hệ thống tự động tốc độ cao. Việc chọn thiết bị phù hợp với nhu cầu đầu ra và mức độ tự động hóa của bạn có thể cải thiện đáng kể năng suất và giảm nhân công. Ngược lại, lựa chọn sai có thể gây ra ùn tắc thường xuyên, lấp đầy không nhất quán, và thời gian ngừng hoạt động – ảnh hưởng đến hiệu quả. Và bởi vì các nhà máy dược phẩm thường xuyên chạy 24/7, bảo trì đột xuất có thể là thảm họa. Vì những lý do này, các chuyên gia trong ngành nhấn mạnh sự lựa chọn cân bằng: “Việc lựa chọn thiết bị đóng gói dược phẩm đòi hỏi phải đánh giá cẩn thận khả năng tuân thủ GMP, sẵn sàng xác nhận, và độ tin cậy hoạt động lâu dài”. Tóm lại, máy phù hợp đảm bảo chất lượng và tuân thủ (tránh sai sót/thu hồi) trong khi tối ưu hóa năng lực sản xuất và chi phí.

Các loại máy đóng gói dược phẩm
“Bao bì dược phẩm” bao gồm nhiều sản phẩm và hình thức. Máy móc rơi vào bao bì sơ cấp (lớp đầu tiên tiếp xúc với thuốc) Và bao bì cấp hai/cấp ba (thùng carton bên ngoài, bó, pallet). Máy điển hình bao gồm:
- Máy chiết rót và định lượng: Vì chất lỏng, kem, hoặc bột trong lọ/chai/gói. Ví dụ: chất độn lỏng, máy làm đầy bột, chất độn xi-rô. Nếu sản phẩm của bạn là một thuốc dạng lỏng, chọn một máy được thiết kế cho chất lỏng (chất độn nhu động hoặc piston). Vì bột/hạt (ví dụ. máy tính bảng số lượng lớn đang được đóng gói), chất độn thể tích hoặc mũi khoan là lý tưởng.
- Thiết bị viên nang/máy tính bảng: Vì viên nang hoặc viên nén, máy chủ chốt là Máy làm đầy viên nang, máy tính bảng ấn, Và máy đếm/làm đầy. Chất làm đầy viên nang hiện đại có thể tự động hóa việc đặt, sự tách biệt, điền vào, và khóa viên nang. Máy ép viên nén bột thành viên. Máy đếm (quầy quang học) cho ăn và đếm liều rắn vào chai hoặc túi. Như ghi chú của JinLuPacking, TRONG liều rắn dòng “mọi thứ đều bắt đầu bằng việc đếm chính xác” – cảm biến đa kênh tốc độ cao đảm bảo mỗi chai/túi có số đếm chính xác.
- Máy đóng gói vỉ: Đối với viên nén/viên nang trong vỉ. Những khay định hình nhiệt này sẽ niêm phong viên thuốc dưới lớp giấy bạc. Máy ép vỉ đến như tấm phẳng (được lập chỉ mục) hoặc quay liên tục các loại, với nhiều định dạng khác nhau (Giấy bạc dạng lạnh hoặc dạng PVC). Chúng lý tưởng cho việc đóng gói theo đơn vị liều lượng.
- Máy đóng hộp/đấm bốc: Đóng gói thứ cấp. thùng carton gấp và dán thùng carton, thường xuyên chèn các vỉ hoặc chai cùng với tờ rơi của bệnh nhân. Họ có thể nằm ngang (tải phụ) hoặc thẳng đứng (tải hàng đầu) Máy đóng thùng tự động dựng hộp, đang tải nội dung, và niêm phong – giảm lao động thủ công và đảm bảo đóng hộp nhất quán (quan trọng đối với việc truy xuất nguồn gốc của GMP). Máy đóng thùng hiện đại sử dụng thiết kế vệ sinh bằng thép không gỉ và con dấu chống giả mạo để đáp ứng các quy định nghiêm ngặt.
- Người dán nhãn và thanh tra: Máy ghi nhãn dán nhãn in sẵn trên chai/thùng có mã lô, ngày hết hạn, và mã vạch. Hệ thống kiểm tra (tầm nhìn của máy ảnh, máy dò kim loại, tia X) kiểm tra độ chính xác điền, hạt lạ, sự hiện diện của nhãn, và khả năng đọc mã. Để tuân thủ, tìm kiếm thiết bị hỗ trợ tuần tự hóa (mã vạch có thể theo dõi) và từ chối lỗi.
- Dây chuyền đóng gói tích hợp: Trong thực tế, nhà máy dược phẩm thường sử dụng dòng hoàn chỉnh kết hợp các máy trên. Ví dụ, dây chuyền sản xuất máy tính bảng sang thùng carton có thể bao gồm máy ép viên, máy hút ẩm, quầy thuốc viên, Chai Unscrambler, đơn vị làm đầy, máy đóng nắp, niêm phong cảm ứng, ghi nhãn, thùng carton, và băng tải kiểm tra cuối cùng. Các hệ thống đầu cuối này có thể được lắp ráp trước và xác nhận dưới dạng một dây chuyền, giúp đơn giản hóa trình độ chuyên môn và dấu chân. .
Tóm lại, chọn máy phù hợp với dạng bào chế và hình thức đóng gói của bạn. Như một người hướng dẫn khuyên, “Máy đóng gói vỉ rất lý tưởng cho máy tính bảng, trong khi hệ thống đóng dấu theo mẫu nằm ngang được sử dụng để đóng gói thông lượng cao”. Hiểu sản phẩm của bạn (viên thuốc, viên nang, chất lỏng, bột) và gói mong muốn (mụn rộp, cái chai, gói, túi làm sẵn, vân vân.) là bước đầu tiên trong việc chọn máy.

Các yếu tố chính cần xem xét
Khi đánh giá máy móc hoặc nhà cung cấp cụ thể, hãy ghi nhớ những yếu tố quan trọng này:
- Sản phẩm & Khả năng tương thích định dạng bao bì: Đảm bảo thiết bị có thể xử lý hình thức và thùng chứa sản phẩm chính xác của bạn. Dạng bào chế và loại bao bì vấn đề: máy tính bảng, viên nang, chất lỏng, tất cả các loại bột đều có nhu cầu xử lý riêng. Ví dụ, chất rắn có thể đếm được sử dụng hệ thống đếm hoặc vỉ, trong khi chất lỏng yêu cầu chất làm đầy chất lỏng có chất bịt kín. Đồng thời kiểm tra kích thước và hình học: một số máy có thể xử lý nhiều trọng lượng đổ đầy hoặc kích thước thùng chứa, những người khác thì tận tâm. Tính linh hoạt là chìa khóa nếu bạn lập kế hoạch cho nhiều SKU. Mô tả máy thường chỉ định các định dạng tương thích; xác minh bằng các thử nghiệm sản phẩm mẫu nếu có thể.
- Tốc độ sản xuất & Dung tích: Kết hợp công suất của máy với nhu cầu hiện tại và tương lai của bạn. Máy có “tốc độ định mức” (ví dụ. chai/phút hoặc vỉ/phút) và đầu ra thực tế (có tính đến sự thay đổi). Thông số kỹ thuật quá mức (tốc độ rất cao) lãng phí tiền bạc và không gian; thông số kỹ thuật dưới mức gây ra tắc nghẽn. Nhiều nhà sản xuất (giống như JinLu) báo giá tốc độ: ví dụ. một số dòng viên nang có thể sản xuất hàng trăm nghìn viên nang mỗi giờ, máy đếm >99.98% chính xác ở mức ~6.000 chai/h, và thùng carton lên đến 450 thùng/phút. Không chỉ xem xét sản lượng hiện tại mà còn cả tốc độ tăng trưởng – JinLuPacking khuyên bạn nên chọn công suất “phù hợp với nhu cầu hiện tại của bạn đồng thời vẫn có dư địa để tăng trưởng”. Tìm kiếm các máy mô-đun hoặc tùy chọn nhiều làn nếu bạn thấy trước âm lượng sẽ tăng.
- Sự chính xác & Kiểm soát chất lượng: Độ chính xác cao là điều không thể thương lượng trong ngành dược phẩm. Thiết bị chiết rót, đếm phải đảm bảo không có thêm hoặc thiếu thuốc, đúng liều lượng, và một gói được niêm phong đáng tin cậy. Các dòng hàng đầu nhằm mục đích “không thừa một miếng nào và không thiếu một miếng nào” trong mỗi thùng chứa. Đánh giá từng máy thông số kỹ thuật chính xác: ví dụ. một người điền số đếm có thể yêu cầu 99.98% sự chính xác. Chúng tôi khuyên dùng cảm biến quang học đa kênh hoặc định lượng điều khiển bằng servo để đạt được dung sai chặt chẽ. Đồng thời kiểm tra các tính năng chất lượng tích hợp: ví dụ. hệ thống thị giác để xác minh số lượng, phát hiện rò rỉ, cân kiểm tra trọng lượng. Máy nên tích hợp kiểm soát trong quá trình để bắt lỗi (từ chối hàng loạt, cảnh báo) để đảm bảo chất lượng ổn định.
- GMP & Tuân thủ quy định: Máy dược phẩm phải đáp ứng các tiêu chuẩn nghiêm ngặt về vệ sinh và tài liệu. tìm kiếm thiết kế tuân thủ cGMP: tất cả các bộ phận tiếp xúc với sản phẩm trong Thép không gỉ (SUS316L) hoặc các vật liệu cấp dược phẩm khác, bề mặt nhẵn (không có chân cụt hoặc kẽ hở), và dễ dàng tháo lắp để vệ sinh. Bề mặt phải chống ăn mòn/ô nhiễm. Máy nên có nhân viên bảo vệ an toàn, bảng điều khiển kín, và thay đổi ít công cụ hơn nếu có thể. Điều quan trọng không kém là việc tuân thủ phần mềm: các dòng dược phẩm hiện đại thường thuộc FDA 21 Phần CFR 11 (hồ sơ điện tử) và Phụ lục EU GMP 11 (hệ thống máy tính). Chúng tôi khuyên bạn nên đảm bảo hỗ trợ thiết bị hồ sơ điện tử, dấu vết kiểm toán, và quyền truy cập dựa trên vai trò, và đi kèm với gói xác nhận toàn diện (tài liệu thiết kế, Giao thức FAT/SAT, Mẫu IQ/OQ/PQ). Tóm lại, máy phải được sẵn sàng xác thực. Ưu tiên máy có dấu CE hoặc chứng nhận địa phương khác nếu xuất khẩu. Hãy nhớ rằng hướng dẫn đóng gói (AI, FDA, EU) mọi căng thẳng trong việc duy trì tính nguyên vẹn của sản phẩm thông qua việc làm sạch, quy trình được xác nhận.
- Linh hoạt & Thời gian chuyển đổi: Các dòng dược phẩm thường chạy nhiều sản phẩm. Tìm kiếm những chiếc máy có khả năng chuyển đổi nhanh chóng. Các tính năng có thể bao gồm các đầu điều chỉnh được điều khiển bằng servo, bộ nhớ "công thức nấu ăn" cho cài đặt, và thay thế khuôn hoặc thanh dẫn hướng mà không cần dụng cụ. JinLuPacking nhấn mạnh “không cần công cụ, chuyển đổi nhanh chóng với tính năng lưu trữ công thức” để xử lý nhiều SKU. Chiếc máy có thể chuyển đổi định dạng trong vài phút (vs. giờ) được đền đáp bằng cách giảm thời gian ngừng hoạt động và tăng OEE. Máy bán tự động điều chỉnh đơn giản hơn nhưng yêu cầu người vận hành; máy hoàn toàn tự động thường thay đổi công cụ nhanh hơn nếu được thiết kế tốt. Đồng thời xem xét tính linh hoạt của bố cục – nguồn cấp dữ liệu dọc và ngang, làn đường, vân vân., để phù hợp với hình dạng gói khác nhau.
- Mức độ tự động hóa: Quyết định giữa bán tự động (một số bước thủ công) Và Hoàn toàn tự động hệ thống. Dây chuyền đóng gói tự động giảm thiểu lao động và lỗi của con người, đạt được sự nhất quán 24/7 thông lượng. Ví dụ, dựng thùng carton tự động, tải và niêm phong các thùng carton với đầu vào tối thiểu của người vận hành, trong khi máy đóng thùng bán tự động cần nạp thùng carton thủ công. Dây chuyền đếm/làm đầy hoàn toàn tự động tích hợp bộ cấp liệu rung và cánh tay gắp và đặt cho máy tính bảng, loại bỏ việc đếm thủ công. Sự đánh đổi: máy tự động đắt hơn và cài đặt phức tạp hơn. Máy bán tự động có giá thành rẻ hơn nhưng lại bó buộc công nhân và chạy ở tốc độ thấp hơn. Chúng tôi so sánh chúng trong bảng dưới đây:
| Tính năng |
Máy tự động |
Máy bán tự động |
| Thông lượng |
Rất cao (liên tục, 24/7) |
Vừa phải (hàng loạt hoặc không liên tục) |
| Yêu cầu lao động |
Thấp (phi hành đoàn nhỏ để giám sát) |
Cao hơn (người vận hành cho ăn/xử lý) |
| Linh hoạt |
Trung bình (định dạng chuyên dụng; nhanh hơn về âm lượng) |
Cao (xử lý thủ công cho phép các định dạng khác nhau) |
| Thời gian chuyển đổi |
Có thể lâu hơn (thiết lập phức tạp) |
Thường ngắn hơn (cơ học đơn giản hơn) |
| Chi phí ban đầu |
Cao hơn (điều khiển phức tạp & người máy) |
Thấp hơn |
| BẢO TRÌ |
Tổ hợp (yêu cầu kỹ thuật viên được đào tạo) |
Đơn giản hơn (các bộ phận người dùng có thể sử dụng được) |
Tóm lại, sử dụng máy tự động để âm lượng lớn, sản xuất ổn định. Nếu đợt chạy của bạn nhỏ hoặc không thường xuyên, bán tự động có thể đủ ngân sách. (Ví dụ, máy ép vỉ bán tự động chỉ có thể hàn nhiệt các vỉ trong khi người vận hành nạp thuốc bằng tay.) Dù bạn chọn cấp độ nào, đảm bảo các điều khiển hiện đại (PLC/HMI, khóa liên động an toàn) và giao diện người dùng trực quan.
- Vệ sinh & Khả năng làm sạch: Máy móc dược phẩm phải được dễ dàng làm sạch và khử trùng để ngăn ngừa lây nhiễm chéo. Kiểm tra xem tất cả các bộ phận tiếp xúc có thể tiếp cận được để rửa sạch không, và không có góc bẫy bột hoặc chất lỏng. Nhiều máy dược phẩm sử dụng các bộ phận tháo nhanh không cần dụng cụ để làm sạch. Xem xét máy có hỗ trợ không CIP (sạch sẽ tại chỗ) hoặc chu trình làm sạch lau chùi. Thiết kế nên cho phép giặt khô (Ví dụ, các bộ phận có thể hoán đổi cho nhau để chứa bột). Đồng thời đánh giá các bộ lọc và thông gió nếu có liên quan đến bột. Cần có quy trình vệ sinh nghiêm ngặt trong môi trường dược phẩm, vì vậy hãy lên kế hoạch cho các thói quen bảo trì hàng ngày/hàng tuần. (Cho tiền boa: Đọc lời khuyên của HMC – các dòng dược phẩm yêu cầu “quy trình làm sạch nghiêm ngặt để loại bỏ cặn và ngăn ngừa lây nhiễm chéo giữa các lô”.)
- Tính toàn vẹn dữ liệu & Truy xuất nguồn gốc: Máy sẽ tạo điều kiện thuận lợi cho việc truy xuất nguồn gốc hàng loạt. Các tính năng cần tìm bao gồm mã vạch nối tiếp (đối với gói đơn vị liều), ghi dữ liệu của các thông số quá trình, và tích hợp với hệ thống MES/ERP của nhà máy. Các máy hàng đầu cung cấp các bản kiểm tra cho tất cả các thông số quan trọng (nhiệt độ, trọng lượng, đếm). Để tuần tự hóa (được yêu cầu bởi nhiều thị trường như DSCSA ở Mỹ hay FMD ở EU), đảm bảo dây chuyền có thể xử lý việc kiểm tra trực quan mã 1D/2D, từ chối mã xấu, và tổng hợp dữ liệu cha-con (VÍ DỤ., liên kết các gói vỉ riêng lẻ với thùng carton của chúng). Về cơ bản, thiết bị sẽ giúp bạn đáp ứng các yêu cầu truy tìm của FDA và EU. Máy móc hiện đại thường có OEE (Hiệu quả thiết bị tổng thể) giám sát bảng điều khiển là tốt.
- Tổng chi phí sở hữu (TCO): Nhìn xa hơn giá nhãn dán. Coi như Tiêu thụ năng lượng (yếu tố làm nóng, máy bơm chân không, động cơ), chi phí tiêu hao (phim ảnh, giấy bạc, bột, phụ tùng thay thế như con dấu), Và yêu cầu dịch vụ. Một chiếc máy rất rẻ có thể có thời gian ngừng hoạt động cao hoặc sử dụng các bộ phận không đạt tiêu chuẩn. Ngược lại, một chiếc máy chất lượng có thể sử dụng nhiều năng lượng hơn nhưng tạo ra ít chất thải hơn. Các yếu tố bao gồm: lao động bảo trì (tần suất và mức độ khó duy trì), thời gian giao phụ tùng thay thế (quan trọng đối với máy nhập khẩu), và tài chính/ROI. Như ghi chú blog Jinlu dành cho máy đóng vỉ, “Máy chất lượng cao có thể có chi phí trả trước cao hơn nhưng có thể giảm lãng phí và thời gian ngừng hoạt động”. Tóm lại, tính toán chi phí vòng đời: giá mua + chi phí vận hành + BẢO TRÌ. Các nhà cung cấp thường cung cấp các công cụ hoặc máy tính TCO cho việc này.
- Kinh nghiệm của nhà cung cấp & Ủng hộ: Cuối cùng, lựa chọn nhà sản xuất uy tín, có kinh nghiệm về dược. Thiết bị đóng gói là khoản đầu tư dài hạn; bạn muốn có một đối tác, không chỉ là một nhà cung cấp. Kiểm tra hồ sơ theo dõi của nhà cung cấp: họ có nhà máy tham khảo ở khu vực/ngành của bạn không? Máy của họ có được xác nhận cho khách hàng dược phẩm không? Ưu tiên các nhà cung cấp có chuyên môn về GMP đã được chứng minh, hỗ trợ xác nhận toàn diện, và hồ sơ theo dõi đã được chứng minh trong các ngành được quản lý. Đồng thời đánh giá dịch vụ sau bán hàng: họ có cung cấp hỗ trợ vận hành không (MỠ/SAT/IQ/OQ), tồn kho phụ tùng, chẩn đoán từ xa, và đào tạo? Một nhà cung cấp tốt sẽ giúp tối ưu hóa bố cục của bạn, hỗ trợ kiểm toán theo quy định, và xử lý các sự cố khẩn cấp. Tránh những người bán hàng đêm – tìm kiếm các chứng chỉ trong ngành (ISO, CN), bảo đảm, và mạng lưới toàn cầu.
Dưới đây là một sơ đồ tóm tắt quá trình lựa chọn:
Tự động so với. Bao bì bán tự động: Một sự so sánh
Khi lựa chọn phạm vi, nó giúp so sánh các dây chuyền hoàn toàn tự động với các lựa chọn thay thế bán tự động. Dây chuyền tự động tích hợp cho ăn, hình thành, điền vào, và niêm phong trong một quá trình liên tục, trong khi dây chuyền bán tự động yêu cầu can thiệp thủ công (ví dụ. đóng chai hoặc thùng carton bằng tay). Nói chung:
- Tự động hệ thống cung cấp thông lượng rất cao (thường rất cần thiết cho >100 thùng/phút, hoặc lô viên nang lớn). Họ hoạt động với lao động tối thiểu (1–2 người vận hành để giám sát), làm giảm lỗi của con người. Họ cũng thường tự động thu thập nhật ký dữ liệu đầy đủ. Nhược điểm là chi phí vốn cao hơn và sự phức tạp. Họ yêu cầu các kỹ thuật viên được đào tạo để duy trì.
- Bán tự động máy móc đang chi phí thấp hơn và đơn giản hơn. Họ có thể cần công nhân để cung cấp sản phẩm hoặc thùng carton, làm cho chúng phù hợp với chạy nhỏ và vừa. Chuyển đổi có xu hướng nhanh hơn (ít trục chuyển động hơn), và đào tạo đơn giản hơn. Tuy nhiên, Các bước thủ công làm tăng chi phí lao động và tính biến đổi. Các dòng bán tự động hiếm khi đạt được tốc độ như tự động hoàn toàn.
Dưới đây là so sánh nhanh về các thuộc tính chính:
| Tính năng |
Dòng tự động |
Dây chuyền bán tự động |
| Thông lượng |
Rất cao (100+ đơn vị/phút) |
Vừa phải (tùy thuộc vào tốc độ của người vận hành) |
| Nhân công |
Thấp (1–2 người, chủ yếu là xem) |
Cao hơn (một số bộ phận vận hành cho ăn) |
| Linh hoạt |
Trung bình (được tối ưu hóa cho định dạng cụ thể) |
Cao (người vận hành có thể thích ứng nhanh chóng) |
| Chuyển đổi |
Có thể yêu cầu kỹ thuật (nhiều bộ phận/công cụ hơn) |
Thường nhanh hơn (ít bộ phận hơn để trao đổi) |
| Chi phí ban đầu |
Cao (Plc, người máy, cảm biến) |
Thấp hơn (thiết bị truyền động và băng tải cơ bản) |
| BẢO TRÌ |
Căng (kỹ năng công nghệ chuyên ngành) |
Dễ dàng hơn (bảo trì cơ khí thông thường) |
Trong thực tế, nhiều dòng dược phẩm kết hợp cả hai: ví dụ. một trạm nạp tự động nạp vào thùng carton bán tự động, hoặc ngược lại. Điều quan trọng là phải phù hợp với mức độ tự động hóa phù hợp với khối lượng của bạn. Nếu bạn có kế hoạch mở rộng quy mô hoặc 24/7 chạy, tự động hóa hoàn toàn thường mang lại kết quả. Đối với các đợt thí điểm hoặc hạn chế về ngân sách, một máy để bàn hoặc để bàn bán tự động có thể đủ.

Những cạm bẫy phổ biến cần tránh
- Bỏ qua việc tuân thủ: Không bao giờ tiết kiệm các tính năng của GMP để tiết kiệm chi phí. Máy không tuân thủ sẽ gây ra lỗi kiểm tra hoặc thu hồi.
- Lựa chọn mua hàng “Rẻ nhất”: Giá thầu quá thấp có thể thiếu độ chính xác hoặc hỗ trợ. Xem xét ROI dài hạn, không chỉ giá trả trước.
- Bỏ qua nhu cầu trong tương lai: Đừng mua vì chỉ hôm nay thôi đầu ra; chừa chỗ cho sự tăng trưởng hoặc sản phẩm mới.
- Bỏ qua các cuộc kiểm tra chấp nhận của nhà máy: Luôn kiểm tra máy tại nhà máy với mẫu của bạn (MẬP) trước khi giao hàng. Tương tự như vậy, thực hiện chấp nhận trang web (ĐÃ NGỒI) với nhóm của bạn để đảm bảo nó đáp ứng các thông số kỹ thuật.
- Nhìn ra tiện ích & Tích hợp: Kiểm tra yêu cầu cơ sở vật chất (quyền lực, không khí, Nước) và đảm bảo máy phù hợp với sơ đồ mặt bằng của bạn. Đồng thời đảm bảo nó tích hợp trơn tru với băng tải hoặc hệ thống điều khiển hiện có.

Cách chọn nhà cung cấp phù hợp
Hợp tác với nhà sản xuất thiết bị phù hợp cũng quan trọng như việc lựa chọn máy. tìm kiếm:
- Chuyên môn trong ngành: Các nhà cung cấp chuyên về thiết bị dược phẩm sẽ được cập nhật về các tiêu chuẩn và quy trình xác nhận của FDA/EMA/WHO. Họ có thể tư vấn về cách bố trí và kiểm soát chất lượng. Như một hướng dẫn nêu, “máy bạn chọn phải phù hợp với dạng bào chế của bạn, định dạng bao bì, yêu cầu quy định, và năng lực sản xuất”, vì vậy kinh nghiệm rất quan trọng.
- Hỗ trợ xác thực: Đảm bảo nhà cung cấp cung cấp tài liệu đầy đủ: Kế hoạch kiểm tra nghiệm thu tại nhà máy/địa điểm, Giao thức IQ/OQ/PQ, đánh giá rủi ro, và các gói xác nhận phần mềm. Lý tưởng nhất là họ thậm chí sẽ hỗ trợ thực hiện SAT.
- Dịch vụ hậu mãi: Kiểm tra các điều khoản bảo hành và cách công ty xử lý phụ tùng và dịch vụ. Họ có văn phòng hoặc nhà phân phối địa phương không? Có hỗ trợ từ xa không? Một nhà cung cấp có thể nhanh chóng vận chuyển các bộ phận hoặc cử kỹ thuật viên sẽ giảm thiểu thời gian ngừng hoạt động.
- Danh tiếng & Tài liệu tham khảo: Yêu cầu tài liệu tham khảo hoặc nghiên cứu trường hợp cài đặt tương tự. Lựa chọn các công ty có sự hỗ trợ kỹ thuật mạnh mẽ, đội dịch vụ toàn cầu, dự án dược phẩm được xác nhận, và độ tin cậy của máy đã được chứng minh.
- Khả năng tùy biến: Sản phẩm của bạn có thể có những nhu cầu riêng biệt (vật liệu đặc biệt, thông số phòng sạch, vân vân.). Một nhà cung cấp tốt sẽ tùy chỉnh máy móc (ví dụ. buồng đặc biệt, thêm tự động hóa, ngôn ngữ giao diện người dùng) theo yêu cầu của bạn. Họ, Ví dụ, cung cấp các dây chuyền chìa khóa trao tay phù hợp với nhu cầu về không gian và quy trình.
Bằng cách kiểm tra cẩn thận các nhà cung cấp về những điểm này, bạn giảm rủi ro dự án. Nhớ: “Đối tác thiết bị phù hợp sẽ trở thành một phần mở rộng của đội ngũ kỹ thuật và chất lượng của bạn”.
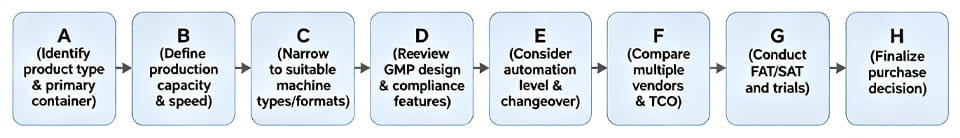
Sơ đồ lựa chọn
Để rõ ràng, đây là một cách đơn giản Sơ đồ các bước lựa chọn:
Các bước tiếp theo
Chọn đúng máy là một quá trình phức tạp nhưng bổ ích. Chúng tôi khuyên bạn nên viết chi tiết Đặc điểm kỹ thuật yêu cầu người dùng (URS) cho đội của bạn, liệt kê tất cả các tính năng phải có ở trên. Sau đó xin báo giá và bảng dữ liệu từ các nhà cung cấp có uy tín. Trước khi mua, yêu cầu một Kiểm tra nghiệm thu tại nhà máy Và, nếu có thể, dùng thử trang web với các sản phẩm thực tế của bạn.
Tại Máy đóng gói JinLu, chúng tôi chuyên về dây chuyền đóng gói dược phẩm từ đầu đến cuối. Sản phẩm của chúng tôi (chất độn viên nang, máy đóng gói vỉ, máy đếm, thùng giấy, vân vân.) được thiết kế để tuân thủ tiêu chuẩn GMP và độ chính xác. Chúng tôi khuyến khích bạn khám phá các trang sản phẩm của chúng tôi (nhìn thấy Liên kết nội bộ dưới) và liên hệ với các kỹ sư của chúng tôi để có giải pháp tùy chỉnh. Cho dù bạn cần một dây chuyền đóng thùng tự động, máy ép vỉ bán tự động, hoặc một dòng chai tích hợp đầy đủ, chúng tôi có thể hướng dẫn bạn trong quá trình lựa chọn và xác nhận.
Câu hỏi thường gặp về máy đóng gói dược phẩm
Chính xác thì “máy đóng gói dược phẩm” là gì??
Đó là bất kỳ thiết bị tự động nào được sử dụng trong ngành dược phẩm để đóng gói thuốc. Điều này có thể bao gồm các máy đổ chất lỏng vào lọ, đếm viên thuốc vào chai, thuốc phồng rộp trong khay, đóng thùng sản phẩm vào hộp, vân vân. Thiết yếu, bất kỳ máy móc nào giúp chứa, nhãn, niêm phong, hoặc thuốc bó là một phần của thiết bị đóng gói dược phẩm.
Tại sao tiêu chuẩn GMP lại quan trọng đối với các máy này?
Thực hành sản xuất tốt (GMP) tiêu chuẩn (FDA 21CFR, EU GMP, Hướng dẫn của WHO) đảm bảo sản phẩm của bạn luôn có chất lượng cao. Máy tuân thủ có thiết kế hợp vệ sinh (thép không gỉ 316L, bề mặt có thể làm sạch), kiểm soát được xác nhận (chính xác, hoạt động truy xuất nguồn gốc), và tài liệu phù hợp. Thiết bị không tuân thủ có nguy cơ gây ô nhiễm hoặc lỗi sản xuất có thể dẫn đến việc thu hồi hoặc trích dẫn theo quy định.
Những máy này giá bao nhiêu?
Chi phí rất khác nhau tùy theo loại máy và độ phức tạp. Một chiếc máy đếm thuốc để bàn bán tự động đơn giản có thể có giá vài nghìn USD, trong khi dây chuyền ép vỉ tự động tốc độ cao hoặc dây chuyền đóng chai tích hợp có thể tốn hàng chục, thậm chí hàng trăm nghìn.. Luôn xem xét tổng chi phí sở hữu (xem ở trên). Tốt nhất bạn nên lấy báo giá từ một số nhà cung cấp sau khi bạn đã xác định được yêu cầu của mình.
Máy đóng gói nào tốt nhất cho máy tính bảng?
Điều đó phụ thuộc vào định dạng của bạn. Để đếm viên thuốc vào chai hoặc túi, một máy đếm/làm đầy tự động là lý tưởng. Đối với bao bì theo đơn vị liều, một máy vỉ là phổ biến. Để đóng gói số lượng lớn trong chai, dây chuyền chiết rót và đóng nắp chai tự động (với một bộ đếm máy tính bảng ngược dòng) được sử dụng. Một số công ty cũng sử dụng máy đóng gói hoặc đóng gói để đóng gói từng viên thuốc. Đánh giá ưu/nhược điểm của từng hình thức (trị giá, sự bảo vệ, tùy chọn người dùng) trước khi chọn máy.
Sản phẩm của tôi nhạy cảm với độ ẩm. Tôi nên cân nhắc điều gì?
Đối với viên nén hoặc viên nang nhạy cảm với độ ẩm, bạn có thể cần dạng lạnh (Mọi thời đại) máy ép vỉ hoặc hệ thống hút ẩm chuyên dụng. Đảm bảo con dấu của máy là loại dược phẩm, và xem xét các phương án sấy tích hợp hoặc làm trơ nitơ. Bản thân vật liệu đóng gói (giấy bạc vs. màng PVC) đều quan trọng như nhau.
Tôi có thể sử dụng một máy cho nhiều sản phẩm được không?
Nhiều máy có thể điều chỉnh được phần nào, nhưng việc chuyển đổi giữa các sản phẩm khác nhau có thể là một thách thức. Chọn thiết bị có công thức/cài đặt cho các định dạng khác nhau. Không có công cụ, Công cụ thay đổi nhanh giúp việc xử lý nhiều SKU trên cùng một máy trở nên khả thi. Tuy nhiên, nếu bạn dự đoán có nhiều loại sản phẩm, máy mô-đun hoặc nhiều dây chuyền chuyên dụng có thể hiệu quả hơn.
Tài liệu tham khảo:
1.Hướng dẫn về bao bì đối với dược phẩm(Phụ lục 9) - AI.
2.Câu hỏi và Câu trả lời về Yêu cầu Thực hành Sản xuất Tốt Hiện hành | Thiết bị - CHÚNG TA. Cục Quản lý Thực phẩm và Dược phẩm.
3.Bao bì GMP tại 2026: Yêu cầu, Hướng dẫn, Điều khiển, và Bao bì Chính và Bao bì Thứ cấp – pharmuni.com.
4.Các quy định và tiêu chuẩn đóng gói toàn cầu - Điều hướng GMP.








