UN turnkey packaging line is a fully integrated, end-to-end packaging system delivered by a single supplier. It covers every stage of production – from initial product feeding to primary packaging, emballage secondaire, and end-of-line boxing/palletizing – and is handed over ready to run under one User Requirements Specification (URS). En pharmacie, turnkey lines ensure single-point responsibility, faster commissioning, and full GMP compliance by design. This article explains the modules of a turnkey packaging line, how it works (flow chart), avantages clés, and how it differs from a multi-vendor setup. We also cover typical pharmaceutical applications, supplier selection tips (with an example of Jinlu’s capabilities), and FAQs.

A true turnkey line goes beyond stitching machines together – one vendor designs, builds, delivers, and validates the entire system under one contract. Par contre, a traditional approach might buy individual machines from different suppliers and try to integrate them later, which often causes delays, extra costs, and validation headaches. The turnkey solution includes automation from bulk product handling through final case packing, along with unified controls, data connectivity, et documentation. Par exemple, Jinlupacking defines a turnkey blister line as covering tablet feeding, blister forming/sealing, printing/coding, mise en carton, and case packing all delivered as one validated system. En pratique, this concept extends to any pharmaceutical format (comprimés, gélules, bouteilles, flacons, sachets, etc.) – meaning one supplier delivers a complete, ready-to-run packaging system rather than a collection of separate machines.
What Does a Turnkey Packaging Line Include?
A turnkey packaging line bundles all the modules needed to go from raw pharmaceutical product (par exemple. comprimés, gélules, poudre, liquide) to fully packaged unit-dose packs and boxed cartons. Key modules typically include:

- Product Feeding & Manutention: Automated feeders or bowl systems that take bulk tablets/capsules/vials/pouches and feed them one-by-one into the line at a controlled rate. For solids, this might be a vibratory pan feeder or capsule unscrambler; pour bouteilles, an unscrambler and conveyor; pour liquides, an orientation/accumulation station. Proper feeding ensures consistent flow into the first packaging machine.
- Emballage principal: The first level of package that directly contains the product. This varies by product type: blister machines for tablets/capsules (thermoforming and sealing foil/pvc into blister packs); liquid-filling machines for vials/ampoules (piston or peristaltic fillers with capping or crimping); tube fillers for gels/creams; sachet or stick-pack machines for powders or granules; pre-made pouch fillers; bottle fillers and cappers for oral liquids; etc.. Each primary packer is designed for the specific dosage form and fill material. Par exemple, blister formers heat PVC film to create pockets for pills, while cold-form (De tout le temps) blister machines press aluminum foil for moisture-sensitive drugs. Every primary step must precisely match the product and foil/pouch/bottle specifications.
- Emballage secondaire (Cartonnage): After primary packing, secondary machinery wraps, boîtes, or cartons the primary packs. Common machines include encartonneuses, which erect folding cartons and insert blister cards or vials along with patient information leaflets. There are side-load (horizontal) and top-load (verticale) cartoners depending on format. Other secondary equipment may include étiqueteuses (apply compliant labels on bottles/cartons), fardeleuses, ou sleeve wrappers to bundle packs. Par exemple, a blister card emerges from the blisterer and is automatically fed into a encartonneuse where a carton is erected, the patient leaflet is inserted, and the carton is sealed. The timing between blister and cartoner is tightly synchronized in a turnkey line to avoid jams – this is handled by PLC controls (see Integration below).
- End-of-Line Packaging: These are tertiary packagers for bulk handling – case packers and palletizers. The case packer takes filled cartons (ou bouteilles) and loads them into corrugated shipping cases (RSC cartons), then seals the case. Palletizers stack the cases onto pallets. Together they prepare product for distribution. Modern case packers often include weight-checking and label-applying stations to verify contents and apply master labels. In a true turnkey solution, even these final steps are integrated so that data (like lot numbers, poids, pallet IDs) flows through to the batch record. Par exemple, at the case pack station the system can weigh each case and apply a label with the case ID, linking it back to the serialized batch.
- Inspection & Contrôle de qualité: Integrated vision and testing equipment ensure compliance at each stage. Typical stations include track-and-trace coding/inspection (printing and verifying batch codes, dates d'expiration, DataMatrix, etc.) et in-line inspection systems (vision de la caméra, détecteurs de métaux, checkweighers or X-ray). Par exemple, a turnkey blister line will print a batch code and 2D DataMatrix on each blister card and immediately verify it with a camera. The system rejects any blister with a missing or illegible code without stopping the whole line. Without a truly integrated line, a bolt-on printer may not reliably keep up, leading to audit failures. Other QC features like fill-level checks, seal integrity tests (par exemple. leak testers for vials), and final carton weight checks are often embedded. All sensors and detectors are tied into the central PLC/HMI, ensuring that any fault flags a hold in that exact unit within the main controller, not a separate computer.
- Intégration & Automation Control: Beyond physical modules, a turnkey line includes the complete automation system – one PLC-based control network, one operator interface (IHM), and fully integrated electrical/software design for the entire line. Rather than each machine having its own controller, the whole line usually runs under a single PLC architecture (par exemple. Siemens S7 running all stations) and one supervisory HMI. This allows line speeds to be matched machine-to-machine, buffers minimized, and data logged centrally. All recipe parameters, speeds, and recipes can be accessed from a unified touchscreen. Surtout, the integration provider writes a single set of control software, does one Factory Acceptance Test (GRAISSE) on the full line, and delivers one validation package (IR/WH/PQ). This ensures the line “hands off” to production in one step. In a proper turnkey line, the mechanical, electrical, and software systems are all delivered together by the integrator, so that “the carton run under unified PLC control, not three separate supplier protocols”.

En résumé, un turnkey packaging system for pharma includes everything from bulk feeding to final case packing, plus coding/inspection equipment and a unified control system. This covers three levels of packaging (primaire, secondaire, tertiaire) in one cohesive line. The turnkey approach means you get one engineered project – one URS, one FAT/SAT, one set of validation documents – rather than several machines to fit together on your floor.
How Does a Turnkey Packaging Line Work?
A turnkey line is carefully designed as a continuous process flow. Products enter in bulk, undergo primary and secondary packaging, and exit as palletized cases. The entire sequence is choreographed by the control system. Below is a simplified process flow diagram:
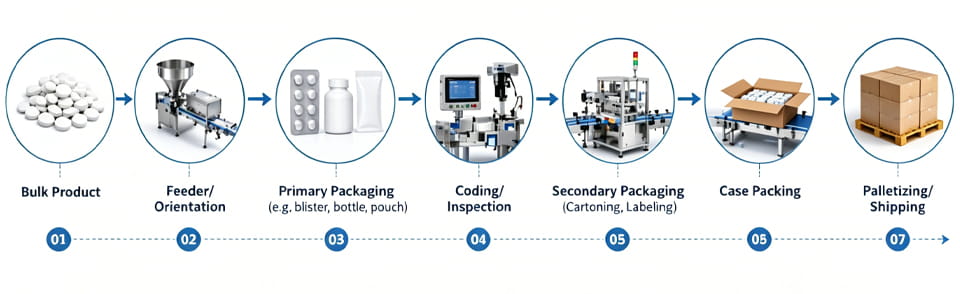
- Bulk Product → Feeder/Orientation: Bulk tablets or capsules are fed into the line via vibratory feeders or unsramblers, which orient the products properly. In bottle lines, unscramblers arrange empty bottles on a conveyor. This stage meters product into the first packer at the exact speed needed.
- Emballage principal: The product enters its first container. Par exemple, tablets go into a emballeuse sous blister where film is formed into pockets and sealed with foil. Liquids might be filled into vials and capped. Powders could be dispensed into stick sachets. The parameters (vitesse, volume de remplissage, seal temperatures) are all pre-programmed. Each step records data (par exemple. poids de remplissage, seal force). In a turnkey line these machines are physically bolted together, so product moves seamlessly from one to the next with no manual intervention.
- Codage & In-Process Inspection: Immediately after primary packing, the line prints serialization and batch codes on each unit. A vision camera reads and verifies codes in real time. If a code is wrong or a pack is damaged, an in-line rejector or bypass automatically removes it. Cela garantit 100% traçabilité. A stand-alone printer would not guarantee this tight integration. The control system logs all print data into an electronic batch record (21 Partie CFR 11 conformité).
- Emballage secondaire (Cartoning/Labeling): Suivant, packs primaires (cartes blister, flacons, pochettes) move into a encartonneuse ou labeling station. A cartoner erects a carton, inserts the product and the printed package insert, then closes and seals the box. Again, this is linked to the main PLC: the cartoner only runs as fast as the blisterer supplies product (buffers are minimized). Modern lines use dual-head or multi-lane cartoners to match high-speed blister output. Labels (if separate) are automatically applied with the same batch/expiry codes. Because all machines share recipes and speed settings in one HMI, changeovers (par exemple. new carton size or product code) are managed uniformly.
- Case Packing & Palletizing: Enfin, sealed cartons proceed to an automated case packer. Here carton cases are erected, groups of cartons are loaded (often in layers), and the case is sealed. The system typically checks case weight and applies a case label linked to the serialized cartons inside. Palletizers then stack the cases on pallets and may wrap/shrink-wrap them. Each pallet is also labeled and traced back. The automation software ties all this together: you get a complete history from bulk input to pallet ID, ensuring regulatory traceability.
Throughout the line, the unified control strategy is key: one PLC system ensures that line speed is balanced, OEE is optimized, and any fault stops only the relevant section, not unrelated machines. Downtime due to line layout or incompatible controls is virtually eliminated in a properly designed turnkey line. An integrated line eliminates downtime caused by the line layout, conception, machine placements or from the controls system, focusing stoppages only on actual machine faults. En pratique, this means the packaging process flows continuously under one “brain,” resulting in higher productivity and predictability.
[jl_youtube src= »https://www.youtube.com/embed/zRe4BB8i9n0″]
Key Benefits of a Turnkey Packaging Line
Adopting a turnkey packaging line brings several advantages over piecemeal setups:
- Single Point of Responsibility: One supplier designs, ingénieurs, and delivers the whole line, and takes responsibility for its performance. You sign one contract and one validation package covering all stations. This greatly reduces management complexity. If something goes wrong at startup or during production, you deal with one integrator rather than negotiating fixes between multiple vendors. As Jinlupacking warns, “the most expensive mistake…is buying the right machines from the wrong vendors”. With a turnkey solution, the supplier “owns the entire process – mechanical, electrical, and software integration”.
- Faster Commissioning & Time-to-Market: Because the line is built and debugged in one place (the supplier’s factory) and shipped fully assembled, commissioning is much quicker. The integrator runs a single FAT/SAT protocol for the entire line. En revanche, multi-vendor projects often see long delays as machines arrive separately and integration issues emerge on-site. Turnkey projects typically go from delivery to production readiness in semaines, not months. Par exemple, Standard turnkey lines can be integrated in 45–60 days, even complex lines in 90–120 days. This compressed timeline means you can hit your launch targets faster.
- Guaranteed Throughput & Efficacité: A turnkey line is engineered to meet your target speeds end-to-end. Since all machines and conveyors are designed together, throughput is optimized (no unseen bottleneck). The supplier can guarantee a certain output (par exemple. “200 blisters/min”) for the full line, not just individual machines. This means higher Overall Equipment Effectiveness (OEE). Turnkey design consistently deliver optimized OEE performance levels by preventing stop-start issues and starvation or blocking in the line. En pratique, a line laid out by one vendor will match speeds and use a single HMI for the entire process, avoiding the costly buffers or manual transfers required in disjointed systems.
- Compliance and Validation Ready: Turnkey lines are built with pharma GMP from the ground up. Design features like all-stainless frames (SUS316L where needed), sanitary layout (no dead legs), and CIP/SIP capability can be specified and delivered. Plus important encore, the documentation comes complete: you receive one comprehensive validation dossier (GRAISSE, QI, QO, PQ) covering the entire line. As notes, a turnkey project includes one IR/WH/PQ for all stations. The line inherently meets GMP standards (21 Partie CFR 211, Annexe UE 1, etc.) and electronic records requirements (21 Partie CFR 11 for data logging). Par exemple, the blister sealing station will log temperature/pressure/dwell per batch to an electronic record, and the case packer will embed serialization data on each case. With a turnkey supplier, you don’t need to cobble together certificates from multiple vendors – all compliance engineering is unified.
- Lower Total Cost of Ownership (TCO): While turnkey lines often require higher initial investment, they save money over time. Because all equipment shares common spare parts and electrical standards, spare inventories are simpler. Service contracts and spare parts can often be with a single source, which can lower overall maintenance costs. According to one industry analysis, a turnkey line typically has “higher upfront, lower total” cost of ownership. En revanche, multi-vendor lines can incur hidden integration costs, multiple spare-part lists, repeated requalification, and costly downtime during troubleshooting. A single-SLA support agreement for the entire line also means faster service. Over a few years, buyers often find that paying a bit more up front for full integration is offset by higher uptime and predictable support.
- Scalability and Customization: Turnkey suppliers can tailor the line to your exact product mix and plant layout. Need a line that can switch from 3ml vials to 10ml bottles? A turnkey integrator will build in changeovers (shared recipes, pièces à changement rapide) from the start. They will also fit the line geometry (par exemple. U-shape for tight spaces) and include all requested features (chargement robotisé, isolator, etc.). This level of customization means you get exactly what you need – an advantage if your product range or facility size is unique. Par exemple, Offres d'emballage Jinlu tailored turnkey lines that match a client’s space and process requirements.

Pris ensemble, these benefits make turnkey packaging lines very attractive for pharma. You avoid much of the risk and uncertainty that comes with buying fragmented equipment. Comme le dit un expert, a genuine turnkey contract means “one FAT/SAT, one validation package, and one team on-site until cGMP production is running” – a clear formula for protecting your launch timeline.
Turnkey vs Multi-Machine: Comparaison côte à côte
The table below contrasts a single-supplier turnkey line with a traditional multi-vendor setup. It highlights why the turnkey approach is often preferred in regulated industries:
| Fonctionnalité |
Turnkey Line (Single Supplier) |
Multi-Vendor System |
| URS & Conception |
One URS covers entire line; supplier owns compliance. All design gaps handled. |
Separate URS documents per machine; buyer must manage integration. |
| Contrôles & Intégration |
Unified PLC, one HMI screen; one communication protocol manages all stations. |
Each machine uses its own controller/PLC. Integration done by buyer/integrator post-delivery, often with costly middleware. |
| FAT/SAT |
Single FAT and SAT documentation for the full line at target throughput. |
Per-machine FATs; full line SAT may be extra effort or missing. Buyer coordinates multiple test reports. |
| Validation (IR/WH/PQ) |
One master validation plan and dossier. No hand-off uncertainties in qualification. |
Individual validation packets per machine; buyer must align protocols and data in-house. |
| Throughput Guarantee |
Vendor guarantees end-to-end speed (no unexpected bottlenecks). |
Each machine rated separately. Overall line often limited by slowest unit, discovered only after assembly. |
| Traçabilité |
Seamless end-to-end serialization & audit trail from primary pack to case. |
Data typically not linked across machines. Custom data transfers or manual logs needed, risking gaps. |
| Accountability |
Single service contact and SLA for entire line. One integrator fixes any issue. |
Multiple vendors; in case of downtime each blames others. Buyer must troubleshoot integration problems. |
| Commissioning Time |
Fast startup: typically few weeks from delivery to production. |
Slow integration: common delays of 2–3 months or more as PLCs and speeds are reconciled. |
| Coût total de possession |
Higher initial price but predictable spares/support. Lower 3-year TCO. |
Lower upfront cost; higher long-term costs due to extra downtime, revalidation, spare inventories, and integration fixes. |
Tableau: Key differences between turnkey (single-supplier) lines and multi-vendor assemblies.
This comparison (adapted from industry data) shows why many pharma companies prefer turnkey solutions despite their higher initial quote. With one system integrator accountable for everything, you avoid the finger-pointing and schedule overruns that plague multi-vendor projects.
Applications of Turnkey Packaging Lines in Pharma
Turnkey packaging lines can be configured for virtually any pharmaceutical product. Common applications include:
- Tablet and Capsule Lines: Highly automated blister lines (PVC or Alu-Alu blister machines + encartonneuses) or bottle lines (tablet counters + filling/capping + étiquetage + mise en carton). Par exemple, a tablet production line might include a tablet press, a multi-head counting machine (filling bottles), an induction capper, Étiquetage, and a cartoner – all integrated as one system. Turnkey blister lines (tablet to carton) and turnkey bottle lines (tablet counter to case packer) are typical for solid-dose drugs.
- Liquide & Injectable Lines: Complete lines for sterile liquids (flacons, ampoules, syringes) are a popular turnkey project. These often include a washer sterilizer, remplissage, stopper inserter, crimper (pour flacons), or vacuum filling/capping (for ampoules), plus lyophilizer loading conveyor, wash/dry, and autoclave integration if needed. Après avoir rempli, the line moves to inspection, étiquetage, and cartoning. Par exemple, an integrated syringe line may automatically assemble, étiquette, and cartoned syringes as shown by one solution provider. En résumé, turnkey sterile filling lines ensure all steps from aseptic fill to final carton are validated together.
- Powder and Granule Packaging (Sachets/Stick Packs): We frequently see turnkey solutions for unit-dose powders (antiacides, oral rehydration, etc.) and blends (produits nutraceutiques). These lines combine a stick or sachet filling machine with a multi-lane cartoner and case packer. Par exemple, Jinlu multi-lane stick-pack cartoning line integrates four machines (stick pack fillers, weigher, grouping conveyor, and cartoner) pour poudres, granulés, liquides ou gels. Such a turnkey system can be customized from 2 à 16 voies, meeting high output needs while ensuring GMP design.
- Solid-dose in Pouches/Bags: Some medicines (par exemple. tablets or powders) are packed in pre-made pouches. A turnkey pouch line would include a counting unit (for tablets or capsules) or auger filler (for powder), a pouch filling machine, scellage, and then secondary cartoning and case packing. These lines are less common but follow the same principle of complete integration.
- Medical Device & Combination Kits: Turnkey integration extends to device assembly and kits. Par exemple, a line could assemble injection pens, label and package them into clamshells or cartons, including automated inspection.
- Sterile and Aseptic Processing: In biotech/pharma, entire aseptic fill–finish suites can be delivered turnkey, including isolators or RABS (restricted access barriers), automated fill lines, and integrated CIP/SIP systems. These are large-scale projects (sometimes called “facility in a box”) but follow the turnkey model: one engineering firm handles all utilities, salle blanche, et équipement.

En pratique, turnkey packaging systems are most prevalent in pharma and healthcare, but are also widely used in related industries like nutraceuticals, produits de beauté, and even food where clean, automated processes are needed. Within pharma, any product that demands high speed, précision, and GMP traceability – tablets, gélules, flacons, syringes, sachets – can be handled by a turnkey line.
How to Choose the Right Turnkey Packaging Line Supplier
Selecting the right supplier is as critical as choosing the line itself. Here are key criteria for buyers:
- Industry Experience & Références: Look for a vendor with proven experience in pharmaceutical packaging projects. They should have études de cas or client references for lines similar to yours. A supplier familiar with pharma regulations (cgMP, FDA/EMA inspections) will better understand your requirements. Ask for examples of past turnkey line installs and performance results.
- Capacité de personnalisation: Every product and facility is different. A good integrator will adapt machines to your needs (par exemple. custom feeders, special sterilizable components, or niche materials). As Jinlu highlights, they “offer turnkey lines tailored to space and process needs”. Inquire how the supplier handles unique products, multi-SKU runs, or cleanroom constraints. See if they can modify layout (U-shape for tight areas) ou de l'équipement (multiple lanes, custom hoppers).
- Validation & Compliance Support: The supplier should provide a full validation package. Confirm they will furnish factory acceptance tests and ready-to-use IQ/OQ protocols. They should also ensure compliance: Par exemple, Jinlu emphasizes that their lines are designed for GMP compliance and come with documentation. Check that the system will support regulatory requirements (electronic records under 21 Partie CFR 11, traçabilité, nettoyabilité). Idéalement, the supplier helps with regulatory filings (par exemple. providing USP <1207> leak test plans, Annexe 1 compatibilité).
- Service après-vente & Soutien: Quick service and spare parts are vital. Does the vendor have local representation or global service teams? Can they offer remote support? Comme le conseille un guide, see if a company can rapidly ship parts or send engineers to minimize your downtime. Warranty length, response time guarantees (ANS), and availability of parts for your region are key. Suppliers like Jinlu often highlight 3-year warranties and 24/7 soutien. A vendor with multilingual manuals and spare parts depots near you is preferable.
- Coût total & Empreinte: Au-delà du prix de la vignette, consider the total life-cycle cost. A supplier who plans for easy maintenance (standard spares, modular parts) can save money long-term. Also verify the line’s physical footprint and utility needs: a good integrator will adapt the layout to fit your facility (par exemple. rotating line, mezzanine conveyors). Don’t overpay for unused capacity – match the line’s speed and size to your current and projected volume. Inversement, ensure there’s room to grow if demand increases.
- Corporate Credentials: Check the manufacturer’s certifications: ISO 9001 (quality management) et ISO 13485 (medical device quality) sont des bonus. For pharma-specific concerns, ensure they acknowledge CE requirements and applicable electrical/EMC standards. (CE marking is mandatory in Europe and often required elsewhere to show basic safety compliance.)
Exemple: Machines d'emballage Jinlu, par exemple, markets ourself as an “end-to-end” pharma line provider. Our team can supply charges de capsule, emballeurs de blisters, machines à compter, encartonneuses, etc., all built to GMP standards. We work closely with clients on layout and automation. When evaluating Jinlu or any vendor, ask for detailed quotes based on your URS and compare their proposed integration approach.
Conclusion
En résumé, un turnkey packaging line is an all-inclusive, single-vendor solution for emballage pharmaceutique, handling everything from bulk feeding to final palletizing. It ensures high efficiency, Conformité BPF, and simple project management by delivering one integrated system under a unified URS and validation plan. Key benefits include faster startup, better line performance, and single-point accountability. For pharma manufacturers, a turnkey approach dramatically reduces the risks and headaches of multi-vendor integration. When evaluating turnkey offerings, focus on supplier expertise, personnalisation, and support for GMP validation. With the right partner (par exemple. Jinlu Packing’s end-to-end solutions or similar turnkey providers), you can achieve a smooth, compliant packaging process that is ready to run from day one.
FAQs about Turnkey Packaging Lines
What is the difference between a turnkey line and an integrated packaging line?
In essence they refer to the same concept: a turnkey line is an integrated packaging system delivered ready-to-run. The difference is more in phrasing. A “turnkey” line emphasizes that one supplier handles the whole project under one contract, whereas an “integrated line” stresses that multiple machines are tied together into one system. Both imply full automation from start to finish. Cependant, sometimes “integrated line” can still involve multiple vendors; “turnkey” explicitly means one supplier. The key benefit of turnkey is the single-point accountability and unified validation.
Is a turnkey packaging line suitable for a small or niche pharma company?
Oui, turnkey lines can be scaled to fit smaller operations. Modern integrators are very flexible in design. For limited spaces (par exemple. a small R&D plant or pilot line under 40 m²), vendors offer compact layouts – such as monoblock machines or U-shaped conveyor lines – to minimize footprint. Even if your output is modest, a semi-automatic or single-lane turnkey line can provide big efficiency gains (and easier future scale-up) by reducing manual steps. It’s true the upfront cost might seem high for a small line, but you save time on validation and get faster ROI through efficiency. En bref, turnkey solutions are not only for large factories – they just need to be right-sized by the supplier.
How long does it take to implement a turnkey packaging line?
Turnkey line projects do take time to engineer properly, but integration is faster overall than multi-vendor setups. For a moderately complex line, you might expect 3–6 months from finalizing the design (URS) to factory testing, and then another few weeks on site for installation and SAT. Some vendors report standard turnkey modules ready in 45–60 days, with highly custom lines in the 90–120 day range. You will work closely with the supplier during this period: defining product parameters, approving layouts (often in 3D CAD), then witnessing the FAT at their factory. Keep in mind also the time needed for site preparation (utilitaires, salle blanche). In total, a turnkey line is often completed in under a year for mid-sized projects, whereas a patchwork of separate machines could drag out well beyond that.
What certifications or standards should I look for in a pharmaceutical packaging line?
Packaging equipment itself must meet industrial safety and electrical standards (Par exemple, CE marking in Europe, UL or CSA in North America, and EMC/LVD directives). En pharmacie, there is no single “GMP” certificate for machines, but the design must follow GMP principles (conception sanitaire, validation-ready software, etc.). Donc, ensure the supplier provides a validation package for the line (FAT reports, Protocoles QI/OQ). The software should support FDA 21 Partie CFR 11 (pour les enregistrements électroniques) et Annexe BPF de l'UE 11. De nombreux fabricants (comme Jinlu) highlight CE marking on their machines, which is legally required to sell in Europe. En plus, ask for ISO 9001 (quality management) certification of the manufacturer, and any relevant customer quality audits. En pratique, compliance means your line meets 21 CFR 211 (packaging controls) and Annex 1 (sterile products) standards by design.
Which industries commonly use turnkey packaging lines?
Aside from pharmaceuticals, turnkey packaging lines are used wherever high-speed, automated packaging is needed. This includes nutraceuticals (vitamines, suppléments), produits de beauté, nourriture (especially baby food or confectionery), et produits chimiques. Par exemple, a food manufacturer might install a turnkey frozen goods line similar in concept to a pharma line – integrating portioning, primary wrapping, carton packing, and palletizing. In healthcare specifically, any company looking to streamline production – from big pharma to niche biotech – can benefit from turnkey automation.
Are turnkey packaging systems customizable?
Absolument. Turnkey lines are often designed to each customer’s product, volume, and space. Suppliers routinely offer custom lanes, machine configurations, and special features (par exemple. robotic pick-and-place, isolators, nitrogen purging) as part of the package. The term “custom turnkey” is sometimes used to emphasize this flexibility. What is not customizable is the single-contract nature: even if you add custom machines or modify layouts, it all comes under the turnkey delivery. This means the supplier still provides one integrated system.
Références:
1.21 Partie CFR 211 – Subpart G – Packaging and Labeling Control —— Legal Information Institute
2.CPG Sec. 450.500 Tamper-Resistant Packaging Requirements for Certain Over-the-Counter Human Drug Products —— U.S. Administration des aliments et des médicaments
3.Six Guidelines for Turnkey Packaging Line Integration —— Packaging World
4.Annexe BPF de l'UE 15: Qualification and Validation —— gmp-compliance.org
5.Annexe 9 Lignes directrices sur l'emballage des produits pharmaceutiques -- OMS







