
L'achat de nouveaux équipements pharmaceutiques ne représente que la moitié du travail : la vraie question est la suivante :: est-ce que ça peut passer le QI, QO, et PQ?
Dans fabrication pharmaceutique, une machine qui ne peut pas être correctement qualifiée est un risque, pas un atout. Les régulateurs attendent des preuves claires que l’équipement est correctement installé, fonctionne dans des limites, et fonctionne de manière constante dans des conditions de production réelles . Sans ça, même la ligne de conditionnement la plus avancée ne peut pas être utilisée dans la production GMP.
C'est pourquoi comprendre le QI, QO, et PQ n'est pas seulement un détail technique : il a un impact direct sur votre conformité, calendrier du projet, et si votre investissement génère réellement de la valeur.
QI, QO et QP défendre Qualification d'installation, Qualification opérationnelle, et Qualification des performances. Il s'agit d'étapes séquentielles de qualification des équipements avant utilisation dans la production pharmaceutique GMP.. En bref: QI vérifie que la machine est correctement installée; QO teste qu'il fonctionne correctement sous tous les paramètres requis; PQ vérifie qu'il fonctionne de manière cohérente pendant la production réelle. Selon la FDA, « qualification » désigne la preuve que l'équipement est adapté à l'usage auquel il est destiné.. IQ/OQ/PQ sont les activités documentées pour prouver chaque étape d'adéquation et de fonction. Ensemble, ils font partie de l'ensemble validation procédé pour un système de fabrication, garantir la qualité et la conformité des produits.
Qualification des équipements vs. Validation du processus: IQ/OQ/PQ se concentrent sur la machine (la « qualification des équipements »), tandis que la validation des processus couvre l'ensemble du processus de fabrication. En pratique, la qualification des équipements est souvent une première étape dans un plan directeur de validation. En complétant IQ/OQ/PQ sur un remplisseur de gélules, machine à ampouler, ligne de bouteilles, etc., un fabricant obtient des preuves documentées que l'équipement répond aux spécifications et est prêt pour la production.
| Scène (IR/WH/PQ) | But | Activités clés / Essais | Documents typiques produits |
| QI | Vérifier la bonne installation des équipements | Vérifier l'emplacement, utilitaires (pouvoir, air, eau), installation par manuel; enregistrer les numéros de série et les accessoires | Protocole/liste de contrôle et rapport IQ |
| QO | Vérifier les fonctions opérationnelles dans les limites | Exécutez des tests sur toutes les plages de fonctionnement (vitesses min/max, températures, pressions); tester les alarmes et les verrouillages, vérifier les contrôles | Protocole et rapport OQ |
| PQ | Prouver des performances constantes en production | Exécutez plusieurs lots de production avec le produit réel (ou faire semblant) dans les pires conditions; vérifier les sorties (rendement, précision) | Protocole et rapport PQ |
Tableau 1: Comparaison du QI, QO, Étapes PQ (adapté des normes de l'industrie).

Équipement pharmaceutique doit se conformer à des réglementations strictes en matière de BPF. Aux États-Unis, FDA 21 Partie CFR 211 (pharmaceutique) et 21 CFR 820 (dispositifs médicaux) exiger explicitement la validation des systèmes de fabrication. IQ/OQ/PQ constituent l’épine dorsale de cette conformité. Par exemple, la FDA déclare que les activités de qualification « précèdent nécessairement la fabrication des produits à l’échelle commerciale ». Dans l'UE, Annexe BPF 15 exige également la qualification des équipements tout au long de leur cycle de vie. L’objectif est de garantir que l’équipement produira de manière fiable des produits sûrs.
Sans QI/OQ/PQ approprié, les entreprises sont confrontées à de sérieux risques: défauts du produit, rappels de lots, citations réglementaires, et les risques pour la sécurité. Une qualification appropriée renforce la certitude que la machine en fait fait ce qu'il est censé faire – par exemple, qu'un blister scelle à la bonne température ou qu'un remplisseur de liquide distribue le volume correct. En pratique, un programme de validation complet permet d'éviter les surprises: "Quand c'est fait correctement, les trois phases de qualification sont minutieuses et chronophages. Cependant, ils sont essentiels pour garantir des processus reproductibles et une qualité de produit stable ». En bref, IQ/OQ/PQ sont obligatoires dans les paramètres GMP. Pour tout ligne de conditionnement (cloque, bouteille, carton, etc.), IQ/OQ/PQ sont requis pour répondre aux exigences de la FDA et de l'UE. Les machines Jinlu sont livrées avec des modèles de documentation complets IQ/OQ/PQ, parce que, comme disent nos experts, "la machine doit être prête pour la validation".
Dans le Phase de QI, nous vérifions que le matériel est livré et mis en place correctement. Cela signifie inspecter la machine et son environnement avant tout test de production.. Les principaux contrôles de QI incluent:
Le résultat d’IQ est un rapport/protocole IQ écrit qui documente chaque vérification et confirme « l’installation comme prévu ». Si quelque chose est hors spécifications, il faut le corriger avant de continuer. Un IQ réussi signifie que « l’équipement est installé comme prévu » – une condition préalable à tout test. (En pratique, certains tests peuvent commencer dans le QI en tant qu'éléments « prêts pour le test », mais les critères d'acceptation viennent en OQ/PQ.)
OQ teste les fonctions de l’équipement dans des conditions contrôlées. Maintenant que la machine est correctement installée, nous vérifions tous les paramètres de fonctionnement et systèmes de contrôle. OQ comprend généralement:
Le Rapport blanc montrera les résultats de chaque test et les comparera aux critères d’acceptation (qui proviennent de spécifications de conception ou d'URS). Ce n'est qu'après le passage de l'OQ que nous procédons. (Parfois, OQ et IQ peuvent se chevaucher dans un rapport combiné appelé IOQ., mais la logique reste séquentielle.)
Un objectif clé d’OQ est: « Dans toutes les conditions spécifiées, la machine fait-elle ce qu'elle est censée faire?» Si quelque chose échoue (par exemple. un capteur est mal aligné), c'est corrigé et testé à nouveau.
Le PQ est la dernière étape, où nous prouvons que l'équipement fonctionne correctement pendant la production réelle. Alors qu'OQ aurait pu être vide, PQ utilise un produit réel ou simulé. Les étapes comprennent:
À la fin du PQ, nous avons la preuve documentée que "L'équipement qualifié produit systématiquement un produit conforme dans des conditions réelles." Autrement dit, la machine est prête pour la production GMP.
Par exemple, si PQ réussit sur une machine de remplissage de gélules, nous savons que sa précision de dosage, insertion de capsules, et la vitesse du rotor fonctionne sans dérive. Cela complète le « triangle » de validation et donne l’assurance au service qualité/réglementation que la ligne ne produira pas de mauvais lots.. Ensuite, l'équipement est libéré pour la fabrication de routine et inclus dans les programmes de maintenance réguliers..

Il est important de clarifier la terminologie. Qualification fait généralement référence à l'équipement, alors que validation fait souvent référence à des processus ou des systèmes. Dans de nombreux frameworks GMP, IQ/OQ/PQ sont pris en compte parties de validation d'un système de fabrication. En ce sens:
Ainsi, équipement IQ/OQ/PQ qualifie les machines. Un séparé Validation du processus relierait ces machines au processus de production réel (comme la compression et le revêtement des comprimés, etc.). En fait, IQ/OQ/PQ sont parfois appelés Qualification des équipements (égaliseur) mesures. UN Qualification de conception (QD) l'étape peut précéder le QI, s'assurer que la conception répond aux exigences des utilisateurs (URS) avant de construire ou d’acheter du matériel.
Voici un flux simple des phases du cycle de vie d’un projet:
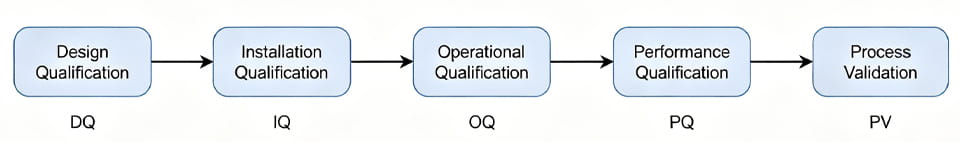
Chaque flèche est un transfert: tu dois finir DQ (vérifier que la conception répond à l'URS) avant le QI (vérifier la bonne installation). Ce n'est qu'une fois la PQ terminée que vous pouvez affirmer que l'équipement est validé et commencer la validation du processus. (par exemple. mener des campagnes de production réelles).
La qualification des équipements fait partie de l'équipement global cycle de vie:
Chaque étape produit une documentation. Par exemple, Les machines d'emballage Jinlu sont livrées avec un Kit de validation QI/OQ/PQ et protocoles FAT/SAT. Cela signifie que l'acheteur peut gagner du temps en utilisant les modèles du fabricant lors de la qualification.. Un calendrier typique peut s'étendre sur des semaines, voire des mois, selon la complexité..
Selon FDA/UE, équipes d’assurance qualité et d’ingénierie formées (et parfois des consultants extérieurs) devrait mener ou être témoin de ces étapes. L'essentiel: un discipliné, un flux documenté depuis la conception jusqu'au PQ garantit que votre ligne est conforme.
Chaque type de machine d'emballage dans une usine GMP doit être qualifié. Voici des exemples courants:
Image: Une remplisseuse de gélules semi-automatique (Jinlu CGNT-209). La validation de ces machines implique des contrôles minutieux de l’orientation des capsules, précision du dosage, et scellement pendant IQ/OQ/PQ.
Exemple de cas: Machine de remplissage de gélules IQ/OQ/PQ
Pour illustrer, imaginez qualifier un remplisseuse de capsules entièrement automatique pour les vitamines:
Lorsque ces étapes sont terminées avec des résultats satisfaisants, le remplisseur de capsules est qualifié pour la production. Il est prêt à commencer à emballer des produits réels en toute confiance.
Malgré son importance, la qualification des équipements se heurte souvent à des obstacles:
L'expérience de Jinlu est la suivante la meilleure façon de surmonter ces défis est de planifier. Commencez tôt la planification de la validation, définir l'URS et le plan de test avec le fournisseur, et s'assurer que toutes les parties prenantes s'accordent sur les critères. L'utilisation de modèles et de listes de contrôle accélère également le processus.

Pour rationaliser IQ/OQ/PQ et garantir un résultat conforme:
En suivant ces bonnes pratiques, les entreprises peuvent réduire les surprises lors des audits. Comme le souligne souvent le fondateur de Jinlu, Petty Fu: choisir des machines auprès de fournisseurs GMP expérimentés « garantit qu'ils sont livrés avec un résultat précis, certificats de conformité, et un soutien local ». Autrement dit, validez en toute confiance et choisissez un partenaire qui parle déjà validation.

Dans la fabrication pharmaceutique, QI, QO, et PQ ne sont pas négociables. Ces étapes de validation structurées prouvent que votre équipement – des remplisseuses de gélules aux conditionneuses sous blister en passant par les lignes de bouteilles – est adapté à son usage et est conforme aux BPF.. Un programme IQ/OQ/PQ bien exécuté permet d’éviter des erreurs coûteuses et garantit une qualité constante.
Choisir un fournisseur avec un solide support de validation est essentiel. Machines de conditionnement pharmaceutique de Jinlu Packing venir avec Conception prête pour les BPF et documentation complète (Protocoles FAT/SAT/IQ/OQ/PQ) pour rationaliser la conformité. Notre équipe peut vous aider à définir l'URS, effectuer des tests de QI/OQ, et générer les rapports requis.
👉 Vous recherchez équipement pharmaceutique qui simplifie la conformité? Contactez Jinlu aujourd'hui pour les machines d'emballage avec un support complet IQ/OQ/PQ et des services de validation clé en main. Nous veillerons à ce que votre nouvelle ligne soit installée, testé, et documenté pour une certification GMP sans tracas.
En termes simples, Qualification d'installation (QI) s'assure que l'équipement est installé correctement (bon emplacement, utilitaires, et composants). Qualification opérationnelle (QO) teste les fonctions de l’équipement dans des conditions normales et extrêmes (confirmer tous les contrôles, alarmes, et les limites). Qualification des performances (PQ) confirme que l'équipement peut fonctionner de manière constante dans la production réelle, en utilisant des matériaux et des conditions réelles. Ensemble, Document IQ/OQ/PQ attestant que la machine fonctionne comme prévu.
Oui. Lignes directrices réglementaires (FDA, UE, OMS) exiger que tous les équipements critiques d’une usine pharmaceutique soient qualifiés avant utilisation. Par exemple, FDA 21 CFR 211 et Annexe BPF de l'UE 15 les deux exigent une qualification rigoureuse des équipements. Ignorer IQ/OQ/PQ risque de violations de la réglementation et de rappels potentiels de produits. En pratique, toute nouvelle machine de conditionnement dans une ligne pharmaceutique (ampoules, charges, encartonneuses, etc.) doit être validé via IQ/OQ/PQ s'il touche le produit.
Il s’agit généralement d’un effort conjoint. Le fournisseur de machines apporte souvent son aide en fournissant de la documentation et en exécutant des tests d'acceptation en usine.. L’équipe qualité/validation de l’acheteur effectue (ou des témoins) le site IQ/OQ/PQ. Fréquemment, le fabricant d'équipement d'origine (Ils, Par exemple) propose des services de validation ou des conseils dans le cadre de la mise en service. Finalement, l'entreprise acheteuse est responsable de s'assurer que les IQ/OQ/PQ sont complétés et documentés avant la fabrication du produit.
PQ vient après que IQ et OQ aient été complétés avec succès. Une fois la machine installée (QI) et les tests fonctionnels réussissent (QO), Le PQ est effectué lors de cycles de production réels ou simulés. C'est généralement la dernière étape: Par exemple, exécuter trois lots consécutifs ou un essai de production à grande échelle tout en surveillant les performances. Le PQ se produit souvent pendant une période de mise en service initiale, avant que la ligne ne soit officiellement mise en production de routine.
Absolument. Même si l'emballage semble simple, les régulateurs le traitent comme faisant partie du processus de fabrication contrôlé. Chaque machine de conditionnement primaire doit être qualifiée. Par exemple, comme indiqué sur le site de Jinlu, « pour les produits réglementés, effectuer l'installation, qualification opérationnelle et de performance (IR/WH/PQ) selon les besoins ». Une encartonneuse qui manipule votre produit fini doit fonctionner correctement. (par exemple. plie les cartons de manière fiable, insère des étiquettes), quels IQ/OQ/PQ accomplissent. En substance, tout équipement pouvant affecter la qualité ou la traçabilité du produit nécessite une qualification.
Références:
1.Document Officiel de Qualification et de Validation – Annexe BPF de l’UE 15.
2.GUIDE DES BONNES PRATIQUES DE FABRICATION DES MÉDICAMENTS ANNEXES – Annexe du Guide PIC/S BPF 15 (Norme mondiale BPF).
3.L'histoire & L'avenir de la validation – ispe.org.
4.Qualification (QD, QI, QO, PQ) – gempex.com.

Petit Fu, Fondateur de Jinlupacking, amène 20 années d'expertise dans le secteur des machines pharmaceutiques. Sous sa direction, Jinlu est devenu un fournisseur de confiance intégrant la conception, production, et ventes. Petty est passionné par le partage de ses connaissances approfondies de l'industrie pour aider ses clients à naviguer dans les complexités de l'emballage pharmaceutique., s'assurer qu'ils reçoivent non seulement du matériel, mais un véritable partenariat de services à guichet unique adapté à leurs objectifs de production.