
L'emballage des capsules fait référence à la façon dont les capsules pharmaceutiques (coque dure ou softgel) sont joints pour distribution et utilisation. Il comprend emballage primaire (le récipient immédiat en contact avec les gélules, par exemple. cavités ou bouteilles de blisters) et emballage secondaire (cartons extérieurs, boîtes, et des étiquettes qui protègent et identifient davantage le produit). Un bon emballage des capsules doit protéger le médicament de l’humidité, lumière, et contamination, assurer la stabilité tout au long de la durée de conservation, et répondre aux exigences réglementaires. Par exemple, L'OMS note que « les composants de l'emballage entourent le produit depuis la production jusqu'à son utilisation,» et doit remplir des fonctions comme la protection, stabilité, et identification. Réglementations BPF de la FDA (21 CFR 211) exiger des procédures écrites pour inspecter et tester les matériaux d’emballage dès leur réception, et exiger un étiquetage sécurisé et des contrôles d’inviolabilité. En pratique, nous devons faire correspondre le produit en capsules (forme posologique, sensibilité, dosage, etc.) au format et au matériau d'emballage appropriés.

L'emballage primaire des capsules pharmaceutiques est essentiel pour protection du produit, conformité, et la sécurité des patients: Il garde les capsules au sec, non contaminé, et clairement étiqueté. Par exemple, les plaquettes thermoformées et les flacons ambrés sont connus pour être idéaux pour les médicaments sensibles à l'humidité. Le bon emballage favorise également l’observance du patient (dosage sous blister en dose unitaire, bouteilles avec sécurité enfant, étiquetage clair) et les besoins de la chaîne d'approvisionnement (sérialisation, logistique). Ci-dessous, nous examinons chaque option en détail.
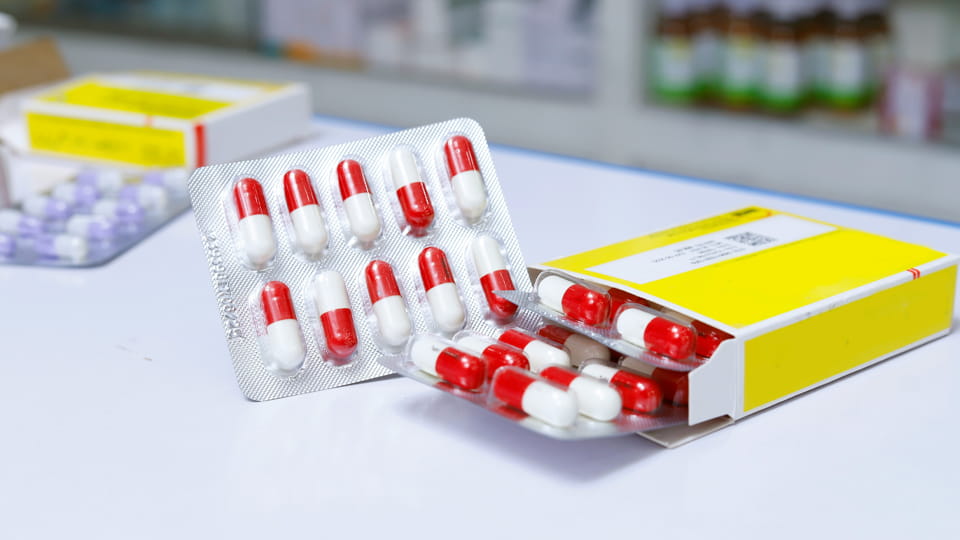
Packs de cloques constitué de cavités en plastique préformées (« ampoules ») qui contiennent des capsules individuelles, scellé avec un support (papier d'aluminium ou plastique). Ce emballage primaire offres protection en dose unitaire. Les blisters fournissent excellente barrière propriétés: ils peuvent être fabriqués avec matériaux comme le PVC/PVDC ou le film (Alu-PVC, De tout le temps) pour bloquer l'humidité, oxygène et lumière. Par exemple, La blistereuse DPP-180Pro de Jinlu peut former et sceller jusqu'à 4,800 blisters de capsules par heure en stratifiés Alu-PVC ou Alu-Alu. Les principales caractéristiques de l'emballage sous blister des capsules comprennent:
Chiffre:Machine d'emballage sous blister Jinlu JL-180Pro formant des ampoules de capsules (Alu-PVC), 4,800 cavités/heure.
Les blisters de Jinlu (par exemple. le DPP-180Pro) peut automatiquement former des cavités, capsules alimentaires, joint, plaques découpées, et même imprimer les codes de lot. Ils fonctionnent généralement à une vitesse moyenne-élevée (des milliers de cavités par heure), idéal pour la production de masse. Différents formats de blisters (Alu-PVC vs Alu-Alu) sont sélectionnés par produit: De tout le temps (feuille-feuille) offre la barrière la plus élevée (idéal pour les médicaments très sensibles à l'humidité), alors que le PVC/PVDC est courant pour les capsules standard. L'emballage blister simplifie également le comptage et l'inspection: la plupart des lignes intègrent facilement des machines d'inspection par caméra ou de comptage de comprimés pour vérifier le remplissage des capsules.
Les flacons sont le récipient multidose classique. Les capsules sont remplies dans des bouteilles (en plastique ou en verre) qui sont ensuite plafonnés. Ce format est bien connu pour les vitamines, suppléments, et de nombreuses pilules sur ordonnance. Principales caractéristiques:
Chiffre: Ligne d'embouteillage de capsules/comprimés Jinlu JL-16H (100 bouteilles/min) avec décrypteur, filler, capsuleuse, et étiqueteuse.
Les lignes d'embouteillage de capsules intègrent plusieurs machines: un bouteille de bouteille (ou trémie d'alimentation), un machine de comptage-remplissage (qui dépose X capsules dans chaque bouteille), un machine de capsulage/scellage (appliquer et serrer les capuchons), et un étiqueteuse. Par exemple, La gamme JL-16H de Jinlu peut gérer des capsules de 3 à 40 mm, atteindre ~100 bouteilles/min avec >99.8% précision de comptage. Les bouteilles peuvent être remplies en vrac très rapidement. Pour respecter la réglementation, les lignes de bouteilles comprennent souvent une station pour l'insertion des dessicants (par exemple. packs de silice) et une station de sceau inviolable. Les bouchons de sécurité enfants sont une autre caractéristique courante des médicaments pédiatriques.
Dans le choix de l'emballage des bouteilles, tenir compte du volume et de la stabilité du produit: produits à grand volume (par exemple. vitamines quotidiennes) bouteilles de costume, alors que les capsules à faible volume ou très sensibles peuvent préférer les ampoules. Notez que les bouteilles offrent généralement moins de protection inhérente contre l’humidité et l’oxygène que les ampoules à haute barrière. Le verre ambré ou les récipients opaques peuvent ajouter une protection contre la lumière, et joints intérieurs (doublures en papier d'aluminium) peut améliorer la barrière.

Sachets (et des packs de bâtons) sont paquets flexibles qui contiennent une dose de poudre, granulés ou liquide. Bien que ce ne soit pas aussi courant pour les capsules, ils sont pertinents pour les formes posologiques apparentées (par exemple. capsules nutraceutiques réduites en poudre, ou du mastic en poudre lui-même). Néanmoins, sachets et stick packs méritent d'être mentionnés dans un guide d'emballage des capsules:
Les sachets et les stick packs sont produits par formage-remplissage-scellement vertical (Vff) ou machines FFS horizontales. Série JL-VP de Jinlu, par exemple, est une machine FFS à bâtons/sachets multivoies (40 sacs / min / voie, Précision de ± 0,02 g) pour poudres, granulés, liquides ou gels. Si les gélules elles-mêmes devaient être conditionnées en sachets, on ouvrirait le sachet pour récupérer la dose – mais le plus souvent, les gélules sont conditionnées intactes dans un blister ou un flacon, tandis que les sachets/stick packs traitent le contenu en poudre.
Note réglementaire: les sachets et les stick packs sont autorisés pour les médicaments solides et semi-solides (selon les définitions de l'OMS). Ils sont considérés comme emballage primaire s’ils contiennent directement le produit. Comme des ampoules, les sachets peuvent être conçus avec une sécurité enfants ou avec des encoches de déchirure.
Paquets de bandes (également appelés bandes d'aluminium ou « bandes de dose ») sont une autre forme de conditionnement primaire en dose unitaire. Une bande est généralement une bande étroite de matériau (souvent en aluminium ou en stratifié multicouche) plié et scellé, avec perforations entre les doses. Contrairement aux ampoules, les paquets de bandes ne forment pas de cavités profondes, mais, la capsule est prise en sandwich entre deux couches de film. Les paquets de bandes sont largement utilisés en Asie et pour les produits où une protection élevée contre l'humidité est nécessaire mais où les blisters sont trop volumineux.. Points clés:

Du point de vue de la sélection, les packs de bandes occupent un juste milieu entre les bouteilles (en gros) et une ampoule (rigidité). Ils sont souvent choisis lorsque très haute barrière contre l'humidité est nécessaire mais dans un cadre mince, forme légère. (Note: les paquets de bandes peuvent ne pas fournir de preuve d'inviolabilité aussi clairement que les blisters, à moins que des scellés supplémentaires ne soient utilisés.)
Après avoir choisi un pack principal, les capsules sont généralement placées dans emballage secondaire comme des boîtes en carton ou emballé sous film rétractable cas. Les cartons offrent une protection supplémentaire pendant l'expédition, permettre un étiquetage supplémentaire (informations sur les médicaments, marque, scellés inviolables), et aider à organiser des produits multi-pack. Considérations clés pour l’emballage secondaire:
En résumé, l'emballage secondaire enveloppe le produit pour la distribution finale et garantit que toutes les caractéristiques d'étiquetage et de conformité sont en place. Un bon design améliore ici l’image de marque et le confort de l’utilisateur (cartons à ouverture facile, étiquetage clair).
Choisir le bon matériau d'emballage est essentiel pour protéger les capsules. Les matériaux doivent être compatibles avec le médicament, répondre aux besoins barrières, et respecter la réglementation (ISO 15378 BPF pour les matériaux d'emballage, etc.). Les matériaux courants comprennent:
En général, choisissez la barrière la plus élevée qui convient à votre capsule. Par exemple, une capsule de gélatine sensible à l'humidité peut justifier un blister Alu-Alu ou une bouteille en verre ambré avec dessicant. En revanche, les capsules non sensibles peuvent utiliser un blister en PVC standard ou une bouteille en PEHD. Tenez également compte de la durée de conservation: les produits à plus long terme nécessitent une barrière plus robuste.
Notes de sécurité/recyclage: Les tendances durables privilégient les emballages mono-matériaux (par exemple. Ampoules PP, bouteilles PET recyclables). Matériaux de test pour les extractibles, compatibilité, et respect des limites FDA/UE (par exemple. USP <661> pour plastifiants) fait partie de la conformité des emballages.
Les capsules sont disponibles en différents types et leur formulation dicte les besoins en matière d'emballage.:
En bref, le matériau de la coque de la capsule et type de remplissage affectera le choix de l'emballage. Les formulations très sensibles à l'humidité privilégient toujours la barrière la plus étanche (Blister Alu-Alu, sachet en aluminium, ou flacon scellé avec déshydratant). Les formulations moins sensibles ont plus d'options. Testez toujours la stabilité du capsule emballée finale (ICH Q1A exige une stabilité dans le conteneur proposé).
La sélection d'un emballage de capsule optimal implique l'évaluation de plusieurs facteurs: forme posologique, conformité des patients, sensibilité du produit, durée de conservation, exigences réglementaires, et le coût. Vous trouverez ci-dessous un cadre décisionnel simplifié:
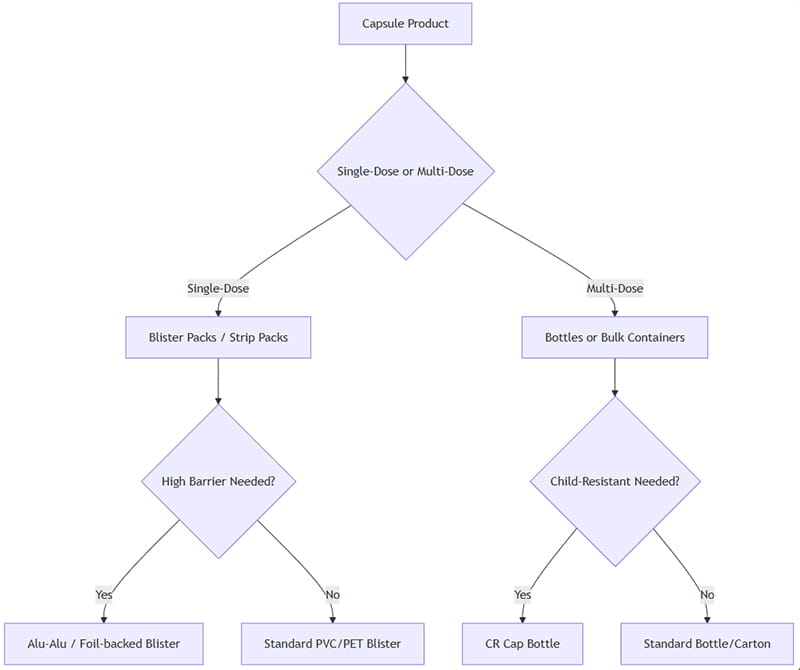
Comment appliquer ceci: Si les capsules sont conçues comme des unités individuelles (par exemple. Dosage serré ou cartes blister de vente au détail), les ampoules/bandes sont préférables. Pour multidose (par exemple. suppléments ou médicaments à forte consommation), les bouteilles sont plus efficaces. Si la protection contre l’humidité et la lumière est essentielle, sélectionner des options à barrière élevée (De tout le temps, bouteille ambrée). Si la sécurité pédiatrique est nécessaire, utiliser des bouchons de bouteille avec sécurité enfants ou des blisters inviolables.
D'autres critères de l'industrie comprennent:
Finalement, essai pilote (études de stabilité, études de consommation) et l'analyse du coût total guidera la décision finale. L'organigramme ci-dessus est un point de départ pour des scénarios typiques.
L'emballage des capsules nécessite des machines spécialisées. Une fois les capsules remplies (par des remplisseurs de capsules), ceux-ci sont transférés vers les lignes de conditionnement:
Conformité des équipements: Toutes les machines d'emballage pour produits pharmaceutiques doivent répondre à la conception GMP (acier inoxydable, surfaces nettoyables, performances validées). Les machines de Jinlu sont certifiées CE/cGMP, souvent avec des protections configurables et des systèmes de rejet pour les pilules non conformes aux spécifications. L'intégration est la clé: Par exemple, une machine à blister peut alimenter directement une encartonneuse qui érige les boîtes et emballe chaque carte blister. Systèmes complets (du remplissage à la boîte finale) permettre l'automatisation de bout en bout du conditionnement des capsules.
L’emballage des capsules pour produits pharmaceutiques doit respecter des réglementations strictes. Les points clés comprennent:
Dans l'ensemble, rencontrer la FDA, Les directives de l’EMA et de l’OMS sont essentielles. De nombreux emballeurs travaillent avec des fournisseurs réglementés (comme Jinlu) qui peut fournir des machines et de la documentation validées (IR/WH/PQ).
Choisir le bon emballage de gélules nécessite une protection équilibrée, convivialité, et le coût. Packs de cloques, bouteilles, sachets, et les bandes ont chacune leur place dans emballage pharmaceutique. Des facteurs tels que la sensibilité à l'humidité, forme posologique, et les besoins du patient guident la décision. Aux côtés, des exigences réglementaires et BPF strictes doivent être respectées.
À Emballage Jinlu, nous sommes spécialisés dans les solutions d'emballage pharmaceutique de bout en bout. Notre équipement – de blistereuses à grande vitesse à comptage entièrement automatisé & lignes d'embouteillage – vous aide à emballer les capsules de manière efficace et conforme. Nous soutenons également les choix durables (par exemple. machines qui utilisent de nouveaux films recyclables) et offrir un service à guichet unique (personnalisation, validation). Notre équipe peut vous aider à évaluer le meilleur système d'emballage de capsules pour votre produit.
Prêt à optimiser votre conditionnement de capsules? Contactez Jinlu Packing aujourd’hui pour des conseils d'experts et des devis. Nos solutions sont soutenues par des normes de qualité mondiales et plus 3,000 clients satisfaits dans le monde entier. Emballons vos capsules pour réussir!
L'emballage des capsules fait référence au processus consistant à enfermer les capsules pharmaceutiques dans des matériaux de protection pour maintenir leur stabilité., sécurité, et conformité. Les formats courants incluent les blisters, bouteilles, et emballage en bandes.
En pratique pharmaceutique, l'emballage n'est pas seulement un confinement : il protège les capsules de l'humidité, oxygène, et contamination, ce qui affecte directement la qualité et la durée de conservation des médicaments.
Il n’existe pas de « meilleure » option : cela dépend de votre produit et de votre marché..
• Emballage blister → idéal pour les médicaments sensibles à l'humidité ou de grande valeur
• Emballage en bouteille → idéal pour le stockage en vrac et les suppléments
• Emballage en bandes → courant sur les marchés sensibles aux coûts
L'emballage sous blister est souvent préféré car il offre une protection en dose unitaire et une meilleure isolation environnementale..
Utilisez des matériaux qui bloquent les principales menaces (humidité, oxygène, lumière). Par exemple, Les blisters en aluminium Alu-Alu ou les flacons en verre ambré offrent une excellente barrière pour les capsules sensibles à l'humidité.. Les blisters en PVC ou PET avec revêtement PVDC sont courants et moins chers pour les produits stables. Les bouteilles en PEHD sont standard mais peuvent nécessiter une pochette déshydratante si les capsules sont sensibles. Évaluer les taux de transmission de la vapeur d'eau (WVTR) de films. Dans une production soucieuse de la durabilité, envisager des monomatériaux recyclables (par exemple. Ampoules PP). Testez toujours la compatibilité des emballages via des études de stabilité.
Les capsules sont sensibles aux facteurs environnementaux tels que l'humidité, oxygène, et la lumière.
Emballage approprié:
• Empêche l'absorption de l'humidité
• Réduit l'oxydation
• Maintient la stabilité chimique
Emballage, en particulier, crée des micro-environnements scellés pour chaque capsule, prolonge considérablement la durée de conservation.
Cela dépend du format. Pour les ampoules, vous avez besoin d'une machine de formage/scellage de blisters (avec des moules à cavités pour s'adapter à votre capsule) plus éventuellement une encartonneuse. Pour les bouteilles, vous avez besoin d'une ligne de remplissage/bouchage de bouteilles (redresseur, contre-remplisseur, capsuleuse, Étiquetage). Pour les packs de sticks, utiliser une machine à sachets verticale pour former, remplir et sceller. Jinlu propose tout cela: par exemple. nos blisters série DPP, Ligne de comptage JL-16H, et emballeuse de sachets JL-VP. Choisir la bonne machine implique de la capacité, taille des capsules, et intégrations requises (systèmes de vision, rejeter les stations, etc.).
Tenez compte de facteurs tels que le dosage (célibataire contre. multiple), besoins de stabilité, et utilisation par les patients. Pour la commodité d’une dose unique et une protection élevée (spécialement pour les capsules sensibles à l'humidité et à la lumière), les ampoules sont souvent préférées. Pour des raisons de volume ou économiques, les bouteilles sont généralement choisies. Aussi, si des fonctionnalités de sécurité enfants ou adaptées aux personnes âgées sont nécessaires, les bouteilles peuvent offrir plus d'options. En règle générale, faites correspondre les propriétés de votre capsule à l’emballage: par exemple. gélules très hygroscopiques → blister Alu-Alu; suppléments très demandés → bouteilles.
Références:
1.Annexe 9 Lignes directrices sur l'emballage des produits pharmaceutiques -- OMS
2.Exigences techniques pour les médicaments et les produits de santé -- Organisation Mondiale de la Santé
3.Matériaux d'emballage pharmaceutique et sécurité des médicaments: Une mini-revue ——MDPI
4.Emballages et notices pharmaceutiques —— Bibliothèque nationale de médecine
5.Une revue sur la caractérisation et la recyclabilité des blisters pharmaceutiques ——ScienceDirect

Petit Fu, Fondateur de Jinlupacking, amène 20 années d'expertise dans le secteur des machines pharmaceutiques. Sous sa direction, Jinlu est devenu un fournisseur de confiance intégrant la conception, production, et ventes. Petty est passionné par le partage de ses connaissances approfondies de l'industrie pour aider ses clients à naviguer dans les complexités de l'emballage pharmaceutique., s'assurer qu'ils reçoivent non seulement du matériel, mais un véritable partenariat de services à guichet unique adapté à leurs objectifs de production.