
Pharmaceutical companies today are under pressure from every direction. Hospitals want safer medication handling. Retail markets want lower packaging costs. Regulators expect better traceability and serialization. En même temps, manufacturers are trying to improve production efficiency without creating unnecessary packaging waste. That is why the discussion around conditionnement en dose unitaire et bulk packaging has become much more important in recent years. One format focuses on dose-level protection and patient safety, while the other prioritizes speed, simplicité, and cost efficiency. Neither option is automatically better for every product. The right choice depends on your drug type, production goals, distribution channel, and packaging line capabilities. Pour les fabricants pharmaceutiques, procurement teams, and operations managers, understanding the real differences between unit dose and bulk packaging is essential before investing in a new pharmaceutical packaging system or upgrading an existing ligne de conditionnement.

Unit dose packaging (aussi appelé single-dose packaging) means each dose of medication is individually sealed in its own package. Par exemple, one pill might be in its own blister pocket or pouch. Jinlu Packing explains that “unit dose packaging means each individual dose of a medication is pre-packaged and sealed by itself”. En pratique, this can be a single-tablet blister card, un paquet de bandes (one tablet per cavity), a prefilled syringe, a vial, or a sachet containing one dose of liquid or powder. Each package carries labels (nom du médicament, dose, expiration) so a nurse or patient can confirm the medicine without counting pills from a bottle. In US hospitals, sur 75% of oral meds are dispensed in unit-dose form.
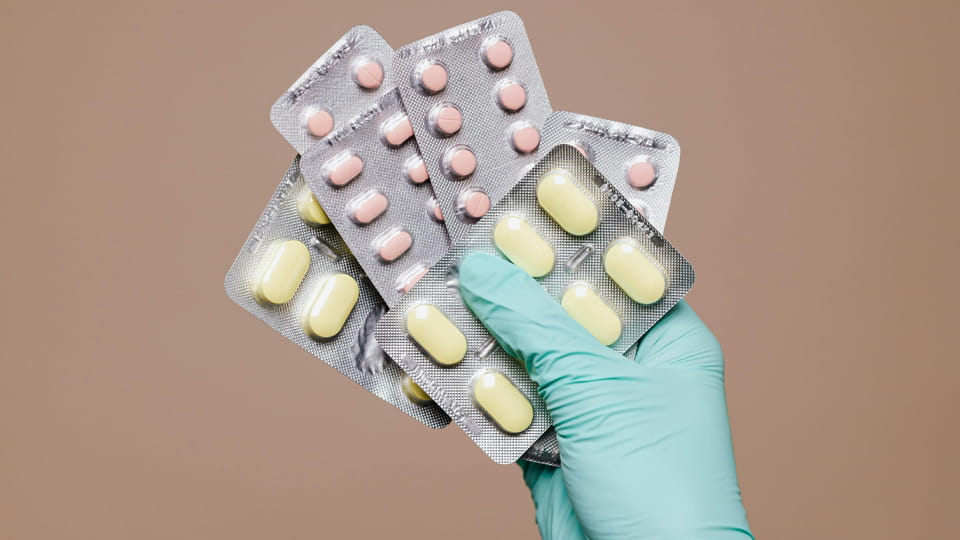
Unit-dose packs are common in hôpitaux, cliniques, long-term care facilities, and clinical trials – essentially anywhere precise dose control and traceability are paramount. They help prevent dosing errors because each packet contains exactly one dose and is clearly labeled. Autrement dit, as Jinlu notes, unit-dose packs require no counting – you “just grab one package and open it”, reducing handling mistakes. Other terms used include single-dose, unit-of-use, ou unit-of-medication conditionnement. Typical formats include:
En résumé, conditionnement en dose unitaire isolates every dose individually, aiding safety, conformité des patients, et traçabilité.
En revanche, bulk packaging refers to multi-dose or collective packaging of drugs. Common bulk formats include large pill bottles, pots, multi-dose tubes, and bulk bags or totes. Par exemple, a 100-tablet prescription bottle or a vitamin jar with 30 capsules is bulk packaging. These containers hold many doses together. Bulk packaging is the norm for consumer pharmacy products, médicaments en vente libre, suppléments nutritionnels, and most generic tablets. It’s also used for intermediate stages of production (par exemple. bulk sachets or drums).

Bulk packaging is usually more cost-effective per dose and is suited to high-volume, commodity products. The equipment for bulk lines includes tablet/capsule counting and bottle filling machines, plafonnement, scellage, and labeling equipment. Par exemple, a tablet-counting line may use an automatic counting machine to fill bottles, a capper to seal them, and a labeler to finish. Bulk lines often run faster (counting hundreds of tablets per minute) but offer less dose-by-dose control than unit-dose systems. Bulk packaging is ideal for mass-market OTC drugs, suppléments nutritionnels, generic medications, and any product where price per dose is critical and individual dosing control is less important.
The table below compares the two approaches on important factors:
| Facteur | Emballage en dose unitaire | Emballage en vrac |
| Medication Safety | Haut. Each dose sealed, reducing contamination. | Inférieur. Multiple doses in one container. |
| Conformité des patients | Mieux. Individual doses clearly labeled; easier to verify. | Inférieur. Patients count their own doses. |
| Traçabilité & Sérialisation | Excellent. Easy to serialize/track each dose. | Limité. Only container is tracked, not each pill. |
| Packaging Cost | Higher per dose. More packaging material & manutention. | Lower per dose. One package holds many units. |
| Production Throughput | Modéré. More steps per dose (par exemple. formation de cloques). | Plus haut. Simple counting/filling is faster. |
| Packaging Waste | Plus. One-dose packs increase material use. | Moins. Minimal packaging per dose (par exemple. one bottle vs. 100 clochards). |
| Shelf-life/Protection | Mieux. Blisters/vials often have high-barrier films. | Modéré. Bulk bottles protect less per unit, though shelf life can be similar. |
| Mieux pour | Hospitals, essais cliniques, high-value drugs, controlled substances, personalized meds. | Retail OTC, generic drugs, vitamines, suppléments, large-volume consumer products. |
As Jinlu Packing notes, think of it like a bottle of 100 comprimés (en gros) versus a blister with one tablet (unit dose). Bulk bottles clearly win on cost efficiency and speed, but unit-dose formats excel on safety and control. Par exemple, blister packaging individually protects each pill from moisture and tampering, whereas bulk bottles expose all tablets to the same environment.

Unit-dose systems offer several key benefits:
En résumé, conditionnement en dose unitaire maximizes safety, traçabilité, and patient-centric dosing, at the expense of higher packaging and handling cost. As Jinlu summarizes, unit-dose units are “designed to hold a quantity of drug intended for administration as a single dose”, ensuring “the right medication in the correct strength reaches the right patient at the right time, every time”.
Bulk (multi-dose) packaging has its own strengths:
Bulk packaging shines in cost-efficiency and scale, making it ideal for high-volume products where the economy of scale outweighs the need for individual-dose control. En fait, Jinlu’s experience as a supplier shows that bulk bottle lines (including counting, plafonnement, and labeling units) form the backbone of many nutraceutical and generic drug production lines.
Different end-markets and use-cases have clear preferences. The table below summarizes typical industry choices:
| Industry/Use Case | Preferred Packaging |
| Hospitals & Soins de santé (acute care) | Unit Dose (clochards, bandes, seringues) |
| Long-Term Care & LTC Pharmacy | Unit Dose (cartes blister, pre-filled packs) |
| Clinical Trials & Recherche | Unit Dose (precise dosage control for trials) |
| High-Potency / Controlled Substances | Unit Dose (security, dosage) |
| Contract Manufacturing (CMOs producing hospital meds) | Unit Dose |
| Over-the-Counter (De gré à gré) Drogue | Bulk (bottles/tubes of tablets, gélules) |
| Nutraceutiques & Suppléments | Bulk (vitamin bottles, sachets) |
| Generic Pharmaceuticals (mass market) | Bulk (cost focus) |
| Emerging Consumer Health (produits de beauté, nutraceutical combos) | Bulk |
| Veterinary / Animal Health | Bulk (often retail multi-dose vials or feed additives) |
| Emergency Kits / First Aid | Unit Dose (single-use pouches for portability) |
Par exemple, Jinlu notes that unit-dose systems originated in hospitals and now “vastly expanded” into other fields due to safety advantages. En effet, sur 75% of U.S. hospital oral medications are unit-dosed, reflecting the sector’s preference. En revanche, consumer-facing markets like supplements and OTC pain relievers typically use bulk containers because they demand lower unit cost and higher volume. Clinical and hospital environments favor the dosing control of blisters or sachets, while generic-drug manufacturers and retailers focus on the efficiency of bottle lines.
Choosing between unit-dose and bulk has significant cost implications. Consider the following cost factors:
En résumé, unit-dose packaging typically demands higher CAPEX and per-unit packaging cost, but can yield savings from fewer errors and waste. Bulk packaging offers lower material and production costs per dose. The “better” option depends on analyzing these costs alongside the product’s pricing strategy and compliance requirements.
Unit-dose packaging lines use specialized equipment. Key machines include:

En bref, un unit-dose line might consist of a blister or sachet machine (for primary packaging), followed by a cartoner (and optionally labeler or printer). Par exemple, a typical hospital drug line is: Tablet orientation feeder → Blister machine → Automatic cartoner → Case packer. Jinlu’s machinery for these steps can be linked in one flow (see that “can be connected to blister machines»).
[jl_youtube src= »https://www.youtube.com/embed/k0uyYbGttCU »]
Bulk lines use equipment optimized for speed and volume:
En vigueur, un bulk packaging line typically looks like: Bottle unscrambler → Tablet counter filling machine → Desiccant inserter → Capper → (Foil sealer) → Labeler → Cartoner. These modular machines can also handle diverse products (gummies, poudres, liquides) as long as counting/filling and capping are adjusted. Jinlu’s and pages illustrate how such lines are integrated.
Par exemple, the JL-16R gummy line “automatically complete the entire process of unscramble bottles, counting … inserting desiccant, plafonnement, aluminum foil sealing and labeling” at jusqu'à 80 bouteilles/min. These advanced lines emphasize throughput and consistency, making them ideal for bulk product packaging.

Choosing the right approach depends on multiple factors. Use the checklist and flowchart below as a guide:
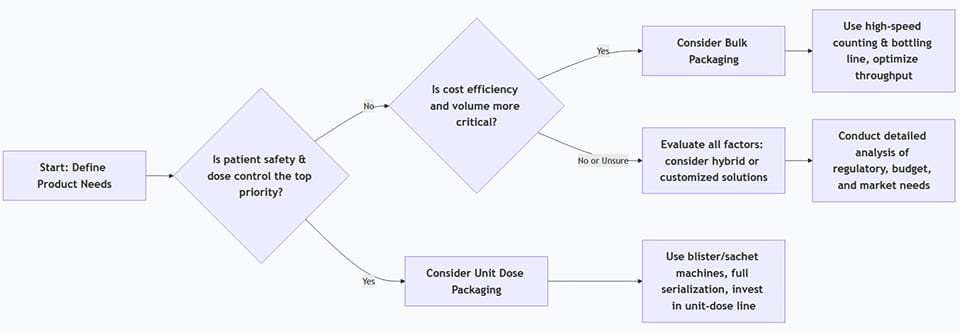
This chart shows a simplified guide: if medical safety/tracability outweighs cost, lean towards unit-dose. If budget and volume dominate, bulk is likely best. In real cases, mixed strategies (par exemple. bulk bottle inside a serialized carton) can also be considered.
Il n’y a pas de réponse universelle. The choice between unit dose and bulk packaging depends on your product’s requirements and your business strategy. Unit-dose packaging offers unmatched safety, conformité, and convenience for hospitals, essais cliniques, and high-value drugs, but it requires more investment in machinery (blister or sachet packers, encartonneuses) et matériel d'emballage. Bulk packaging (bouteilles, pots, tubes) excels at low cost and high throughput, ideal for retail drugs and supplements, but gives less control over individual dosing.
En pratique, many companies use les deux methods for different products. Par exemple, a drug with a narrow therapeutic index might be packed in single-dose blisters for hospital sale, while the same medication’s OTC version is sold in 30-count bottles. When designing your line, Considérez le coût total de possession (équipement + matériels + errors) and regulatory needs.
To determine the best approach, review the factors above: target market, forme posologique, volume, compliance rules, et budget. A packaging machinery expert (ou Jinlu’s customization team) can design an appropriate line. Par exemple, Jinlu Packing fournit turnkey solutions – from high-speed blister machines to integrated tablet-counting and bottling lines – to fit both unit-dose and bulk needs.
Unit dose packaging means each medication dose is individually sealed and labeled, such as one tablet per blister cavity or one sachet per use. Bulk packaging stores multiple doses together in a bottle, récipient, or larger package. Unit dose packaging focuses more on safety, traçabilité, and dose control, while bulk packaging is usually more cost-efficient for high-volume production.
There is no single answer for every pharmaceutical product. Unit dose packaging is often better for hospitals, clinical environments, and high-value medications because it improves medication safety and tracking. Bulk packaging is usually preferred for nutraceuticals, OTC products, and large-scale retail distribution because it reduces packaging cost and improves production speed.
Hospitals commonly use unit dose packaging because each dose is clearly identified and protected until administration. This helps reduce medication errors, supports barcode scanning systems, and improves patient safety during dispensing.
Oui, in most cases bulk packaging has lower packaging material costs and higher line efficiency. Bottle filling lines and tablet counting systems can usually run at faster speeds than individual dose packaging systems. Cependant, total cost should also consider medication waste, travail, compliance requirements, et la protection des produits.
Bulk packaging is widely used for:
• Vitamins and supplements
• Generic tablets and capsules
• OTC medications
• Nutraceutical products
• High-volume retail pharmaceuticals
These products are often packed using counting machines, bottle filling lines, machines à capsuler, and labeling systems.
Common unit dose packaging equipment includes:
• Machines d'emballage sous blister
• Strip packing machines
• Machines d'emballage de sachets
• Stick pack machines
• Encartonneuses
• Systèmes d'inspection visuelle
These pharmaceutical packaging machines help create individually sealed doses with better product protection and traceability.
Oui. Unit dose packaging helps lower the risk of contamination, incorrect dosing, and handling mistakes because each dose remains sealed until use. Many healthcare facilities use unit dose systems to support safer dispensing workflows and barcode verification processes.
Oui. Pharmaceutical blister packaging is one of the most common forms of unit dose packaging. Each tablet or capsule is stored in a separate cavity, which helps protect the product from moisture, oxygène, and contamination while also improving dose tracking.
Bulk packaging is commonly used in:
• Nutraceutical manufacturing
• Supplement production
• Retail pharmaceutical distribution
• Médecine vétérinaire
• High-output tablet and capsule production
Manufacturers choose bulk pharmaceutical packaging when production efficiency and lower packaging costs are the main priorities.
The best choice depends on several factors, y compris:
• Product type
• Target market
• Exigences réglementaires
• Packaging budget
• Volume de production
• Shelf-life needs
• Distribution channels
Par exemple, hospitals and clinical products often benefit from unit dose packaging, while retail supplements and high-volume generic drugs are usually more suitable for bulk packaging systems.
Références:
1. Sec. CPG 430.100 Unit Dose Labeling for Solid and Liquid Oral Dosage Forms -- NOUS. Administration des aliments et des médicaments
2.Data Standards Manual (monographs) Package Type -- NOUS. Administration des aliments et des médicaments
3.Emballage de médicaments —— WikiPedia
4.Guidance on good manufacturing practice and good distribution practice: Questions and answers —— Agence européenne des médicaments
5.ISO 15378 attestation – Primary Packaging Material for Medicinal standard

Petit Fu, Fondateur de Jinlupacking, amène 30 années d'expertise dans le secteur des machines pharmaceutiques. Sous sa direction, Jinlu est devenu un fournisseur de confiance intégrant la conception, production, et ventes. Petty est passionné par le partage de ses connaissances approfondies de l'industrie pour aider ses clients à naviguer dans les complexités de l'emballage pharmaceutique., s'assurer qu'ils reçoivent non seulement du matériel, mais un véritable partenariat de services à guichet unique adapté à leurs objectifs de production.