
Struggling with leaking blister packs? Let’s learn how this blister defect occurs and how to solve this problem!
No matter you’re handling Embalaje de ampolla líquida, or dealing with solid forms like embalaje de ampolla de cápsulas y pill blister packaging, leakage is always a headache for pharma manufacturers. In the realm of blister packaging in pharmaceutical industry, data indicates that even a seemingly negligible defect rate of just 3%, caused by leaks or blister sealing failure, results in over 15,000 unqualified products per month. En otras palabras, the resulting hidden loss of profit could amount to between $50,000 y $100,000 anualmente.
What an astonishing number! So understanding the leakage problem, exploring the root causes of blister defects, and finding common packaging machine issues and solutions become worthwhile and necessary for each pharma and supplement manufacturer.
Conclusiones clave
Bart, a health-conscious senior analyst, relies on premium supplements to sustain his high-performance lifestyle. One morning, he discovered a discolored, ineffective pill caused by a blister seal gap that allowed moisture to ruin the active ingredients. Furious at this breach of trust, Bart discarded the blister box and permanently blacklisted the brand. For the manufacturer, this tiny leak may result in far more than a film waste but losses of customers’ trust.
En su esencia, blister packaging leaks is quite simple to understand: the blister seal bonding the blister plastic to the sealing foil has failed. The moment a breach occurs in this seal, outside air, humedad, and bacteria can easily slip inside, rendering the protective barrier completely ineffective.
The ensuing consequences are extremely severe, with their specific manifestations depending on the nature of the contents within the packaging. Taking solid oral dosage forms like pill blister packaging or capsule blister packaging as an example, a leak can lead to elevated humidity levels within the cavity; The humidity, Sucesivamente, degrades and inactivate the Active Pharmaceutical Ingredient (API).
For liquid blister packaging, the issue is even more critical: a leak signifies the actual loss of the liquid contents, potentially resulting in the complete failure of the entire blister tray, and even the total rejection of the entire product batch.
Absolutely not. While occasional mechanical malfunctions cannot be entirely ruled out, treating blister defect as a «normal» occurrence equals to ignoring a boat that is taking on water, and the consequences could be catastrophic.
This is particularly true for liquid blister packaging; even the slightest inconsistency in pressure or temperature can result in leakage. If the liquid is food such as ketchup, the cost is manageable; sin embargo, if the liquid is expensive, the resulting financial losses could be incalculable.
Dr. Sarah Jenkins, Chief Materials Scientist at the Global Packaging Institute, notes: «Manufacturers often mistake material incompatibility for mechanical failure; yet, en 80% of liquid leakage cases, the root cause of blister sealing failure lies in minute variations in sealing temperature or pressure.» This insight reminds us of blister packaging complexity that blister defect is a result of a complex of factors. En las siguientes secciones, we will delve in greater detail into the underlying causes of leakage in blister packaging.

From understanding blister defects to diagnosing their root causes of blister defects, this is an essential skill for any pharmaceutical or health supplement manufacturer. To better facilitate the development of solutions, we will first focus on the underlying causes, undertaking a systematic classification and analysis.
Table 1-Classification and Description of Reasons for Blister Packs Leakage
| Categoría | Specific Reason | Brief Description |
| Machine Calibration | Uneven Sealing Pressure | The Blister Packaging Machine fails to distribute force equally across the blister tray packaging. |
| Thermal Variation | Incorrect Sealing Temperature | Heat is either too low to melt the adhesive or too high, scorching the blister plastic. |
| Material Issues | Incompatible Films | The forming film and lidding foil (P.EJ., blister paper packaging vs aluminum blister packaging) don’t bond well. |
| Mechanical Wear | Worn Sealing Molds | Molds wear creates gaps in the blister seal. |

The image above depicts a typical blister pack produced under uneven pressure. As is evident, the textured pattern on one side is deep, while on the other, it is shallow. Sin embargo, this is not merely an aesthetic issue; it reflects an underlying problem: leakage in the blister pack.
Uneven sealing pressure primarily stems from two factors: single-cylinder systems and worn molds. por un lado, many modern blister packaging sealing machine adopt a single pneumatic cylinder at the heat-sealing station. Since this cylinder serves as the primary source of heat-sealing pressure, this single-cylinder design frequently proves inadequate when processing blister packs made of rigid PET or those with a large surface area. This uneven distribution inevitably leads to wrinkled blister packaging and leaks.

Por otro lado, the impact of mold damage, resulting from prolonged use, also cannot be overlooked. When the heat-sealing mold lacks sufficient surface parallelism, it inevitably leads to uneven pressure distribution across the blister pack during the sealing process.
When it comes to temperature control, producing a flawless blister box requires the same meticulous attention to «heat management» as cooking a gourmet meal. Inconsistent temperature is the silent killer of the Embalaje blister Alu-Alu proceso. If the temperature is too low, the heat-seal coating on the pharma packaging foil will fail to activate for cohesion. En cambio, if the temperature is too high, the blister plastic may melt completely. Dr. Roberto Vance, Chief Engineer at the Packaging Science Foundation’s Thermodynamics Division, explica: «Inconsistent temperature is the silent killer of blister seals. A fluctuation of just 5℃ can compromise seal integrity by as much as 40%.»
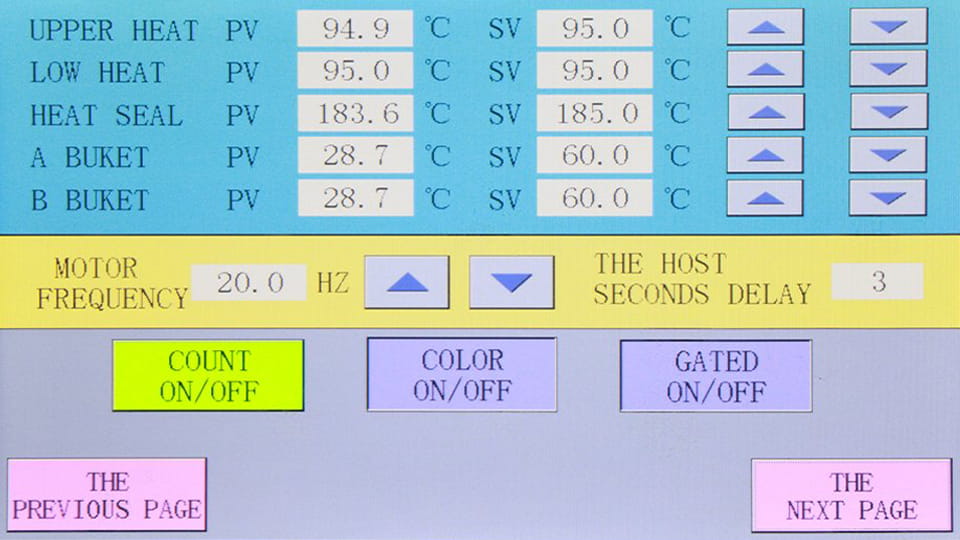
Improper heating element layout and a delayed response in temperature control systems are the primary culprits behind these temperature fluctuations. If the electric heating rods embedded within the heating plate are sparsely distributed or unevenly arranged, «hot spots» and «cold zones» will inevitably emerge across the plate’s surface. Además, if the sensors within the temperature control system are positioned too far from the sealing interface, or if the feedback loop operates too slowly, it can lead to significant temperature instability.
When switching blister packing supplies, some manufacturers may inadvertently introduce materials that do not bond effectively under their current blister packaging machine setting. Por ejemplo, mixing paper blister packaging adhesives with incompatible films. This incompatibility typically manifests as delamination of the blister packaging which indicates a failure to adhere properly. Además, certain emerging blister materials require specialized adhesives, which is hard for older machinery to process.
Now that we have identified the culprits behind these blister defects, it is time to take action to optimize your blister line packaging. Learning how to deal with these problems is the final step to transform your blister packs into the perfect ones.


Fixing blister defects requires more than just good intentions; it requires world-class equipment. Y Jeinlu eligiendo, we have partnered with Fortune 500 pharmaceutical giants like Robinson, UCB, and US Pharma to address the blister packaging issues. Compliant with CE and cGMP certifications, Jinlu Packing ensures unparalleled quality and compliance for your embalaje de ampolla médica necesidades.

Nuestro máquina de envases de ampollas, que van desde laboratory blister machine a rotary blister packaging machine, is engineered to tackle the most stubborn common packaging machine issues with:
Dual-Cylinder Design:Delivers massive, perfectly uniform pressure.
Alu-PVC and Alu-Alu Dual Use: Versatility that saves costs.
Drawer-Type Mold Slot Design: Quick mold changeover
Balcony-Style Design: Convenient cleaning with no dead corners
Beyond our cutting-edge máquina de envases de ampollas tecnología, our commitment to support your blister line packaging extends long after the sale. Ofrecemos:
Blister defects may occur in everyday production. It can be either significant or minor, depending on your knowledge about how to handle the blister packaging complexity. But one thing for sure, an advanced máquina de envases de ampollas is a must if you want your production line runs smoothly without worrying about the potential shutdown. Contáctenos for further details!
En términos generales, most medications can be packaged in blister packaging, with the exception of a very few specific medication such as those that are highly reactive or extremely sensitive to light, which sometimes require the use of glass bottles made from specialized materials.
Blister sealing is the critical process that determines the integrity of a blister pack. Whether it’s a cold seal blister pack or vacuum forming blister packaging, a blister packaging machine uses heat and pressure to bond the materials together.
The most frequent issues include a blister defect like leakage, wrinkled foil, and uneven embossing. These issues often relates to uneven pressure of the heat sealing station.
No necesariamente! Unreasonable increase in temperature results in material melting on your blister packaging machine. It’s finding the perfect balance that matter in solving the leaking issue.
Surprisingly, no! A high speed blister packing machine with a dual-cylinder design applies pressure more efficiently, reducing the need for repeated sealing cycles and ultimately saving energy while preventing blister defects.
Referencias
1.Investigación de gran vista: Tamaño del mercado de envases farmacéuticos, Compartir & Informe de análisis de tendencias
2.International Journal of Pharmaceutics: Impact of Sealing Mold Wear on the Integrity of Medical Blister Packaging
3.Global Packaging Institute: Thermal Dynamics in Liquid Blister Packaging Integrity

Petty Fu, Fundador de Jinlupacking, trae 30 años de experiencia al sector de maquinaria farmacéutica. Bajo su liderazgo, Jinlu se ha convertido en un proveedor confiable que integra diseño, producción, y ventas. A Petty le apasiona compartir su profundo conocimiento de la industria para ayudar a los clientes a navegar las complejidades del empaque farmacéutico., garantizar que reciban no sólo equipos, sino una verdadera asociación de servicio integral adaptada a sus objetivos de producción..