
Η συσκευασία κάψουλας αναφέρεται στον τρόπο με τον οποίο οι φαρμακευτικές κάψουλες (σκληρού κελύφους ή μαλακής γέλης) εσωκλείονται για διανομή και χρήση. Περιλαμβάνει πρωταρχική συσκευασία (το άμεσο δοχείο σε επαφή με τις κάψουλες, π.χ.. κοιλότητες φυσαλίδων ή μπουκάλια) και δευτερεύουσα συσκευασία (εξωτερικά χαρτοκιβώτια, κουτιά, και ετικέτες που προστατεύουν και προσδιορίζουν περαιτέρω το προϊόν). Η καλή συσκευασία της κάψουλας πρέπει να προστατεύει το φάρμακο από την υγρασία, φως, και μόλυνση, εξασφαλίζουν σταθερότητα κατά τη διάρκεια ζωής, και πληρούν τις κανονιστικές απαιτήσεις. Για παράδειγμα, Ο ΠΟΥ το σημειώνει «Τα εξαρτήματα συσκευασίας περιβάλλουν το προϊόν από την παραγωγή μέχρι τη χρήση,” και πρέπει να ικανοποιεί λειτουργίες όπως η προστασία, σταθερότητα, και ταυτοποίηση. Κανονισμοί GMP του FDA (21 CFR 211) απαιτούν γραπτές διαδικασίες για την επιθεώρηση και τη δοκιμή των υλικών συσκευασίας κατά την παραλαβή, και επιβάλλουν ασφαλείς ελέγχους επισήμανσης και παραβίασης. Στην πράξη, πρέπει να ταιριάξουμε το προϊόν κάψουλας (δοσολογική μορφή, ευαισθησία, δοσολογία, και τα λοιπά.) στην κατάλληλη μορφή και υλικό συσκευασίας.

Η κύρια συσκευασία των φαρμακευτικών καψουλών – είναι κρίσιμη για προστασία του προϊόντος, συμμόρφωση, και την ασφάλεια των ασθενών: Διατηρεί τις κάψουλες στεγνές, μη μολυσμένο, και με σαφή σήμανση. Για παράδειγμα, Οι συσκευασίες blister και τα κεχριμπαρένια μπουκάλια είναι γνωστό ότι είναι ιδανικά για φάρμακα ευαίσθητα στην υγρασία. Η σωστή συσκευασία υποστηρίζει επίσης τη συμμόρφωση του ασθενούς (δοσολογία κυψέλης μονάδας δόσης, μπουκάλια ανθεκτικά στα παιδιά, σαφής επισήμανση) και τις ανάγκες της εφοδιαστικής αλυσίδας (σειριοποίηση, επιμελητεία). Παρακάτω εξετάζουμε κάθε επιλογή λεπτομερώς.
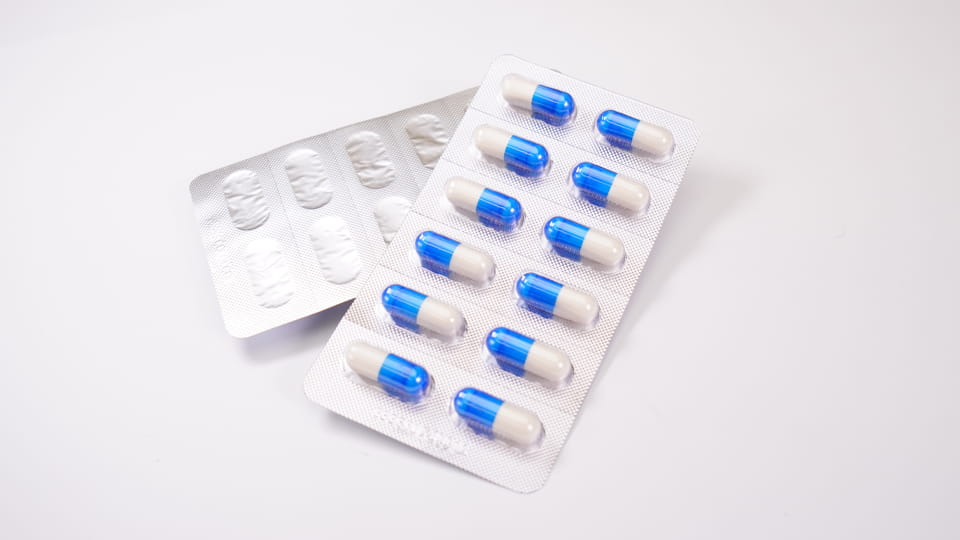
Φουσκάλες αποτελούνται από προσχηματισμένες πλαστικές κοιλότητες ("φουσκάλες") που χωρούν μεμονωμένες κάψουλες, σφραγισμένο με ένα υπόστρωμα (αλουμινόχαρτο ή πλαστικό). Αυτό πρωταρχική συσκευασία προσφορές προστασία μονάδας δόσης. Οι φουσκάλες παρέχουν εξαιρετικό εμπόδιο σκηνικά θέατρου: μπορούν να γίνουν με υλικά όπως PVC/PVDC ή αλουμινόχαρτο (Alu-PVC, Alu-Alu) για να μπλοκάρει την υγρασία, οξυγόνο και φως. Για παράδειγμα, Η μηχανή κυψελών DPP-180Pro της Jinlu μπορεί να σχηματίσει και να σφραγίσει μέχρι 4,800 κυψέλες κάψουλας ανά ώρα σε ελάσματα Alu-PVC ή Alu-Alu. Τα βασικά χαρακτηριστικά της συσκευασίας κυψέλης κάψουλας περιλαμβάνουν:
Εικόνα:Jinlu JL-180Pro μηχανή συσκευασίας κυψελών που σχηματίζει κυψέλες κάψουλας (Alu-PVC), 4,800 κοιλότητες/ώρα.
Μηχανές blister της Jinlu (π.χ.. το DPP-180Pro) μπορεί να σχηματίσει αυτόματα κοιλότητες, κάψουλες τροφοδοσίας, σφραγίδα, κομμένα πιάτα, και ακόμη και να εκτυπώσετε κωδικούς παρτίδας. Συνήθως τρέχουν με μέτρια προς υψηλή ταχύτητα (χιλιάδες κοιλότητες ανά ώρα), Ιδανικό για μαζική παραγωγή. Διαφορετικές μορφές blister (Alu-PVC εναντίον Alu-Alu) επιλέγονται ανά προϊόν: Alu-Alu (αλουμινόχαρτο) προσφέρει το υψηλότερο εμπόδιο (καλύτερο για πολύ ευαίσθητα στην υγρασία φάρμακα), ενώ το PVC/PVDC είναι κοινό για τυπικές κάψουλες. Η συσκευασία blister απλοποιεί επίσης την καταμέτρηση και την επιθεώρηση: Οι περισσότερες γραμμές ενσωματώνουν εύκολα μηχανές επιθεώρησης κάμερας ή μέτρησης tablet για να επαληθεύσουν το γέμισμα της κάψουλας.
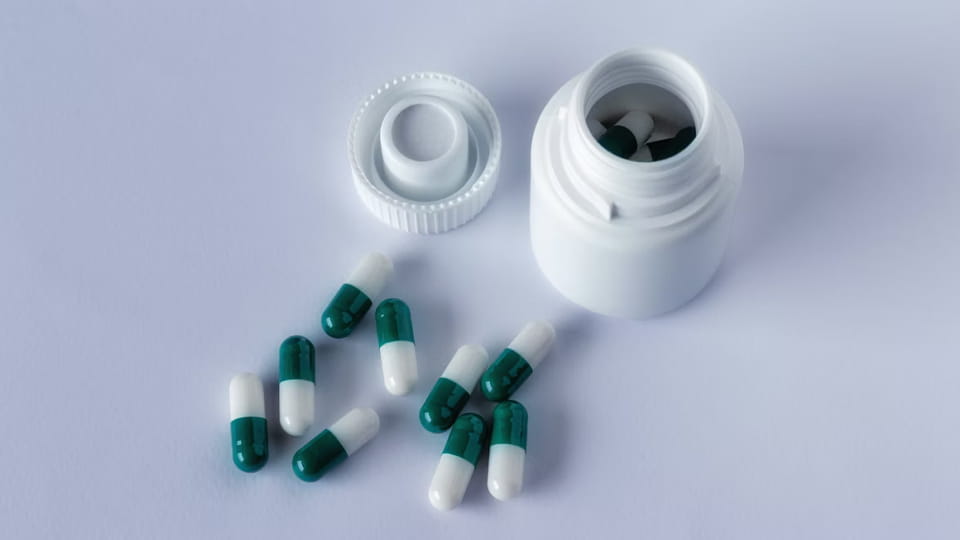
Τα μπουκάλια είναι το κλασικό δοχείο πολλαπλών δόσεων. Οι κάψουλες γεμίζονται σε φιάλες (πλαστικό ή γυαλί) τα οποία στη συνέχεια καλύπτονται. Αυτή η μορφή είναι γνωστή για τις βιταμίνες, συμπληρώματα, και πολλά συνταγογραφούμενα χάπια. Βασικά χαρακτηριστικά:
Εικόνα: Γραμμή εμφιάλωσης καψουλών/δισκίων Jinlu JL-16H (100 μπουκάλια/λεπτό) με unscrambler, ράβδος, καπάκι, και ετικετογράφος.
Οι γραμμές εμφιάλωσης καψουλών ενσωματώνουν πολλαπλές μηχανές: ένα μπουκαλάκι unscrambler (ή χοάνη τροφοδοσίας), ένα μηχανή μέτρησης-γεμίσματος (που ρίχνει Χ κάψουλες σε κάθε φιάλη), ένα μηχανή κάλυψης/σφράγισης (εφαρμόστε και σφίξτε τα καπάκια), και α μηχάνημα επισήμανσης. Για παράδειγμα, Η σειρά JL-16H της Jinlu μπορεί να χειριστεί κάψουλες 3–40 mm, επιτυγχάνοντας ~100 μπουκάλια/λεπτό με >99.8% ακρίβεια μέτρησης. Τα μπουκάλια μπορούν να γεμιστούν χύμα πολύ γρήγορα. Για την τήρηση των κανονισμών, Οι γραμμές φιαλών συχνά περιλαμβάνουν σταθμό για την εισαγωγή αποξηραντικών (π.χ.. συσκευασίες πυριτίου) και έναν σταθμό σφραγίδας που δεν έχει παραβιάσεις. Τα καπάκια ανθεκτικά στα παιδιά είναι ένα άλλο κοινό χαρακτηριστικό για τα παιδιατρικά φάρμακα.
Στην επιλογή συσκευασίας μπουκαλιών, λάβετε υπόψη τον όγκο και τη σταθερότητα του προϊόντος: προϊόντα μεγάλου όγκου (π.χ.. καθημερινές βιταμίνες) μπουκάλια κοστουμιού, ενώ οι κάψουλες χαμηλού όγκου ή υψηλής ευαισθησίας μπορεί να προτιμούν τις φουσκάλες. Τα μπουκάλια σημείωσης προσφέρουν γενικά λιγότερη εγγενής προστασία υγρασίας/οξυγόνου από τις κυψέλες υψηλού φραγμού. Τα πορτοκαλί γυάλινα ή αδιαφανή δοχεία μπορούν να προσθέσουν προστασία από το φως, και εσωτερικές σφραγίδες (επενδύσεις από φύλλο αλουμινίου) μπορεί να βελτιώσει το εμπόδιο.

φακελάκια (και κολλάνε πακέτα) εκτάριο ευέλικτα πακέτα που κρατούν μία δόση σκόνης, κόκκους ή υγρό. Αν και δεν είναι τόσο συνηθισμένο για κάψουλες, είναι σχετικές με τις σχετικές δοσολογικές μορφές (π.χ.. διατροφικές κάψουλες σπασμένες σε σκόνη, ή το ίδιο το πληρωτικό σε σκόνη). Παρόλα αυτά, φακελάκια και πακέτα ραβδιών αξίζει να αναφέρεται σε έναν οδηγό συσκευασίας κάψουλας:
Τόσο τα φακελάκια όσο και οι συσκευασίες stick παράγονται από κατακόρυφη μορφή-γεμίσματος-σφραγίδα (VFFS) ή οριζόντιες μηχανές FFS. Η σειρά JL-VP του Jinlu, για παράδειγμα, είναι ένα μηχάνημα FFS stick/σακετάκι πολλαπλών λωρίδων (40 τσάντες/λεπτό/λωρίδα, Ακρίβεια ±0,02 g) για σκόνες, κόκκους, υγρά ή τζελ. Εάν οι ίδιες οι κάψουλες πρόκειται να συσκευαστούν σε φακελάκια, κάποιος θα άνοιγε το φακελάκι για να ανακτήσει τη δόση – αλλά πιο συχνά, Οι κάψουλες συσκευάζονται άθικτες σε blister ή φιάλη, ενώ τα φακελάκια/πακετάκια με ραβδί χειρίζονται περιεχόμενο σε σκόνη.
Κανονιστικό σημείωμα: Τα φακελάκια και οι συσκευασίες stick επιτρέπονται για στερεά και ημιστερεά φάρμακα (σύμφωνα με τους ορισμούς του ΠΟΥ). Θεωρούνται πρωταρχική συσκευασία εάν περιέχουν απευθείας το προϊόν. Σαν φουσκάλες, Τα φακελάκια μπορούν να σχεδιαστούν ανθεκτικά στα παιδιά ή με εγκοπές σχίσιμου.
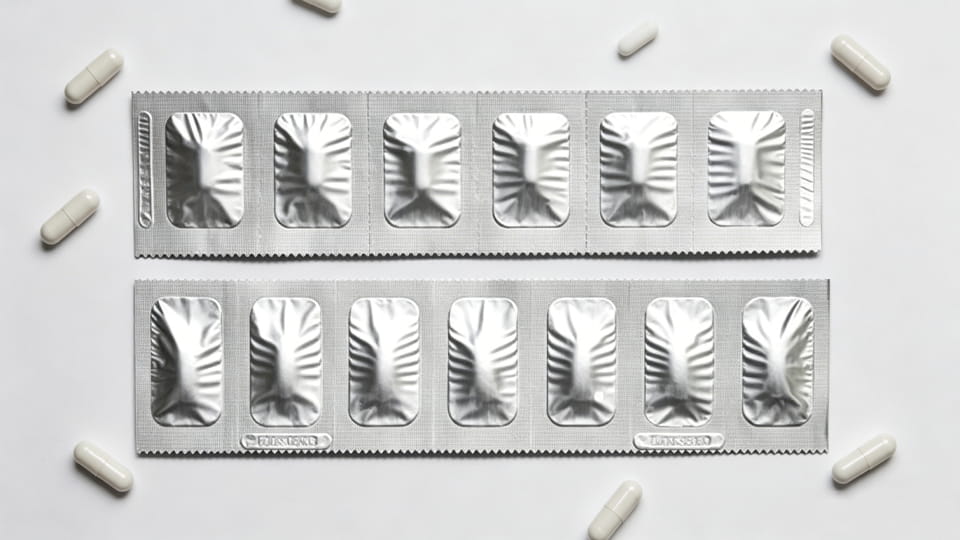
Πακέτα απογυμνώσεων (ονομάζονται επίσης ταινίες αλουμινίου ή "ζώνες δόσης") είναι μια άλλη μορφή πρωτογενούς συσκευασίας μοναδιαίας δόσης. Μια λωρίδα είναι συνήθως ένας στενός ιστός υλικού (συχνά αλουμίνιο ή πολυστρωματικό laminate) διπλωμένο και σφραγισμένο, με διατρήσεις μεταξύ των δόσεων. Σε αντίθεση με τις φουσκάλες, Αντίθετα, οι λωρίδες δεν σχηματίζουν βαθιές κοιλότητες, η κάψουλα τοποθετείται ανάμεσα σε δύο στρώσεις φιλμ. Τα strip pack χρησιμοποιούνται ευρέως στην Ασία και για προϊόντα όπου απαιτείται υψηλή προστασία από την υγρασία, αλλά οι φουσκάλες είναι πολύ ογκώδεις. Βασικά σημεία:

Από άποψη επιλογής, Τα πακέτα ταινιών καταλαμβάνουν μια μέση λύση μεταξύ του μπουκαλιού (όγκος) και φουσκάλα (ακαμψία). Συχνά επιλέγονται όταν πολύ υψηλό φράγμα υγρασίας χρειάζεται αλλά σε ένα λεπτό, ελαφριά μορφή. (Σημείωμα: Τα πακέτα ταινιών ενδέχεται να μην παρέχουν στοιχεία παραβίασης τόσο καθαρά όσο οι κυψέλες, εκτός εάν χρησιμοποιούνται πρόσθετες σφραγίδες.)
Αφού επιλέξετε ένα κύριο πακέτο, κάψουλες τοποθετούνται συνήθως σε δευτερεύουσα συσκευασία όπως χαρτοκιβώτια ή συρρικνωμένο-τυλιγμένο περιπτώσεις. Τα χαρτοκιβώτια παρέχουν επιπλέον προστασία κατά την αποστολή, επιτρέπουν πρόσθετη επισήμανση (πληροφορίες φαρμάκων, επωνυμία, σφραγίδες που δεν παραβιάζονται), και βοηθούν στην οργάνωση προϊόντων πολλαπλών συσκευασιών. Βασικά στοιχεία για τη δευτερεύουσα συσκευασία:
Συνοπτικά, Η δευτερεύουσα συσκευασία τυλίγει το προϊόν για τελική διανομή και διασφαλίζει ότι όλα τα χαρακτηριστικά επισήμανσης και συμμόρφωσης είναι στη θέση τους. Ο καλός σχεδιασμός εδώ βελτιώνει την εικόνα της επωνυμίας και την ευκολία του χρήστη (χαρτοκιβώτια που ανοίγουν εύκολα, σαφής επισήμανση).
Η επιλογή του σωστού υλικού συσκευασίας είναι κρίσιμη για την προστασία των καψουλών. Τα υλικά πρέπει να είναι συμβατά με το φάρμακο, ανταποκρίνονται στις ανάγκες φραγμού, και συμμορφώνονται με τους κανονισμούς (ISO 15378 GMP για υλικά συσκευασίας, και τα λοιπά.). Τα κοινά υλικά περιλαμβάνουν:
Γενικά, επιλέξτε το υψηλότερο φράγμα που έχει νόημα για την κάψουλα σας. Για παράδειγμα, μια ευαίσθητη στην υγρασία κάψουλα ζελατίνης μπορεί να δικαιολογεί κυψέλη Alu-Alu ή ένα πορτοκαλί γυάλινο μπουκάλι με ξηραντικό. Σε αντίθεση, Μη ευαίσθητες κάψουλες μπορεί να χρησιμοποιούν τυπική κυψέλη PVC ή φιάλη HDPE. Εξετάστε επίσης τη διάρκεια ζωής: Τα μακροπρόθεσμα προϊόντα χρειάζονται πιο ισχυρό φράγμα.
Σημειώσεις ασφαλείας/ανακύκλωσης: Οι βιώσιμες τάσεις ευνοούν τις συσκευασίες ενός υλικού (π.χ.. Κυψέλες PP, ανακυκλώσιμα μπουκάλια PET). Δοκιμές υλικών για εκχυλίσματα, αρμονία, και πληρούν τα όρια FDA/ΕΕ (π.χ.. USP <661> για πλαστικοποιητές) αποτελεί μέρος της συμμόρφωσης με τη συσκευασία.
Οι κάψουλες κυκλοφορούν σε διάφορους τύπους και η σύνθεσή τους υπαγορεύει τις ανάγκες συσκευασίας:
Εν συντομία, ο υλικό κελύφους κάψουλας και τύπος πλήρωσης θα επηρεάσει την επιλογή συσκευασίας. Οι εξαιρετικά ευαίσθητες στην υγρασία συνθέσεις ευνοούν πάντα το πιο σφιχτό φράγμα (Κυψέλη Alu-Alu, φακελάκι από αλουμινόχαρτο, ή σφραγισμένη φιάλη με ξηραντικό). Τα λιγότερο ευαίσθητα σκευάσματα έχουν περισσότερες επιλογές. Πάντα να δοκιμάζετε τη σταθερότητα του τελική συσκευασμένη κάψουλα (Το ICH Q1A απαιτεί σταθερότητα στο προτεινόμενο δοχείο).
Η επιλογή μιας βέλτιστης συσκευασίας κάψουλας περιλαμβάνει την αξιολόγηση πολλών παραγόντων: δοσολογική μορφή, συμμόρφωση, ευαισθησία προϊόντος, διάρκεια ζωής, κανονιστικές απαιτήσεις, και κόστος. Ακολουθεί ένα απλοποιημένο πλαίσιο απόφασης:
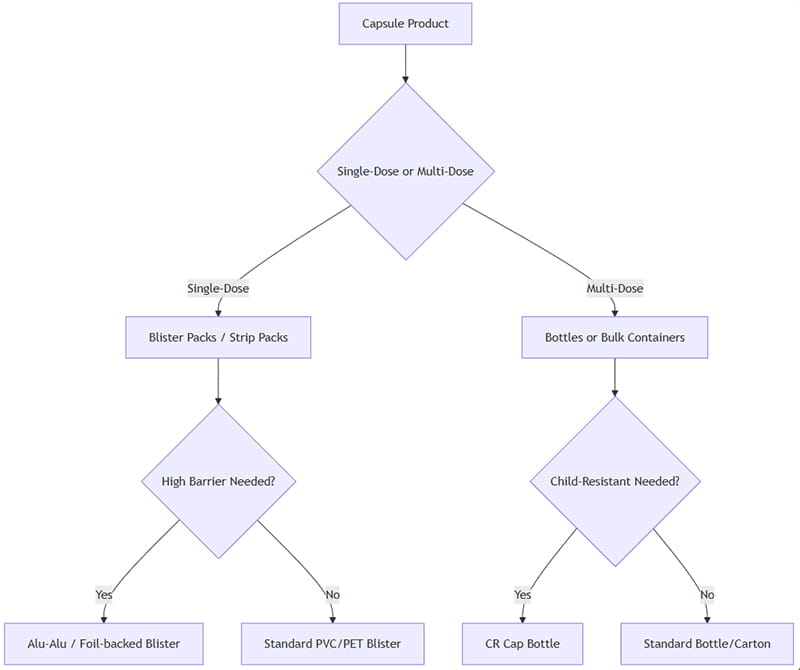
Πώς να το εφαρμόσετε αυτό: Εάν οι κάψουλες προορίζονται ως μεμονωμένες μονάδες (π.χ.. σφιχτές δόσεις ή κάρτες λιανικής με κυψέλες), προτιμώνται φουσκάλες/λωρίδες. Για πολλαπλές δόσεις (π.χ.. συμπληρώματα ή φάρμακα υψηλής χρήσης), τα μπουκάλια είναι πιο αποτελεσματικά. Εάν η προστασία υγρασίας/φωτός είναι κρίσιμη, επιλέξτε επιλογές υψηλού φραγμού (Alu-Alu, κεχριμπαρένιο μπουκάλι). Εάν απαιτείται παιδιατρική ασφάλεια, χρησιμοποιήστε πώματα για μπουκάλια ανθεκτικά για παιδιά ή σχέδια κυψέλες που δεν παραβιάζονται.
Άλλα κριτήρια από τον κλάδο περιλαμβάνουν:
Τελικά, πιλοτική δοκιμή (μελέτες σταθερότητας, καταναλωτικές μελέτες) και η ανάλυση συνολικού κόστους θα καθοδηγήσει την τελική απόφαση. Το παραπάνω διάγραμμα ροής είναι ένα σημείο εκκίνησης για τυπικά σενάρια.
Η συσκευασία των καψουλών απαιτεί εξειδικευμένα μηχανήματα. Αφού γεμίσουν οι κάψουλες (με πληρωτικά καψουλών), Αυτά μεταφέρονται σε γραμμές συσκευασίας:
Συμμόρφωση με τον εξοπλισμό: Όλες οι μηχανές συσκευασίας για φαρμακευτικά προϊόντα πρέπει να πληρούν το σχέδιο GMP (ανοξείδωτο χάλυβα, επιφάνειες που καθαρίζονται, επικυρωμένη απόδοση). Τα μηχανήματα της Jinlu έχουν πιστοποίηση CE/cGMP, συχνά με ρυθμιζόμενα προστατευτικά και συστήματα απόρριψης για χάπια εκτός προδιαγραφών. Η ενσωμάτωση είναι το κλειδί: για παράδειγμα, μια μηχανή blister μπορεί να τροφοδοτηθεί απευθείας σε ένα χαρτόνι που στήνει κουτιά και συσκευάζει κάθε κάρτα blister. Ολοκληρωμένα συστήματα (από το γέμισμα στο τελικό κουτί) επιτρέπουν την αυτοματοποίηση από άκρο σε άκρο της συσκευασίας των καψουλών.
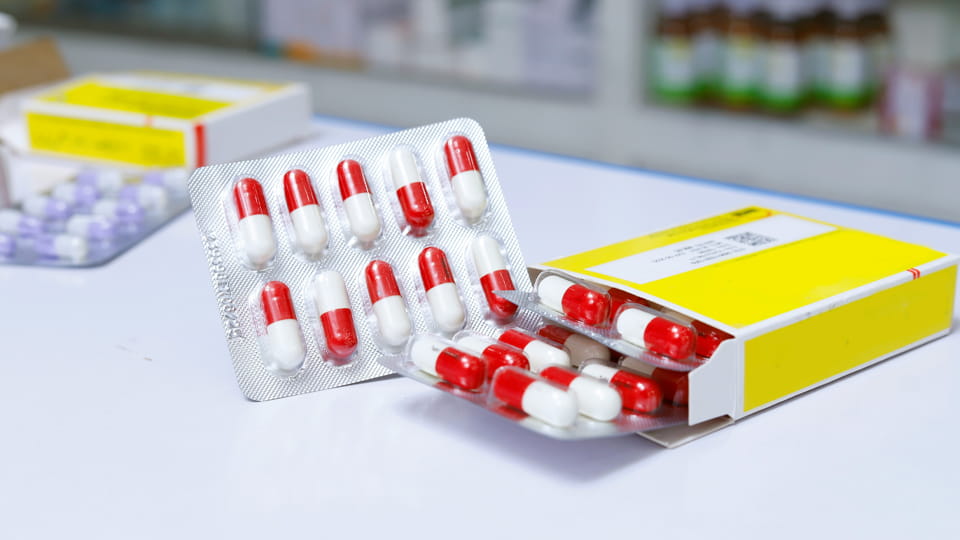
Οι συσκευασίες καψουλών για φαρμακευτικά προϊόντα πρέπει να τηρούν αυστηρούς κανονισμούς. Τα βασικά σημεία περιλαμβάνουν:
Ολικός, συνάντηση FDA, Οι οδηγίες του EMA και του ΠΟΥ είναι απαραίτητες. Πολλοί συσκευαστές συνεργάζονται με ελεγχόμενους προμηθευτές (όπως η Τζινλού) που μπορεί να παρέχει επικυρωμένα μηχανήματα και τεκμηρίωση (IR/WH/PQ).
Η επιλογή της σωστής συσκευασίας κάψουλας απαιτεί προστασία εξισορρόπησης, χρηστικότητα, και κόστος. Φουσκάλες, μπουκάλια, φακελάκια, και οι λωρίδες έχουν τη θέση τους φαρμακευτική συσκευασία. Παράγοντες όπως η ευαισθησία στην υγρασία, μορφή δόσης, και οι ανάγκες των ασθενών καθοδηγούν την απόφαση. Κατά μήκος της πλευράς, πρέπει να πληρούνται αυστηρές κανονιστικές απαιτήσεις και απαιτήσεις GMP.
Στο Συσκευασία Jinlu, ειδικευόμαστε σε ολοκληρωμένες λύσεις συσκευασίας φαρμακευτικών προϊόντων. Ο εξοπλισμός μας – από μηχανές κυψελών υψηλής ταχύτητας να πλήρως αυτοματοποιημένη καταμέτρηση & γραμμές εμφιάλωσης – σας βοηθά να συσκευάζετε τις κάψουλες αποτελεσματικά και με συμμόρφωση. Υποστηρίζουμε επίσης βιώσιμες επιλογές (π.χ.. μηχανήματα που λειτουργούν νέες ανακυκλώσιμες μεμβράνες) και προσφέρουν υπηρεσία μίας στάσης (προσαρμογή, νομιμοποίηση). Η ομάδα μας μπορεί να σας βοηθήσει να αξιολογήσετε το καλύτερο σύστημα συσκευασίας καψουλών για το προϊόν σας.
Έτοιμοι να βελτιστοποιήσετε τη συσκευασία της κάψουλάς σας? Επικοινωνήστε με την Jinlu Packing σήμερα για καθοδήγηση και προσφορές ειδικών. Οι λύσεις μας υποστηρίζονται από παγκόσμια πρότυπα ποιότητας και άνω 3,000 ικανοποιημένους πελάτες παγκοσμίως. Ας συσκευάσουμε τις κάψουλές σας για επιτυχία!
Η συσκευασία κάψουλας αναφέρεται στη διαδικασία εγκλεισμού φαρμακευτικών καψουλών σε προστατευτικά υλικά για τη διατήρηση της σταθερότητας, ασφάλεια, και συμμόρφωση. Οι συνήθεις μορφές περιλαμβάνουν συσκευασίες blister, μπουκάλια, και συσκευασία strip.
Στη φαρμακευτική πράξη, Η συσκευασία δεν είναι απλώς περιορισμός - προστατεύει τις κάψουλες από την υγρασία, οξυγόνο, και μόλυνση, που επηρεάζει άμεσα την ποιότητα και τη διάρκεια ζωής του φαρμάκου.
Δεν υπάρχει ενιαία «καλύτερη» επιλογή—εξαρτάται από το προϊόν και την αγορά σας.
• Συσκευασία blister → καλύτερη για ευαίσθητα στην υγρασία ή φάρμακα υψηλής αξίας
• Συσκευασία μπουκαλιών → καλύτερη για μαζική αποθήκευση και συμπληρώματα
• Συσκευασία strip → κοινή σε ευαίσθητες ως προς το κόστος αγορές
Συχνά προτιμάται η συσκευασία με κυψέλες επειδή παρέχει προστασία μοναδιαίας δόσης και καλύτερη περιβαλλοντική απομόνωση.
Χρησιμοποιήστε υλικά που μπλοκάρουν τις βασικές απειλές (υγρασία, οξυγόνο, φως). Για παράδειγμα, Οι κυψέλες από αλουμινόχαρτο Alu-Alu ή τα κεχριμπαρένια γυάλινα μπουκάλια προσφέρουν εξαιρετικό φράγμα για κάψουλες ευαίσθητες στην υγρασία. Οι κυψέλες PVC ή PET με επίστρωση PVDC είναι κοινές και φθηνότερες για σταθερά προϊόντα. Οι φιάλες HDPE είναι στάνταρ, αλλά μπορεί να χρειαστούν μια θήκη αποξηραντικού εάν οι κάψουλες είναι ευαίσθητες. Αξιολογήστε τους ρυθμούς μετάδοσης υδρατμών (WVTR) των ταινιών. Στην παραγωγή με συνείδηση της βιωσιμότητας, εξετάστε ανακυκλώσιμα μονοϋλικά (π.χ.. Κυψέλες PP). Να ελέγχετε πάντα τη συμβατότητα της συσκευασίας μέσω μελετών σταθερότητας.
Οι κάψουλες είναι ευαίσθητες σε περιβαλλοντικούς παράγοντες όπως η υγρασία, οξυγόνο, και φως.
Σωστή συσκευασία:
• Αποτρέπει την απορρόφηση υγρασίας
• Μειώνει την οξείδωση
• Διατηρεί τη χημική σταθερότητα
Συσκευασία φουσκάλας, προπαντός, δημιουργεί σφραγισμένα μικρο-περιβάλλοντα για κάθε κάψουλα, παρατείνει σημαντικά τη διάρκεια ζωής.
Εξαρτάται από τη μορφή. Για φουσκάλες, χρειάζεστε μια μηχανή σχηματισμού/σφράγισης κυψελών (με καλούπια κοιλότητας για να ταιριάζει στην κάψουλα σας) συν πιθανώς ένα χαρτοκιβώτιο. Για μπουκάλια, χρειάζεστε μια γραμμή πλήρωσης/καλύμματος φιάλης (unscrambler, αντι-πληρωτικό, καπάκι, Ετικέτα). Για πακέτα ραβδιών, χρησιμοποιήστε μια μηχανή φακελίσκου κάθετης μορφής-γεμίσματος-σφράγισης. Το Jinlu προσφέρει όλα αυτά: π.χ.. οι κυψέλες της σειράς DPP μας, Γραμμή μέτρησης JL-16H, και συσκευαστής φακελλίσκων JL-VP. Η επιλογή του σωστού μηχανήματος απαιτεί χωρητικότητα, μέγεθος κάψουλας, και απαιτούνται ενσωματώσεις (συστήματα όρασης, απόρριψη σταθμών, και τα λοιπά.).
Λάβετε υπόψη παράγοντες όπως η δοσολογία (single vs. πολλαπλούς), ανάγκες σταθερότητας, και χρήση από τον ασθενή. Για ευκολία μιας δόσης και υψηλή προστασία (ειδικά για κάψουλες ευαίσθητες στην υγρασία/φως), Συχνά προτιμώνται οι φουσκάλες. Για μεγάλου όγκου ή οικονομικούς λόγους, συνήθως επιλέγονται μπουκάλια. Επίσης, εάν χρειάζονται χαρακτηριστικά ανθεκτικά στα παιδιά ή για ηλικιωμένους, τα μπουκάλια μπορεί να προσφέρουν περισσότερες επιλογές. Κατά κανόνα, αντιστοιχίστε τις ιδιότητες της κάψουλάς σας με τη συσκευασία: π.χ.. πολύ υγροσκοπικές κάψουλες → Alu-Alu blister; συμπληρώματα υψηλής ζήτησης → μπουκάλια.
Αναφορές:
1.Παράρτημα 9 Οδηγίες για τη συσκευασία φαρμακευτικών προϊόντων -- ΠΟΥ
2.Τεχνικές Απαιτήσεις Φαρμάκων και Προϊόντων Υγείας -- Παγκόσμιος Οργανισμός Υγείας
3.Ασφάλεια Φαρμακευτικών Υλικών Συσκευασίας και Φαρμάκων: Μια μίνι κριτική —— MDPI
4.Συσκευασία και ένθετα φαρμακείου —— Εθνική Βιβλιοθήκη Ιατρικής
5.Ανασκόπηση για τον χαρακτηρισμό και την ανακυκλωσιμότητα των φαρμακευτικών blisters —— ScienceDirect

Πέτι Φου, Ιδρυτής της Jinlupacking, φέρνει πάνω 20 χρόνια εμπειρίας στον τομέα των φαρμακευτικών μηχανημάτων. Υπό την ηγεσία του, Η Jinlu έχει εξελιχθεί σε έναν αξιόπιστο προμηθευτή που ενσωματώνει το σχεδιασμό, παραγωγή, και πωλήσεις. Ο Petty είναι παθιασμένος με το να μοιράζεται τις βαθιές του γνώσεις στον κλάδο για να βοηθήσει τους πελάτες να πλοηγηθούν στην πολυπλοκότητα της συσκευασίας φαρμάκων, εξασφαλίζοντας ότι δεν λαμβάνουν μόνο εξοπλισμό, αλλά μια πραγματική συνεργασία ενιαίας εξυπηρέτησης προσαρμοσμένη στους στόχους παραγωγής τους.