
การซื้ออุปกรณ์ยาใหม่เป็นเพียงครึ่งหนึ่งของงาน แต่คำถามที่แท้จริงก็คือ: มันจะผ่าน IQ ได้ไหม, โอคิว, และพีคิวเอ?
ใน การผลิตยา, เครื่องจักรที่ไม่ผ่านการรับรองอย่างเหมาะสมถือเป็นความเสี่ยง, ไม่ใช่สินทรัพย์. หน่วยงานกำกับดูแลคาดหวังหลักฐานที่ชัดเจนว่าอุปกรณ์ได้รับการติดตั้งอย่างถูกต้อง, ดำเนินการภายในขอบเขต, และดำเนินการอย่างสม่ำเสมอในสภาวะการผลิตจริง . หากไม่มีสิ่งนั้น, แม้แต่สายการบรรจุที่ทันสมัยที่สุดก็ไม่สามารถนำมาใช้ในการผลิต GMP ได้.
นั่นเป็นเหตุผลที่เข้าใจ IQ, โอคิว, และ PQ ไม่ได้เป็นเพียงรายละเอียดทางเทคนิคเท่านั้น แต่ยังส่งผลกระทบโดยตรงต่อการปฏิบัติตามข้อกำหนดของคุณ, เส้นเวลาของโครงการ, และการลงทุนของคุณให้คุณค่าจริงหรือไม่.
ไอคิว, OQ และ PQ ยืนหยัดเพื่อ คุณสมบัติการติดตั้ง, คุณสมบัติการปฏิบัติงาน, และ คุณสมบัติการปฏิบัติงาน. ขั้นตอนเหล่านี้เป็นขั้นตอนตามลำดับในอุปกรณ์ที่มีคุณสมบัติตามที่กำหนดก่อนใช้ในการผลิตยา GMP. ในระยะสั้น: ไอคิว ตรวจสอบว่าเครื่องได้รับการติดตั้งอย่างถูกต้อง; โอคิว ทดสอบว่าทำงานได้อย่างถูกต้องภายใต้การตั้งค่าที่จำเป็นทั้งหมด; PQ ตรวจสอบว่ามีประสิทธิภาพสม่ำเสมอในระหว่างการผลิตจริง. ตามที่อย, “คุณสมบัติ” หมายความว่า การแสดงว่าอุปกรณ์เหมาะสมกับการใช้งานตามวัตถุประสงค์. IQ/OQ/PQ เป็นกิจกรรมที่บันทึกไว้เพื่อพิสูจน์ความเหมาะสมและการทำงานในแต่ละขั้นตอน. ด้วยกัน, พวกเขาเป็นส่วนหนึ่งของภาพรวม การตรวจสอบ กระบวนการสำหรับระบบการผลิต, สร้างความมั่นใจในคุณภาพของผลิตภัณฑ์และการปฏิบัติตามข้อกำหนด.
คุณสมบัติของอุปกรณ์เทียบกับ. การตรวจสอบกระบวนการ: IQ/OQ/PQ มุ่งเน้นไปที่เครื่องจักร (“คุณสมบัติอุปกรณ์”), ในขณะที่การตรวจสอบกระบวนการครอบคลุมกระบวนการผลิตทั้งหมด. ในทางปฏิบัติ, คุณสมบัติของอุปกรณ์มักเป็นขั้นตอนแรกในแผนแม่บทการตรวจสอบ. โดยการเติม IQ/OQ/PQ ด้วยฟิลเลอร์แคปซูล, เครื่องตุ่ม, สายขวด, ฯลฯ, ผู้ผลิตได้รับเอกสารหลักฐานที่แสดงว่าอุปกรณ์นั้น ตรงตามข้อกำหนด และพร้อมสำหรับการผลิต.
| เวที (IR/WH/PQ) | วัตถุประสงค์ | กิจกรรมสำคัญ / การทดสอบ | เอกสารทั่วไปที่ผลิต |
| ไอคิว | ตรวจสอบการติดตั้งอุปกรณ์ที่ถูกต้อง | ตรวจสอบสถานที่, สาธารณูปโภค (พลัง, อากาศ, น้ำ), การติดตั้งต่อคู่มือ; บันทึกหมายเลขซีเรียลและอุปกรณ์เสริม | โปรโตคอล IQ/รายการตรวจสอบและรายงาน |
| โอคิว | ตรวจสอบฟังก์ชันการทำงานภายใต้ขีดจำกัด | ทำการทดสอบในทุกช่วงการทำงาน (ความเร็วต่ำสุด/สูงสุด, อุณหภูมิ, ความกดดัน); ทดสอบสัญญาณเตือนและลูกโซ่, ตรวจสอบการควบคุม | โปรโตคอล OQ และรายงาน |
| PQ | พิสูจน์ประสิทธิภาพที่สม่ำเสมอในการผลิต | รันชุดการผลิตหลายชุดกับผลิตภัณฑ์จริง (หรือแกล้งทำเป็น) ในสภาวะที่เลวร้ายที่สุด; ตรวจสอบผลลัพธ์ (ผลผลิต, ความแม่นยำ) | โปรโตคอลและรายงาน PQ |
โต๊ะ 1: การเปรียบเทียบไอคิว, โอคิว, ขั้นตอน PQ (ดัดแปลงมาจากมาตรฐานอุตสาหกรรม).

อุปกรณ์ทางเภสัชกรรม ต้องปฏิบัติตามกฎระเบียบ GMP ที่เข้มงวด. ในสหรัฐอเมริกา, อย 21 ส่วนซีเอฟอาร์ 211 (ยา) และ 21 ซีเอฟอาร์ 820 (อุปกรณ์การแพทย์) ต้องมีการตรวจสอบระบบการผลิตอย่างชัดเจน. IQ/OQ/PQ เป็นหัวใจสำคัญของการปฏิบัติตามข้อกำหนดนี้. ตัวอย่างเช่น, FDA ระบุว่ากิจกรรมการรับรองคุณสมบัติ “จำเป็นต้องนำหน้าผลิตภัณฑ์การผลิตในระดับเชิงพาณิชย์”. ในสหภาพยุโรป, ภาคผนวก GMP 15 ในทำนองเดียวกันจะกำหนดคุณสมบัติของอุปกรณ์ตลอดวงจรการใช้งาน. จุดมุ่งหมายคือเพื่อให้แน่ใจว่าอุปกรณ์จะผลิตผลิตภัณฑ์ที่ปลอดภัยได้อย่างน่าเชื่อถือ.
ไม่มี IQ/OQ/PQ ที่เหมาะสม, บริษัทต่างๆ ต้องเผชิญกับความเสี่ยงร้ายแรง: ข้อบกพร่องของผลิตภัณฑ์, การเรียกคืนแบทช์, การอ้างอิงตามกฎระเบียบ, และอันตรายด้านความปลอดภัย. คุณสมบัติที่เหมาะสมสร้างความมั่นใจให้กับตัวเครื่อง จริงๆ แล้ว ทำในสิ่งที่ควรทำ เช่น, ว่าเครื่องบรรจุภัณฑ์พลาสติกปิดผนึกที่อุณหภูมิที่เหมาะสมหรือตัวเติมของเหลวจะจ่ายปริมาตรที่ถูกต้อง. ในทางปฏิบัติ, โปรแกรมการตรวจสอบที่สมบูรณ์จะช่วยหลีกเลี่ยงเรื่องไม่คาดคิด: “เมื่อทำถูกต้องแล้ว, การรับรองทั้งสามขั้นตอนมีความพิถีพิถันและใช้เวลานาน. อย่างไรก็ตาม, สิ่งเหล่านี้มีความสำคัญอย่างยิ่งต่อการรับประกันกระบวนการที่ทำซ้ำได้และคุณภาพของผลิตภัณฑ์ที่มั่นคง”. ในระยะสั้น, จำเป็นต้องมี IQ/OQ/PQ ในการตั้งค่า GMP. สำหรับอย่างใดอย่างหนึ่ง สายการบรรจุ (พุพอง, ขวด, ผู้บรรจุกล่อง, ฯลฯ), IQ/OQ/PQ จะต้องเป็นไปตามข้อกำหนดของทั้ง FDA และ EU. เครื่องจักรของ Jinlu มาพร้อมกับเทมเพลตเอกสาร IQ/OQ/PQ เต็มรูปแบบ, เพราะ, ตามที่ผู้เชี่ยวชาญของเรากล่าว, “เครื่องจะต้องพร้อมสำหรับการตรวจสอบ”.
ใน ระยะไอคิว, เราตรวจสอบว่ามีการส่งมอบอุปกรณ์และตั้งค่าอย่างถูกต้อง. ซึ่งหมายถึงการตรวจสอบเครื่องจักรและสภาพแวดล้อมก่อนการทดสอบการผลิตใดๆ. การตรวจไอคิวที่สำคัญได้แก่:
ผลลัพธ์ของ IQ คือรายงาน/โปรโตคอล IQ ที่เป็นลายลักษณ์อักษร ซึ่งจะบันทึกการตรวจสอบแต่ละรายการและยืนยันว่า "ติดตั้งตามที่คาดไว้" หากมีสิ่งใดไม่ตรงตามสเปก, จะต้องได้รับการแก้ไขก่อนที่จะดำเนินการต่อ. IQ ที่ประสบความสำเร็จหมายถึง “อุปกรณ์ได้รับการติดตั้งตามที่ตั้งใจไว้” ซึ่งเป็นข้อกำหนดเบื้องต้นสำหรับการทดสอบใดๆ. (ในทางปฏิบัติ, การทดสอบบางอย่างอาจเริ่มต้นจาก IQ เป็นรายการ "พร้อมสำหรับการทดสอบ", แต่เกณฑ์การยอมรับจะอยู่ใน OQ/PQ)
OQ ทดสอบการทำงานของอุปกรณ์ ภายใต้สภาวะที่ได้รับการควบคุม. ตอนนี้เครื่องได้รับการติดตั้งอย่างถูกต้องแล้ว, เราตรวจสอบ พารามิเตอร์การทำงานทั้งหมด และระบบควบคุม. โดยทั่วไป OQ จะประกอบด้วย:
ที่ รายงานสีขาว จะแสดงผลการทดสอบแต่ละครั้งและเปรียบเทียบกับเกณฑ์การยอมรับ (ซึ่งมาจากข้อกำหนดการออกแบบหรือ URS). หลังจากที่ OQ ผ่านแล้วเท่านั้นที่เราจะดำเนินการต่อ. (บางครั้ง OQ และ IQ อาจทับซ้อนกันในรายงานรวมที่เรียกว่า IOQ, แต่ตรรกะยังคงเป็นไปตามลำดับ)
เป้าหมายสำคัญของ OQ คือ: “ภายใต้เงื่อนไขที่กำหนดทั้งหมด, เครื่องทำสิ่งที่ควรทำหรือไม่?- หากสิ่งใดล้มเหลว (E.G. เซ็นเซอร์ไม่ตรงแนว), มันได้รับการแก้ไขและทดสอบอีกครั้ง.
PQ เป็นขั้นตอนสุดท้าย, โดยที่เราพิสูจน์ว่าอุปกรณ์ทำงานอย่างถูกต้องระหว่างการผลิตจริง. ในขณะที่ OQ อาจจะว่างเปล่าไปแล้ว, PQ ใช้สินค้าจริงหรือสินค้าจำลอง. ขั้นตอนได้แก่:
ในตอนท้ายของ PQ, เราได้บันทึกหลักฐานไว้แล้ว “อุปกรณ์ที่ผ่านการรับรองจะผลิตผลิตภัณฑ์ที่สอดคล้องภายใต้สภาวะจริงอย่างสม่ำเสมอ” กล่าวอีกนัยหนึ่ง, เครื่องพร้อมสำหรับการผลิต GMP.
ตัวอย่างเช่น, ถ้า PQ ประสบผลสำเร็จบนเครื่องบรรจุแคปซูล, เรารู้ว่าความแม่นยำในการจ่ายสาร, การใส่แคปซูล, และความเร็วของโรเตอร์ทำงานข้ามกะโดยไม่มีการดริฟท์. การดำเนินการนี้ทำให้การตรวจสอบ "สามเหลี่ยม" เสร็จสมบูรณ์ และให้ความมั่นใจแก่ QA/หน่วยงานกำกับดูแลว่าสายการผลิตจะไม่สร้างชุดงานที่ไม่ดี. จากนั้นอุปกรณ์จะถูกปล่อยสำหรับการผลิตตามปกติและรวมอยู่ในตารางการบำรุงรักษาตามปกติ.

สิ่งสำคัญคือต้องชี้แจงคำศัพท์. คุณสมบัติ มักจะหมายถึงอุปกรณ์, ในทางตรงกันข้าม การตรวจสอบ มักหมายถึงกระบวนการหรือระบบ. ในกรอบ GMP จำนวนมาก, มีการพิจารณา IQ/OQ/PQ ส่วนของการตรวจสอบ ของระบบการผลิต. ในแง่นี้:
ดังนั้น, อุปกรณ์ IQ/OQ/PQ มีคุณสมบัติตรงตามคุณสมบัติของเครื่องจักร. แบบแยก การตรวจสอบกระบวนการ จะเชื่อมโยงเครื่องจักรเหล่านั้นเข้ากับกระบวนการผลิตจริง (เช่นการบีบอัดแท็บเล็ตพร้อมการเคลือบ, ฯลฯ). ในความเป็นจริง, บางครั้งเรียกว่า IQ/OQ/PQ คุณสมบัติอุปกรณ์ (อีคิว) ขั้นตอน. ก คุณสมบัติการออกแบบ (ดีคิว) ขั้นตอนอาจมาก่อน IQ, ทำให้มั่นใจได้ว่าการออกแบบตรงตามความต้องการของผู้ใช้ (สสส) ก่อนสร้างหรือซื้ออุปกรณ์.
ต่อไปนี้เป็นขั้นตอนง่ายๆ ในวงจรชีวิตของโครงการ:
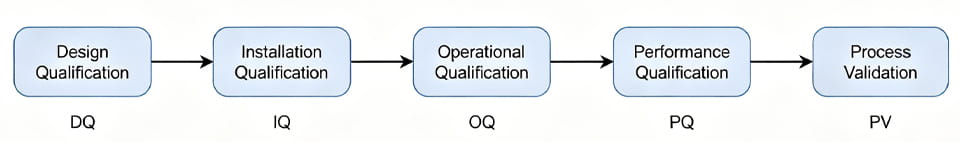
ลูกศรแต่ละอันเป็นแฮนด์ออฟ: คุณต้องจบ DQ (การตรวจสอบการออกแบบตรงตาม URS) ก่อนไอคิว (ตรวจสอบการติดตั้งที่เหมาะสม). หลังจาก PQ เสร็จสมบูรณ์แล้วเท่านั้น คุณจึงจะอ้างสิทธิ์ได้ว่าอุปกรณ์ได้รับการตรวจสอบและเริ่มการตรวจสอบกระบวนการแล้ว (E.G. ดำเนินแคมเปญการผลิตจริง).
คุณสมบัติอุปกรณ์เป็นส่วนหนึ่งของอุปกรณ์โดยรวม วงจรชีวิต:
แต่ละขั้นตอนจัดทำเอกสาร. ตัวอย่างเช่น, เครื่องบรรจุภัณฑ์ของ Jinlu จัดส่งพร้อมกับ ชุดตรวจสอบ IQ/OQ/PQ และโปรโตคอล FAT/SAT. นั่นหมายความว่าผู้ซื้อสามารถประหยัดเวลาได้โดยใช้เทมเพลตของผู้ผลิตในระหว่างการตรวจสอบคุณสมบัติ. ไทม์ไลน์โดยทั่วไปอาจครอบคลุมหลายสัปดาห์ถึงหลายเดือน ขึ้นอยู่กับความซับซ้อน.
ตามที่อย./สหภาพยุโรป, ทีมงาน QA และวิศวกรที่ผ่านการฝึกอบรมแล้ว (และบางครั้งที่ปรึกษาภายนอก) ควรดำเนินการหรือเป็นสักขีพยานในขั้นตอนเหล่านี้. บรรทัดล่าง: มีระเบียบวินัย, ขั้นตอนการออกแบบที่จัดทำเป็นเอกสารผ่าน PQ ช่วยให้มั่นใจได้ว่าสายการผลิตของคุณเป็นไปตามข้อกำหนด.
เครื่องบรรจุภัณฑ์ทุกประเภทในโรงงาน GMP จำเป็นต้องมีคุณสมบัติ. นี่คือตัวอย่างทั่วไป:
ภาพ: เครื่องบรรจุแคปซูลกึ่งอัตโนมัติ (จินหลู CNT-209). การตรวจสอบความถูกต้องของเครื่องจักรดังกล่าวเกี่ยวข้องกับการตรวจสอบการวางแนวแคปซูลอย่างระมัดระวัง, ความแม่นยำในการจ่ายยา, และการปิดผนึกระหว่าง IQ/OQ/PQ.
ตัวอย่างกรณี: เครื่องบรรจุแคปซูล IQ/OQ/PQ
เพื่อแสดงให้เห็น, ลองนึกภาพการเข้ารอบ เครื่องบรรจุแคปซูลอัตโนมัติเต็มรูปแบบ สำหรับวิตามิน:
เมื่อขั้นตอนเหล่านี้ผ่านผลสำเร็จแล้ว, ฟิลเลอร์แคปซูลก็คือ มีคุณสมบัติสำหรับการผลิต. พร้อมเริ่มบรรจุภัณฑ์สินค้าจริงได้อย่างมั่นใจ.
ถึงแม้จะมีความสำคัญก็ตาม, คุณสมบัติของอุปกรณ์มักจะเผชิญกับอุปสรรค:
ประสบการณ์ของ Jinlu ก็คือ วิธีที่ดีที่สุดในการเอาชนะความท้าทายเหล่านี้คือการวางแผน. เริ่มการวางแผนการตรวจสอบตั้งแต่เนิ่นๆ, กำหนด URS และแผนการทดสอบกับผู้ขาย, และตรวจสอบให้แน่ใจว่าผู้มีส่วนได้ส่วนเสียทั้งหมดเห็นด้วยกับหลักเกณฑ์. การใช้เทมเพลตและรายการตรวจสอบยังช่วยเร่งกระบวนการอีกด้วย.

เพื่อปรับปรุง IQ/OQ/PQ และรับประกันผลลัพธ์ที่เป็นไปตามข้อกำหนด:
โดยปฏิบัติตามแนวทางปฏิบัติที่ดีที่สุดเหล่านี้, บริษัทสามารถลดความประหลาดใจในระหว่างการตรวจสอบได้. ดังที่ Petty Fu ผู้ก่อตั้ง Jinlu มักตั้งข้อสังเกตไว้: คัดเลือกเครื่องจักรจากซัพพลายเออร์ GMP ที่มีประสบการณ์ “รับประกันว่าจะมีผลลัพธ์ที่แม่นยำ, ใบรับรองการปฏิบัติตาม, และการสนับสนุนในท้องถิ่น”. กล่าวอีกนัยหนึ่ง, ตรวจสอบด้วยความมั่นใจและเลือกพันธมิตรที่พูดเรื่องการตรวจสอบแล้ว.

ในการผลิตยา, ไอคิว, โอคิว, และ PQ ไม่สามารถต่อรองได้. ขั้นตอนการตรวจสอบความถูกต้องตามโครงสร้างเหล่านี้พิสูจน์ว่าอุปกรณ์ของคุณ ตั้งแต่เครื่องบรรจุแคปซูลไปจนถึงเครื่องบรรจุตุ่ม ไปจนถึงสายการผลิตขวด เหมาะสมกับวัตถุประสงค์และเป็นไปตามมาตรฐาน GMP. โปรแกรม IQ/OQ/PQ ที่ดำเนินการอย่างดีจะช่วยป้องกันข้อผิดพลาดที่มีค่าใช้จ่ายสูงและรับประกันคุณภาพที่สม่ำเสมอ.
การเลือกซัพพลายเออร์ที่มีการสนับสนุนการตรวจสอบที่รัดกุมเป็นสิ่งสำคัญ. เครื่องบรรจุภัณฑ์ยาของ Jinlu Packing มาด้วย การออกแบบที่พร้อมใช้ GMP และ เอกสารที่ครอบคลุม (โปรโตคอล FAT/SAT/IQ/OQ/PQ) เพื่อปรับปรุงการปฏิบัติตามข้อกำหนด. ทีมของเราสามารถช่วยคุณกำหนด URS ได้, ทำการทดสอบ IQ/OQ, และสร้างรายงานที่จำเป็น.
👉ตามหา อุปกรณ์ยา ที่ช่วยลดความยุ่งยากในการปฏิบัติตาม? ติดต่อ Jinlu วันนี้ สำหรับเครื่องจักรบรรจุภัณฑ์ที่รองรับ IQ/OQ/PQ เต็มรูปแบบ และบริการตรวจสอบความถูกต้องแบบครบวงจร. เราจะตรวจสอบให้แน่ใจว่าได้ติดตั้งบรรทัดใหม่ของคุณแล้ว, ทดสอบแล้ว, และจัดทำเป็นเอกสารเพื่อการรับรอง GMP ที่ไม่ยุ่งยาก.
ในแง่ง่ายๆ, คุณสมบัติการติดตั้ง (ไอคิว) ตรวจสอบให้แน่ใจว่าอุปกรณ์ได้รับการติดตั้งอย่างถูกต้อง (ตำแหน่งที่ถูกต้อง, สาธารณูปโภค, และส่วนประกอบ). คุณสมบัติการปฏิบัติงาน (โอคิว) ทดสอบการทำงานของอุปกรณ์ภายใต้การตั้งค่าปกติและสุดขั้ว (ยืนยันการควบคุมทั้งหมด, สัญญาณเตือน, และขีดจำกัด). คุณสมบัติการปฏิบัติงาน (PQ) ยืนยันว่าอุปกรณ์สามารถทำงานได้อย่างต่อเนื่องในการผลิตจริง, โดยใช้วัสดุและเงื่อนไขจริง. ด้วยกัน, เอกสาร IQ/OQ/PQ ที่เครื่องทำงานตามที่ตั้งใจไว้.
ใช่. แนวปฏิบัติด้านกฎระเบียบ (อย, สหภาพยุโรป, WHO) กำหนดให้อุปกรณ์สำคัญทั้งหมดในโรงงานผลิตยาต้องผ่านการรับรองก่อนใช้งาน. ตัวอย่างเช่น, อย 21 ซีเอฟอาร์ 211 และภาคผนวก EU GMP 15 ทั้งอาณัติคุณสมบัติอุปกรณ์ที่เข้มงวด. การข้าม IQ/OQ/PQ มีความเสี่ยงต่อการละเมิดกฎระเบียบและความเป็นไปได้ในการเรียกคืนผลิตภัณฑ์. ในทางปฏิบัติ, เครื่องบรรจุภัณฑ์ใหม่ในสายผลิตภัณฑ์ยา (แผลพุพอง, ฟิลเลอร์, กล่องกระดาษ, ฯลฯ) ควรได้รับการตรวจสอบความถูกต้องผ่าน IQ/OQ/PQ หากสัมผัสกับผลิตภัณฑ์.
โดยทั่วไปแล้วจะเป็นความพยายามร่วมกัน. ซัพพลายเออร์เครื่องจักรมักจะให้ความช่วยเหลือโดยจัดทำเอกสารและดำเนินการทดสอบการยอมรับจากโรงงาน. ทีมงานตรวจสอบคุณภาพ/การตรวจสอบของผู้ซื้อดำเนินการ (หรือพยาน) เว็บไซต์ IQ/OQ/PQ. บ่อย, OEM (พวกเขา, ตัวอย่างเช่น) เสนอบริการตรวจสอบหรือคำแนะนำซึ่งเป็นส่วนหนึ่งของการว่าจ้าง. ในที่สุด, บริษัทจัดซื้อมีหน้าที่รับผิดชอบในการตรวจสอบให้แน่ใจว่า IQ/OQ/PQ เสร็จสมบูรณ์และจัดทำเป็นเอกสารก่อนการผลิตผลิตภัณฑ์.
PQ มาหลังจาก IQ และ OQ สำเร็จแล้ว. เมื่อติดตั้งเครื่องแล้ว (ไอคิว) และผ่านการทดสอบการทำงาน (โอคิว), PQ เสร็จสิ้นระหว่างการดำเนินการผลิตจริงหรือจำลอง. โดยปกติแล้วจะเป็นขั้นตอนสุดท้าย: ตัวอย่างเช่น, ใช้งานสามชุดติดต่อกันหรือทดลองการผลิตเต็มรูปแบบพร้อมทั้งติดตามประสิทธิภาพ. PQ มักเกิดขึ้นในช่วงระยะเวลาการทดสอบเดินเครื่องครั้งแรก, ก่อนที่สายการผลิตจะออกสู่การผลิตตามปกติอย่างเป็นทางการ.
อย่างแน่นอน. แม้ว่าบรรจุภัณฑ์จะดูตรงไปตรงมาก็ตาม, หน่วยงานกำกับดูแลถือว่าเป็นส่วนหนึ่งของกระบวนการผลิตที่ได้รับการควบคุม. เครื่องบรรจุภัณฑ์หลักแต่ละเครื่องจะต้องผ่านการรับรอง. เช่น, ตามที่ระบุไว้ในเว็บไซต์ของ Jinlu, “สำหรับผลิตภัณฑ์ที่มีการควบคุม, ทำการติดตั้ง, คุณสมบัติการปฏิบัติงานและการปฏิบัติงาน (IR/WH/PQ) ตามความจำเป็น”. เครื่องบรรจุกล่องที่จัดการกับผลิตภัณฑ์สำเร็จรูปของคุณจะต้องแสดงให้เห็นว่าทำงานได้อย่างถูกต้อง (E.G. พับกล่องได้อย่างน่าเชื่อถือ, แทรกป้ายกำกับ), IQ/OQ/PQ ที่บรรลุผลสำเร็จ. ในสาระสำคัญ, อุปกรณ์ใดๆ ที่อาจส่งผลต่อคุณภาพผลิตภัณฑ์หรือการตรวจสอบย้อนกลับต้องมีคุณสมบัติ.
อ้างอิง:
1.คุณสมบัติและการตรวจสอบเอกสารอย่างเป็นทางการ – ภาคผนวก EU GMP 15.
2.แนวปฏิบัติที่ดีในการผลิตผลิตภัณฑ์ยา ภาคผนวก – ภาคผนวกคู่มือ PIC/S GMP 15 (มาตรฐาน GMP ระดับโลก).
3.ประวัติศาสตร์ & อนาคตของการตรวจสอบ – ispe.org.
4.คุณสมบัติ (ดีคิว, ไอคิว, โอคิว, PQ) – gempex.com.

เพตตี้ ฟู, ผู้ก่อตั้ง Jinlupacking, นำมาซึ่ง 20 ความเชี่ยวชาญหลายปีในภาคส่วนเครื่องจักรยา. ภายใต้การนำของเขา, Jinlu เติบโตเป็นซัพพลายเออร์ที่เชื่อถือได้โดยผสมผสานการออกแบบ, การผลิต, และการขาย. Petty มีความกระตือรือร้นในการแบ่งปันความรู้เชิงลึกในอุตสาหกรรมของเขา เพื่อช่วยลูกค้าจัดการกับความซับซ้อนของบรรจุภัณฑ์ยา, รับรองว่าพวกเขาจะได้รับไม่ใช่แค่อุปกรณ์เท่านั้น, แต่เป็นพันธมิตรด้านบริการแบบครบวงจรที่แท้จริงซึ่งปรับให้เหมาะกับเป้าหมายการผลิต.



