
캡슐 포장은 의약품 캡슐이 어떻게 포장되는지를 나타냅니다. (하드쉘 또는 소프트젤) 배포 및 사용을 위해 동봉되어 있습니다.. 그것은 다음을 포함합니다 1차 포장 (캡슐과 직접 접촉하는 용기, 예를 들어. 물집 구멍이나 병) 그리고 2차 포장 (외부 상자, 상자, 제품을 더욱 보호하고 식별하는 라벨). 좋은 캡슐 포장은 약물을 습기로부터 보호해야 합니다., 빛, 그리고 오염, 유통기한을 통한 안정성 확보, 규제 요구 사항을 충족합니다.. 예를 들어, WHO는 다음과 같이 지적합니다. “포장 구성 요소는 생산부터 사용까지 제품을 둘러싸고 있습니다.,” 보호와 같은 기능을 충족해야 합니다., 안정, 및 식별. FDA의 GMP 규정 (21 CFR 211) 수령 시 포장재 검사 및 테스트에 대한 서면 절차를 요구합니다., 안전한 라벨링 및 변조 통제를 의무화합니다.. 실제로, 우리는 일치해야합니다 캡슐 제품 (제형, 감광도, 복용량, 등.) 적절한 포장 형식과 재료로.

제약 캡슐의 1차 포장은 매우 중요합니다. 제품 보호, 규정 준수, 환자 안전: 캡슐을 건조하게 유지해줍니다, 오염되지 않은, 그리고 명확하게 라벨이 붙어있습니다. 예를 들어, 블리스터 팩과 호박병은 수분에 민감한 의약품에 이상적인 것으로 알려져 있습니다.. 올바른 포장은 환자의 규정 준수도 지원합니다. (단위 용량 수포 투여, 어린이에게 안전한 병, 명확한 라벨링) 공급망 요구사항 (직렬화, 기호 논리학). 아래에서는 각 옵션을 자세히 검토합니다..
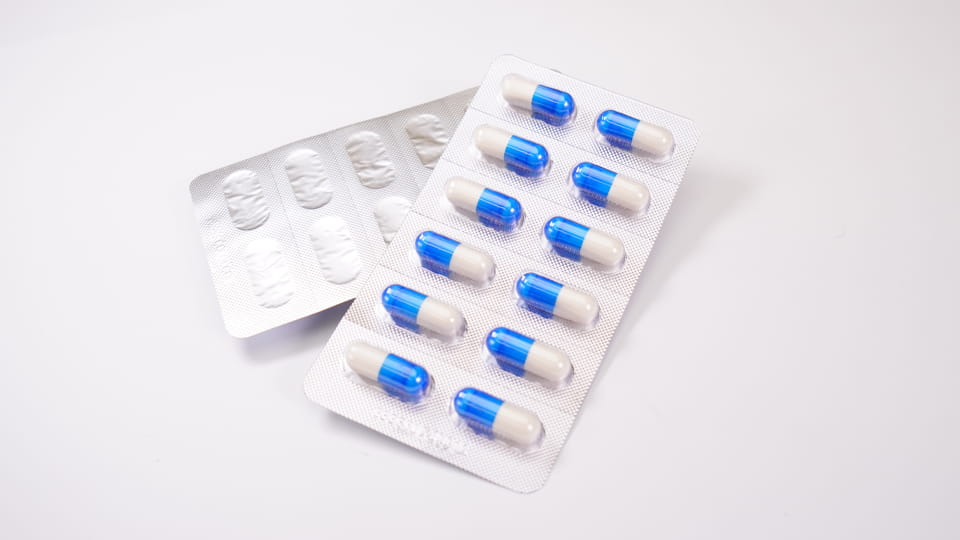
물집 팩 미리 형성된 플라스틱 공동으로 구성 (“물집”) 개별 캡슐을 담는 것, 뒷면으로 밀봉 (호일 또는 플라스틱). 이것 1차 포장 제안 단위 선량 보호. 물집이 제공하는 것 우수한 장벽 속성: 그들은로 만들 수 있습니다 PVC/PVDC 또는 포일과 같은 재료 (알루미늄-PVC, 알루-알루) 습기를 차단하기 위해, 산소와 빛. 예를 들어, Jinlu의 DPP-180Pro 블리스 터 기계는 최대 형성 및 밀봉이 가능합니다. 4,800 시간당 캡슐 물집 Alu-PVC 또는 Alu-Alu 라미네이트. 캡슐 블리스 터 포장의 주요 특징은 다음과 같습니다.:
수치:캡슐 물집을 형성하는 Jinlu JL-180Pro 물집 포장 기계 (알루미늄-PVC), 4,800 충치/시간.
Jinlu의 물집 기계 (예를 들어. DPP-180Pro) 자동으로 충치를 형성할 수 있습니다, 사료 캡슐, 밀봉하다, 컷 플레이트, 로트 코드도 인쇄할 수 있습니다. 일반적으로 중간 정도의 고속으로 실행됩니다. (시간당 수천 개의 충치), 대량 생산에 이상적. 다양한 블리스 터 형식 (Alu-PVC 대 Alu-Alu) 제품별로 선택됩니다: 알루-알루 (호일 포일) 가장 높은 장벽을 제공합니다 (수분에 매우 민감한 약물에 가장 적합), 반면 PVC/PVDC는 표준 캡슐에 일반적입니다.. 블리스터 포장은 계수 및 검사도 단순화합니다.: 대부분의 라인은 카메라 검사 또는 정제 계산 기계를 쉽게 통합하여 캡슐 충전을 확인합니다..
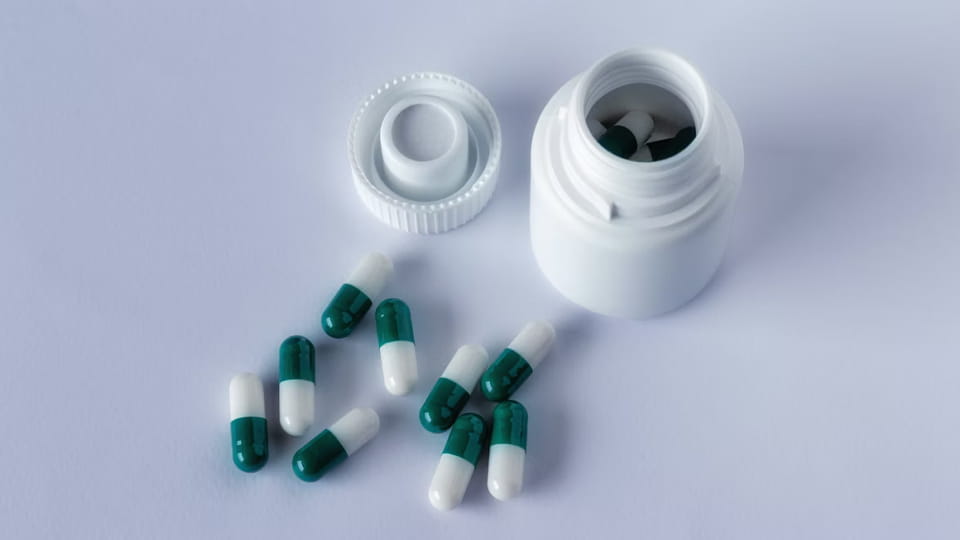
병은 고전적인 다회 용량 용기입니다.. 캡슐은 병에 채워집니다 (플라스틱 또는 유리) 그런 다음 제한됩니다.. 이 형식은 비타민으로 잘 알려져 있습니다., 보충제, 그리고 많은 처방약. 주요 기능:
수치: Jinlu JL-16H 캡슐/정제 병에 넣는 라인 (100 병/분) 언스크램블러와 함께, 막대, 절정, 그리고 라벨러.
캡슐 병입 라인은 여러 기계를 통합합니다.: 에이 병 스크램블러 (또는 피드 호퍼), 에이 계산 충전 기계 (각 병에 X 캡슐을 떨어뜨리는 것입니다.), 에이 캡핑/씰링 기계 (캡을 씌우고 조이세요), 그리고 라벨링 머신. 예를 들어, Jinlu의 JL-16H 라인은 3~40mm 캡슐을 처리할 수 있습니다., 분당 최대 100병 달성 >99.8% 계산 정확도. 병은 매우 빠르게 대량으로 채워질 수 있습니다.. 규정을 충족하려면, 병 라인에는 종종 건조제를 삽입하기 위한 스테이션이 포함되어 있습니다. (예를 들어. 실리카 팩) 변조 방지 봉인 스테이션. 어린이 보호 캡은 소아용 의약품의 또 다른 일반적인 특징입니다..
병 포장을 선택할 때, 제품의 양과 안정성을 고려: 대용량 제품 (예를 들어. 일일 비타민) 정장 병, 반면에 용량이 적거나 매우 민감한 캡슐은 물집을 선호할 수 있습니다.. 참고 병은 일반적으로 제공 하이 배리어 블리스터보다 고유의 수분/산소 보호 능력이 떨어집니다.. 호박색 유리 또는 불투명 용기는 빛 차단 기능을 추가할 수 있습니다., 그리고 내부 씰 (알루미늄 호일 라이너) 장벽을 개선할 수 있다.

향 주머니 (그리고 스틱팩) ~이다 유연한 패킷 1회 분량의 파우더가 들어있는, 과립 또는 액체. 캡슐만큼 흔하지는 않지만, 관련 제형과 관련이 있습니다. (예를 들어. 영양 캡슐을 분말로 쪼개서 만든 것, 또는 분말 충전재 자체). 그래도 여전히, 향 주머니와 스틱 팩 캡슐 포장 가이드에 언급될 가치가 있음:
향낭과 스틱팩 모두 생산됩니다. 수직형 채우기 밀봉 (Vffs) 또는 수평 FFS 기계. Jinlu의 JL-VP 시리즈, 예를 들어, 다중 레인 스틱/봉지 FFS 기계입니다. (40 가방/분/레인, ±0.02g 정확도) 분말용, 과립, 액체 또는 젤. 캡슐 자체를 봉지에 포장하는 경우, 복용량을 회수하기 위해 봉지를 열 것입니다. 그러나 더 일반적으로는, 캡슐은 물집이나 병에 그대로 포장되어 있습니다., 봉지/스틱 팩은 분말 내용물을 취급하는 동안.
규제 사항: 고형제 및 반고형제의 경우 봉지 및 스틱 팩이 허용됩니다. (WHO 정의에 따라). 제품이 직접적으로 포함된 경우 기본 포장으로 간주됩니다.. 물집처럼, 봉지는 어린이에게 안전하도록 설계되거나 눈물 노치가 있을 수 있습니다..
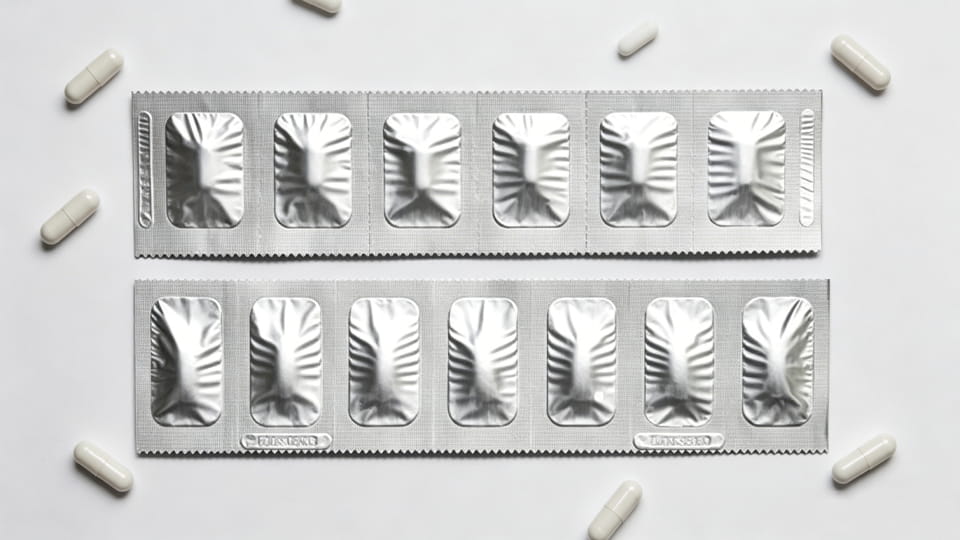
스트립 팩 (호일 스트립 또는 "복용량 밴드"라고도 함) 단위 용량의 1차 팩의 또 다른 형태입니다.. 스트립은 일반적으로 재료의 좁은 웹입니다. (종종 알루미늄 또는 다층 라미네이트) 접어서 밀봉했어요, 복용량 사이에 천공이 있음. 물집과 달리, 스트립 팩은 깊은 구멍을 형성하지 않습니다., 캡슐은 두 겹의 필름 사이에 끼워져 있습니다.. 스트립 팩은 아시아 지역에서 높은 수분 보호가 필요하지만 블리스터의 부피가 너무 큰 제품에 널리 사용됩니다.. 요점:

선택의 관점에서, 스트립 팩은 병과 병 사이의 중간 지점을 차지합니다. (대부분) 물집이 생기고 (엄격). 그들은 종종 다음과 같은 경우에 선택됩니다. 매우 높은 수분 장벽 필요하지만 얇은, 경량 형태. (메모: 스트립 팩은 추가 밀봉을 사용하지 않는 한 블리스터만큼 명확하게 변조 증거를 제공하지 못할 수 있습니다.)
기본 팩을 선택한 후, 캡슐은 일반적으로 2차 포장 판지 상자처럼 또는 수축 포장된 사례. 상자는 배송 중 추가 보호 기능을 제공합니다., 추가 라벨링 허용 (약물 정보, 브랜딩, 변조 방지 씰), 멀티팩 제품 정리를 도와드립니다.. 2차 포장에 대한 주요 고려사항:
요약, 2차 포장은 최종 유통을 위해 제품을 포장하고 모든 라벨링 및 규정 준수 기능이 제대로 갖추어져 있는지 확인합니다.. 여기서 좋은 디자인은 브랜드 이미지와 사용자 편의성을 높여줍니다. (쉽게 열 수 있는 상자, 명확한 라벨링).
캡슐을 보호하려면 올바른 포장재를 선택하는 것이 중요합니다.. 재료는 약물과 호환되어야 합니다., 장벽 요구 사항을 충족, 규정을 준수하고 (ISO 15378 포장재 GMP, 등.). 일반적인 재료는 다음과 같습니다:
일반적으로, 캡슐에 적합한 가장 높은 장벽을 선택하십시오.. 예를 들어, 습기에 민감한 젤라틴 캡슐은 Alu-Alu 물집 또는 건조제가 들어 있는 호박색 유리병을 보증할 수 있습니다.. 대조적으로, 민감하지 않은 캡슐은 표준 PVC 블리스 터 또는 HDPE 병을 사용할 수 있습니다.. 유통기한도 고려하세요: 장기적인 제품에는 더 강력한 장벽이 필요합니다.
안전/재활용 참고 사항: 지속 가능한 추세는 단일 소재 팩을 선호합니다. (예를 들어. PP 물집, 재활용 가능한 페트병). 추출물용 시험물질, 호환성, FDA/EU 기준 충족 (예를 들어. USP <661> 가소제용) 포장 규정 준수의 일부입니다..
캡슐은 다양한 유형으로 제공되며 그 구성에 따라 포장 요구 사항이 결정됩니다.:
요컨대, 그만큼 캡슐 껍질 재료 및 충전 유형 포장 선택에 영향을 미칠 것입니다. 수분에 매우 민감한 제형은 항상 가장 단단한 장벽을 선호합니다. (Alu-Alu 물집, 호일 향 주머니, 또는 건조제가 담긴 밀봉된 병). 덜 민감한 제제에는 더 많은 옵션이 있습니다. 항상 안정성을 테스트하십시오. 최종 포장 캡슐 (ICH Q1A에서는 제안된 용기의 안정성을 요구합니다.).
최적의 캡슐 포장을 선택하려면 여러 요소를 평가해야 합니다.: 제형, 환자 준수, 제품 민감도, 유통기한, 규제 요구 사항, 그리고 비용. 다음은 단순화된 의사결정 프레임워크입니다.:
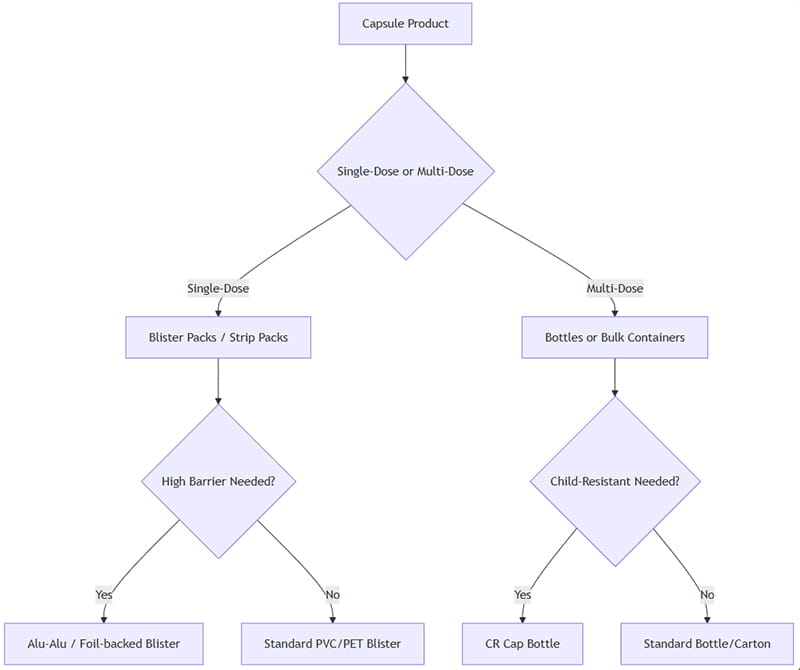
이것을 적용하는 방법: 캡슐이 단일 단위로 의도된 경우 (예를 들어. 꽉 투여하거나 소매용 블리스 터 카드), 물집/스트립이 선호됩니다.. 다회 투여용 (예를 들어. 보충제 또는 다용도 약물), 병이 더 효율적이다. 습기/빛 차단이 중요한 경우, 높은 장벽 옵션을 선택하세요 (알루-알루, 호박색 병). 소아의 안전이 필요한 경우, 어린이에게 안전한 병 마개 또는 변조 방지 블리스 터 디자인을 사용하십시오..
업계의 다른 기준은 다음과 같습니다.:
궁극적으로, 파일럿 테스트 (안정성 연구, 소비자 연구) 총 비용 분석을 통해 최종 결정을 내릴 수 있습니다.. 위의 순서도는 일반적인 시나리오의 시작점입니다..
캡슐 포장에는 특수 기계가 필요합니다.. 캡슐을 가득 채운 후 (캡슐 필러로), 포장라인으로 이송됩니다.:
장비 규정 준수: 모든 의약품 포장 기계는 GMP 설계를 충족해야 합니다. (스테인레스 스틸, 청소 가능한 표면, 검증된 성능). Jinlu의 기계는 CE/cGMP 인증을 받았습니다, 사양을 벗어난 알약에 대한 구성 가능한 보호 및 거부 시스템이 있는 경우가 많습니다.. 통합이 핵심이다: 예를 들어, 블리스 터 기계는 상자를 세우고 각 블리스 터 카드를 포장하는 상자에 직접 공급할 수 있습니다.. 종합 시스템 (충전부터 최종 상자까지) 캡슐 포장의 엔드 투 엔드 자동화 가능.
의약품 캡슐 포장은 엄격한 규정을 준수해야 합니다.. 핵심 사항은 다음과 같습니다:
전반적인, FDA 회의, EMA 및 WHO 지침은 필수적입니다.. 많은 포장업체는 규제 대상 공급업체와 협력합니다. (진루처럼) 검증된 기계와 문서를 제공할 수 있는 사람 (IR/WH/PQ).
올바른 캡슐 포장을 선택하려면 균형 잡힌 보호가 필요합니다., 유용성, 그리고 비용. 물집 팩, 병, 향 주머니, 스트립은 각각 그 자리를 차지합니다. 의약품 포장. 수분 민감도와 같은 요인, 복용량 형태, 환자의 요구에 따라 결정을 내립니다.. 나란히, 엄격한 규제 및 GMP 요구 사항을 충족해야 합니다..
~에 진루 포장, 우리는 엔드 투 엔드 의약품 포장 솔루션을 전문으로 합니다.. 우리 장비 – 고속 물집 기계 에게 완전 자동 계산 & 병입 라인 – 캡슐을 효율적이고 규정에 맞게 포장하는 데 도움이 됩니다.. 우리는 또한 지속 가능한 선택을 지지합니다 (예를 들어. 새로운 재활용 필름을 사용하는 기계) 원스톱 서비스를 제공합니다. (사용자 정의, 확인). 우리 팀은 귀하의 제품에 가장 적합한 캡슐 포장 시스템을 평가하는 데 도움을 드릴 수 있습니다..
캡슐 포장을 최적화할 준비가 되었습니다.? 지금 Jinlu Packing에 문의하세요. 전문가의 안내와 견적을 원하시면. 당사의 솔루션은 글로벌 품질 표준 이상의 지원을 받습니다. 3,000 전세계 고객 만족. 성공을 위해 캡슐을 포장해 보세요!
캡슐 포장은 의약품 캡슐을 보호재에 넣어 안정성을 유지하는 공정을 말합니다., 안전, 및 규정 준수. 일반적인 형식에는 블리스 터 팩이 포함됩니다., 병, 그리고 스트립 포장.
제약 실습에서, 포장은 단순한 봉쇄가 아니라 습기로부터 캡슐을 보호합니다., 산소, 그리고 오염, 의약품의 품질과 유통기한에 직접적인 영향을 미치는.
하나의 "최고" 옵션은 없습니다. 제품과 시장에 따라 다릅니다..
• 블리스터 포장 → 습기에 민감하거나 고가의 의약품에 적합
• 병 포장 → 대량 보관 및 보충제에 가장 적합
• 스트립 포장 → 비용에 민감한 시장에서 일반적임
블리스터 포장은 단위 용량 보호와 더 나은 환경 격리를 제공하기 때문에 선호되는 경우가 많습니다..
주요 위협을 차단하는 소재 사용 (수분, 산소, 빛). 예를 들어, Alu-Alu 호일 블리스터 또는 호박색 유리병은 습기에 민감한 캡슐에 탁월한 차단 기능을 제공합니다.. 안정적인 제품의 경우 PVDC 코팅이 된 PVC 또는 PET 블리스터가 일반적이고 저렴합니다.. HDPE 병은 표준이지만 캡슐이 민감한 경우 건조제 파우치가 필요할 수 있습니다.. 수증기 투과율 평가 (WVTR) 영화의. 지속가능성을 고려한 생산에서, 재활용 가능한 단일 물질을 고려하십시오 (예를 들어. PP 물집). 항상 안정성 연구를 통해 포장 호환성을 테스트하십시오..
캡슐은 습도 등 환경적 요인에 민감합니다., 산소, 그리고 빛.
적절한 포장:
• 수분흡수 방지
• 산화 감소
• 화학적 안정성을 유지합니다.
물집 포장, 특히, 각 캡슐에 대해 밀봉된 미세 환경을 생성합니다., 유통기한 대폭 연장.
형식에 따라 다릅니다. 물집의 경우, 블리스 터 성형 / 밀봉 기계가 필요합니다 (캡슐에 맞는 캐비티 몰드 포함) 그리고 아마도 판지 상자. 병용, 병 채우기/캡핑 라인이 필요합니다. (스크램블러, 카운터필러, 절정, 라벨러). 스틱팩의 경우, 수직형 충전 밀봉 봉지 기계를 사용하세요. Jinlu는 이 모든 것을 제공합니다: 예를 들어. DPP 시리즈 물집, JL-16H 계수 라인, JL-VP 향낭 포장기. 올바른 기계를 선택하려면 용량이 필요합니다, 캡슐 크기, 및 필수 통합 (비전 시스템, 거부 스테이션, 등.).
복용량과 같은 요소를 고려하십시오. (싱글 대. 다수의), 안정성 요구, 그리고 환자 사용. 1회 복용의 편리함과 높은 보호력을 위해 (특히 습기/빛에 민감한 캡슐의 경우), 물집이 선호되는 경우가 많습니다.. 대용량 또는 경제적인 이유로, 병은 일반적으로 선택됩니다. 또한, 어린이에게 안전하거나 노인 친화적인 기능이 필요한 경우, 병은 더 많은 옵션을 제공할 수 있습니다. 원칙적으로, 캡슐의 특성을 포장과 일치시키세요: 예를 들어. 흡습성이 매우 높은 캡슐 → Alu-Alu 물집; 수요가 많은 보충제 → 병.
참고자료:
1.신관 9 의약품 포장에 관한 지침 -- WHO
2.의약품 및 건강 제품에 대한 기술 요구 사항 -- 세계보건기구
3.의약품 포장재 및 의약품 안전: 미니 리뷰 —— MDPI
4.약국 포장 및 삽입물 —— 국립 의학 도서관
5.의약품 블리스 터의 특성화 및 재활용 가능성에 대한 검토 —— 사이언스다이렉트

사소한 푸, 진루패킹 창업자, 가져오다 20 제약 기계 부문에 대한 다년간의 전문 지식. 그의 리더십 아래, Jinlu는 디자인을 통합하는 신뢰할 수 있는 공급업체로 성장했습니다., 생산, 그리고 판매. Petty는 고객이 제약 포장의 복잡성을 헤쳐나갈 수 있도록 깊은 산업 지식을 공유하는 데 열정을 갖고 있습니다., 장비뿐만 아니라 제품도 받을 수 있도록 보장, 생산 목표에 맞춘 진정한 원스톱 서비스 파트너십.



