
El empaque de cápsulas se refiere a cómo las cápsulas farmacéuticas (cápsula dura o cápsula blanda) están cerrados para su distribución y uso.. incluye embalaje primario (el recipiente inmediato en contacto con las cápsulas, p.ej. cavidades de ampolla o botellas) y embalaje secundario (cajas exteriores, cajas, y etiquetas que protegen e identifican aún más el producto). Un buen empaque de cápsulas debe proteger el medicamento de la humedad., luz, y contaminación, garantizar la estabilidad durante la vida útil, y cumplir con los requisitos reglamentarios. Por ejemplo, La OMS señala que “Los componentes del embalaje rodean el producto desde su producción hasta su uso.," y debe cumplir funciones como protección, estabilidad, e identificación. Regulaciones GMP de la FDA (21 CFR 211) Requerir procedimientos escritos para inspeccionar y probar los materiales de embalaje al recibirlos., y exigir etiquetado seguro y controles de manipulación. En la práctica, debemos igualar el producto en cápsula (forma de dosificación, sensibilidad, dosificación, etc.) al formato y material de embalaje adecuados.

El embalaje primario de las cápsulas farmacéuticas es fundamental para protección del producto, cumplimiento, y seguridad del paciente: Mantiene las cápsulas secas., no contaminado, y claramente etiquetado. Por ejemplo, Se sabe que los blísteres y los frascos de color ámbar son ideales para los medicamentos sensibles a la humedad.. El embalaje adecuado también contribuye al cumplimiento del paciente (dosificación en blister de dosis unitarias, biberones a prueba de niños, etiquetado claro) y necesidades de la cadena de suministro (publicación por entregas, logística). A continuación repasamos cada opción en detalle.
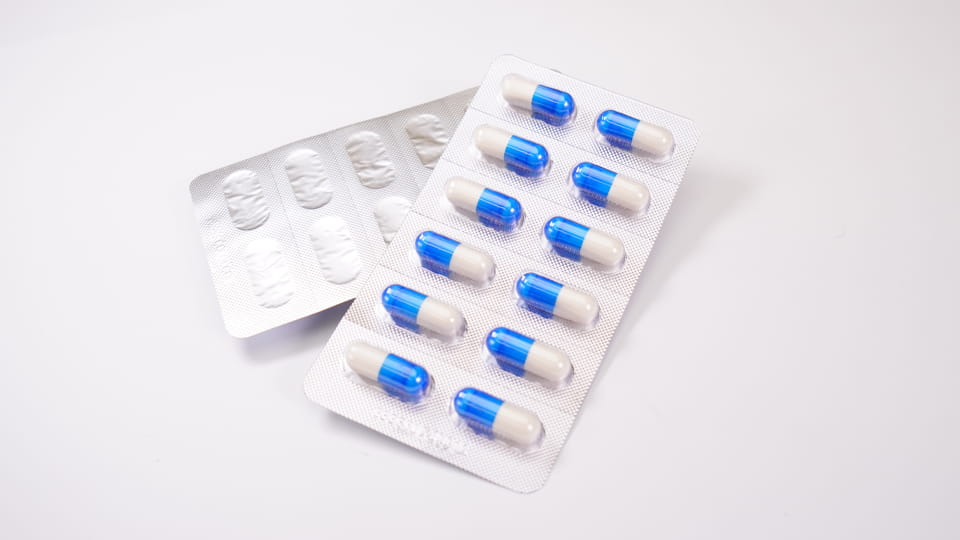
Paquetes de ampollas consisten en cavidades de plástico preformadas (“ampollas”) que contienen cápsulas individuales, sellado con un respaldo (papel de aluminio o plástico). Este embalaje primario ofertas protección de dosis unitaria. Las ampollas proporcionan excelente barrera propiedades: se pueden hacer con Materiales como PVC/PVDC o lámina. (Aluminio-PVC, Alu-Alu) para bloquear la humedad, oxígeno y luz. Por ejemplo, La máquina blister DPP-180Pro de Jinlu puede formar y sellar hasta 4,800 ampollas de cápsulas por hora en laminados Alu-PVC o Alu-Alu. Las características clave del envase tipo blíster de cápsulas incluyen:
Cifra:Máquina envasadora de blister Jinlu JL-180Pro que forma ampollas de cápsulas (Aluminio-PVC), 4,800 caries/hora.
Las máquinas blister de Jinlu (p.ej. el DPP-180Pro) puede formar cavidades automáticamente, cápsulas de alimentación, sello, cortar placas, e incluso imprimir códigos de lote. Suelen correr a velocidad media-alta. (miles de cavidades por hora), Ideal para la producción en masa. Diferentes formatos de blister (Aluminio-PVC vs Aluminio-Alu) son seleccionados por producto: Alu-Alu (papel de aluminio) ofrece la barrera más alta (Lo mejor para medicamentos muy sensibles a la humedad.), mientras que PVC/PVDC es común para las cápsulas estándar. El envasado en blister también simplifica el recuento y la inspección.: la mayoría de las líneas integran fácilmente máquinas de inspección por cámara o de conteo de tabletas para verificar el llenado de las cápsulas..
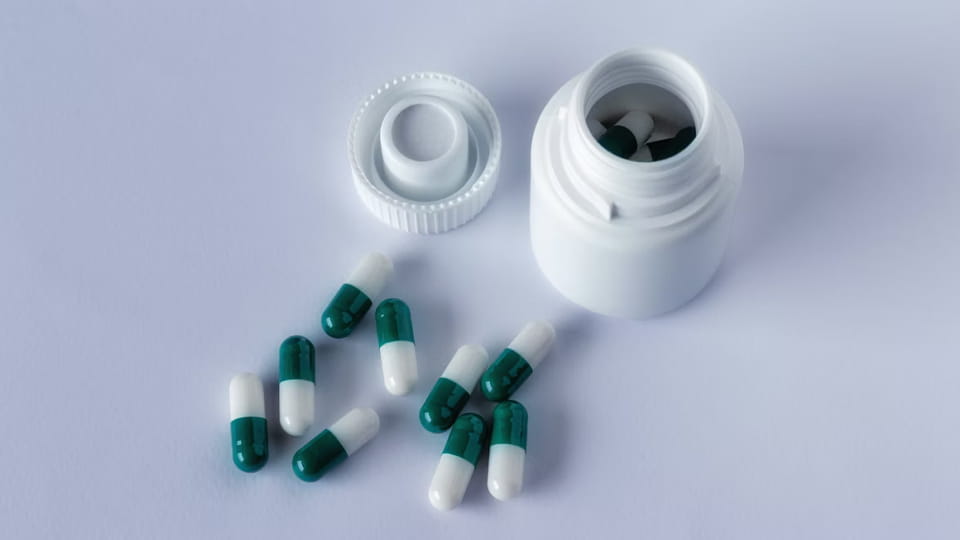
Los biberones son el clásico envase multidosis. Las cápsulas se envasan en botellas. (plástico o vidrio) que luego se tapan. Este formato es muy conocido por las vitaminas., suplementos, y muchas pastillas recetadas. Características clave:
Cifra: Línea de embotellado de cápsulas/tabletas Jinlu JL-16H (100 botellas/min) con descifrador, vara, taponadora, y etiquetadora.
Las líneas de embotellado de cápsulas integran múltiples máquinas: a posicionador de botellas (o tolva de alimentación), a máquina contadora-llenadora (que deja caer X cápsulas en cada botella), a máquina taponadora/selladora (aplicar y apretar las tapas), y un máquina etiquetadora. Por ejemplo, La línea JL-16H de Jinlu puede manejar cápsulas de 3 a 40 mm, logrando ~100 botellas/min con >99.8% Contabilidad de precisión. Las botellas se pueden llenar a granel muy rápidamente. Para cumplir con las regulaciones, Las líneas de botellas a menudo incluyen una estación para insertar desecantes. (p.ej. paquetes de sílice) y una estación de sello a prueba de manipulaciones. Los tapones a prueba de niños son otra característica común de los medicamentos pediátricos.
Al elegir el embalaje de las botellas, considerar el volumen y la estabilidad del producto: productos de gran volumen (p.ej. vitaminas diarias) botellas de traje, mientras que las cápsulas de bajo volumen o muy sensibles pueden preferir ampollas. Las botellas de notas generalmente ofrecen Menos protección inherente contra la humedad y el oxígeno que las ampollas de alta barrera.. Los recipientes de vidrio ámbar o opacos pueden agregar protección contra la luz., y sellos internos (revestimientos de papel de aluminio) puede mejorar la barrera.

Bolsitas (y paquetes de palos) son paquetes flexibles que contienen una dosis de polvo, gránulos o líquido. Si bien no es tan común para las cápsulas, son relevantes para formas farmacéuticas relacionadas (p.ej. cápsulas nutracéuticas rotas en polvo, o relleno en polvo en sí). Sin embargo, bolsitas y paquetes de barras merecen una mención en una guía de envasado de cápsulas:
Tanto los sobres como los stick packs son producidos por forma-llenado-sellado vertical (Vffs) o máquinas FFS horizontales. Serie JL-VP de Jinlu, por ejemplo, es una máquina FFS de barras/bolsitas de varios carriles (40 bolsas/min/carril, Precisión de ±0,02 g) para polvos, gránulos, líquidos o geles. Si las cápsulas se envasaran en bolsitas, uno abriría el sobre para recuperar la dosis, pero más comúnmente, Las cápsulas se envasan intactas en blister o frasco., mientras que los sobres/paquetes en barra contienen contenido en polvo.
Nota regulatoria: Se permiten sobres y paquetes tipo barra para medicamentos sólidos y semisólidos. (según las definiciones de la OMS). Se consideran envases primarios si contienen directamente el producto.. como ampollas, Los sobres pueden diseñarse a prueba de niños o con muescas para rasgar..
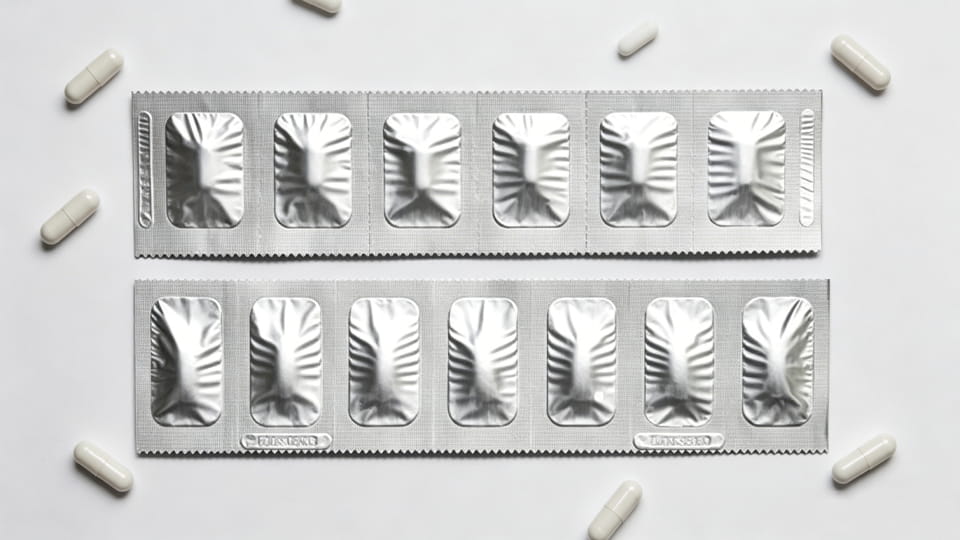
paquetes de tiras (también llamadas tiras de aluminio o “bandas de dosis”) son otra forma de envase primario de dosis unitaria. Una tira suele ser una red estrecha de material. (a menudo aluminio o laminado multicapa) doblado y sellado, con perforaciones entre dosis. A diferencia de las ampollas, Los paquetes de tiras no forman cavidades profundas, sino que, la cápsula está intercalada entre dos capas de película. Los envases en tiras se utilizan ampliamente en Asia y para productos donde se necesita una alta protección contra la humedad pero las ampollas son demasiado voluminosas.. Puntos clave:

Desde el punto de vista de la selección, Los paquetes de tiras ocupan un punto medio entre los de botella. (a granel) y ampolla (rigidez). A menudo son elegidos cuando barrera de humedad muy alta es necesario pero en una delgada, forma ligera. (Nota: Es posible que los paquetes de tiras no proporcionen pruebas de manipulación tan claramente como los blísteres, a menos que se utilicen sellos adicionales.)
Después de elegir un paquete principal, Las cápsulas generalmente se colocan en embalaje secundario como cajas de cartón o envuelto en plástico retráctil casos. Las cajas brindan protección adicional durante el envío, permitir etiquetado adicional (información de drogas, herrada, sellos a prueba de manipulaciones), y ayudar a organizar productos de paquetes múltiples. Consideraciones clave para el embalaje secundario:
En resumen, El embalaje secundario envuelve el producto para su distribución final y garantiza que todas las características de etiquetado y cumplimiento estén en su lugar.. Un buen diseño aquí mejora la imagen de marca y la comodidad del usuario. (cajas de fácil apertura, etiquetado claro).
Elegir el material de embalaje adecuado es fundamental para proteger las cápsulas.. Los materiales deben ser compatibles con el medicamento., satisfacer las necesidades de barrera, y cumplir con las regulaciones (ISO 15378 GMP para materiales de embalaje, etc.). Los materiales comunes incluyen:
Generalmente, elija la barrera más alta que tenga sentido para su cápsula. Por ejemplo, una cápsula de gelatina sensible a la humedad puede requerir un blister Alu-Alu o una botella de vidrio ámbar con desecante. En contraste, Las cápsulas no sensibles pueden utilizar un blister de PVC estándar o una botella de HDPE.. Considere también la vida útil: Los productos a más largo plazo necesitan una barrera más sólida..
Notas de seguridad/reciclaje: Las tendencias sostenibles favorecen los envases monomateriales (p.ej. ampollas de polipropileno, botellas de PET reciclables). Materiales de prueba para extraíbles., compatibilidad, y cumplir con los límites de la FDA/UE (p.ej. USP <661> para plastificantes) es parte del cumplimiento del embalaje.
Las cápsulas vienen en varios tipos y su formulación dicta las necesidades de envasado.:
En breve, el Material de la cubierta de la cápsula y tipo de relleno. afectará la elección del embalaje. Las formulaciones altamente sensibles a la humedad siempre favorecen la barrera más estrecha. (Blíster Alu-Alu, bolsita de aluminio, o botella sellada con desecante). Las formulaciones menos sensibles tienen más opciones. Siempre pruebe la estabilidad del cápsula empaquetada final (ICH Q1A requiere estabilidad en el contenedor propuesto).
Seleccionar un envase de cápsulas óptimo implica evaluar múltiples factores: forma de dosificación, cumplimiento del paciente, sensibilidad del producto, duración, requisitos reglamentarios, y costo. A continuación se muestra un marco de decisión simplificado.:
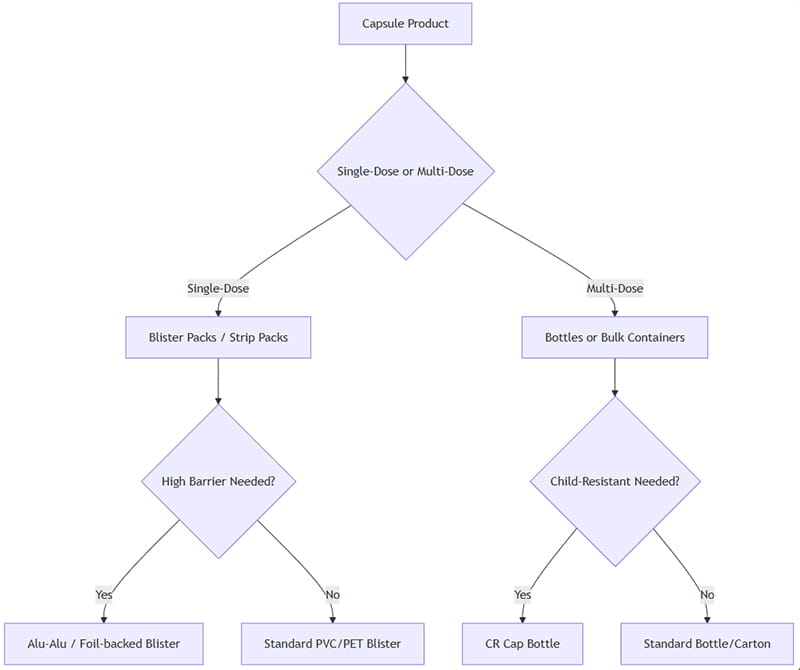
Cómo aplicar esto: Si las cápsulas están pensadas como unidades individuales (p.ej. dosis ajustadas o tarjetas blíster al por menor), Se prefieren ampollas/tiras.. Para multidosis (p.ej. suplementos o medicamentos de alto uso), las botellas son más eficientes. Si la protección contra la humedad y la luz es crítica, seleccionar opciones de alta barrera (Alu-Alu, botella ámbar). Si se necesita seguridad pediátrica, Utilice cierres de botellas a prueba de niños o diseños de blíster a prueba de manipulaciones..
Otros criterios de la industria incluyen:
Al final, prueba piloto (estudios de estabilidad, estudios de consumo) y el análisis del costo total guiará la decisión final. El diagrama de flujo anterior es un punto de partida para escenarios típicos..
El envasado de cápsulas requiere maquinaria especializada. Después de llenar las cápsulas (por rellenos de cápsulas), estos se transfieren a las líneas de envasado:
Cumplimiento del equipo: Todas las máquinas de envasado para productos farmacéuticos deben cumplir con el diseño GMP (acero inoxidable, superficies limpiables, rendimiento validado). Las máquinas de Jinlu cuentan con la certificación CE/cGMP, a menudo con protecciones configurables y sistemas de rechazo para píldoras fuera de especificaciones. La integración es clave: Por ejemplo, una máquina de blister puede alimentar directamente una estuchadora que forma las cajas y envasa cada tarjeta de blister. Sistemas integrales (desde el llenado hasta la caja final) Permitir la automatización de extremo a extremo del envasado de cápsulas..
Los envases de cápsulas para productos farmacéuticos deben cumplir normas estrictas. Los puntos clave incluyen:
En general, reunión de la FDA, Las directrices de la EMA y la OMS son esenciales. Muchos envasadores trabajan con proveedores regulados. (como Jinlu) ¿Quién puede proporcionar máquinas validadas y documentación? (IR/WH/PQ).
Elegir el envase de cápsulas adecuado requiere una protección equilibrada, usabilidad, y costo. Paquetes de ampollas, botellas, bolsitas, y las tiras cada una tiene su lugar en embalaje farmacéutico. Factores como la sensibilidad a la humedad., forma de dosis, y las necesidades del paciente guían la decisión. Junto a, Se deben cumplir estrictos requisitos regulatorios y GMP..
En Embalaje de Jinlu, Nos especializamos en soluciones integrales de embalaje farmacéutico.. Nuestro equipo – desde máquinas blister de alta velocidad a conteo totalmente automatizado & líneas de embotellado – le ayuda a envasar cápsulas de manera eficiente y conforme. También apoyamos las opciones sostenibles (p.ej. máquinas que procesan nuevas películas reciclables) y ofrecer un servicio integral (personalización, validación). Nuestro equipo puede ayudarle a evaluar el mejor sistema de envasado de cápsulas para su producto..
Listo para optimizar el envasado de sus cápsulas? Póngase en contacto con Jinlu Packing hoy para orientación experta y cotizaciones. Nuestras soluciones están respaldadas por estándares de calidad globales y más 3,000 clientes satisfechos en todo el mundo. Empaquemos sus cápsulas para el éxito!
El envasado de cápsulas se refiere al proceso de encerrar cápsulas farmacéuticas en materiales protectores para mantener la estabilidad., seguridad, y cumplimiento. Los formatos comunes incluyen blister., botellas, y embalaje en tiras.
En la práctica farmacéutica, El embalaje no es sólo contención: protege las cápsulas de la humedad., oxígeno, y contaminación, que afecta directamente la calidad y la vida útil del medicamento.
No existe una única "mejor" opción: depende de su producto y mercado..
• Embalaje tipo blíster → mejor para medicamentos sensibles a la humedad o de alto valor
• Envasado en botella → mejor para almacenamiento a granel y suplementos
• Embalaje en tiras → común en mercados sensibles a los costos
A menudo se prefiere el envasado en blister porque proporciona protección en dosis unitarias y un mejor aislamiento ambiental..
Utilice materiales que bloqueen las amenazas clave. (humedad, oxígeno, luz). Por ejemplo, Los blísteres de aluminio Alu-Alu o los frascos de vidrio ámbar ofrecen una excelente barrera para las cápsulas sensibles a la humedad.. Los blísteres de PVC o PET con revestimiento de PVDC son habituales y más económicos para productos estables.. Los frascos de HDPE son estándar, pero es posible que necesiten una bolsa desecante si las cápsulas son sensibles.. Evaluar las tasas de transmisión de vapor de agua. (WVTR) de peliculas. En una producción consciente de la sostenibilidad, considerar monomateriales reciclables (p.ej. ampollas de polipropileno). Pruebe siempre la compatibilidad del embalaje mediante estudios de estabilidad.
Las cápsulas son sensibles a factores ambientales como la humedad., oxígeno, y luz.
Embalaje adecuado:
• Previene la absorción de humedad
• Reduce la oxidación
• Mantiene la estabilidad química
Embalaje, En particular, Crea microambientes sellados para cada cápsula., extendiendo significativamente la vida útil.
Depende del formato. Para ampollas, Necesitas una máquina formadora/selladora de blister (con moldes de cavidad para adaptarse a su cápsula) más posiblemente un estuchador. para botellas, necesitas una línea de llenado/tapado de botellas (descifrador, contra-relleno, taponadora, etiquetadora). Para paquetes de barras, Utilice una máquina vertical para formar, llenar y sellar bolsitas.. Jinlu ofrece todo esto: p.ej. nuestras ampollas serie DPP, Línea de conteo JL-16H, y empacadora de bolsitas JL-VP. Seleccionar la máquina adecuada implica capacidad, tamaño de la cápsula, e integraciones requeridas (sistemas de visión, estaciones de rechazo, etc.).
Considere factores como la dosis (soltero vs.. múltiple), necesidades de estabilidad, y uso del paciente. Para comodidad de dosis única y alta protección (especialmente para cápsulas sensibles a la humedad/luz), A menudo se prefieren las ampollas.. Por motivos económicos o de gran volumen, Generalmente se eligen botellas.. También, si se necesitan características a prueba de niños o aptas para personas mayores, las botellas pueden ofrecer más opciones. Por regla general, Haga coincidir las propiedades de su cápsula con el empaque.: p.ej. cápsulas muy higroscópicas → blister Alu-Alu; suplementos de alta demanda → botellas.
Referencias:
1.Anexo 9 Directrices sobre envases para productos farmacéuticos. -- OMS
2.Requisitos Técnicos para Medicamentos y Productos Sanitarios -- Organización Mundial de la Salud
3.Materiales de embalaje farmacéutico y seguridad de los medicamentos: Una mini revisión —— MDPI
4.Envases y prospectos de farmacia —— Biblioteca Nacional de Medicina
5.Una revisión sobre la caracterización y reciclabilidad de los blisters farmacéuticos. —— Ciencia Directa

Petty Fu, Fundador de Jinlupacking, trae 20 años de experiencia al sector de maquinaria farmacéutica. Bajo su liderazgo, Jinlu se ha convertido en un proveedor confiable que integra diseño, producción, y ventas. A Petty le apasiona compartir su profundo conocimiento de la industria para ayudar a los clientes a navegar las complejidades del empaque farmacéutico., garantizar que reciban no sólo equipos, sino una verdadera asociación de servicio integral adaptada a sus objetivos de producción..