
В фармацевтической промышленности, проверка упаковки is a critical subset of валидация процесса focused on ensuring that packaging equipment and processes consistently protect drug product quality. It involves a documented program of qualification (Установка, Operational, Производительность) and testing (например. container-closure integrity, стабильность, транспорт) to prove that packaging systems perform as intended under GMP conditions. Effective packaging validation safeguards patient safety (by preventing contamination, degradation or mix-ups) and ensures regulatory compliance (FDA, ЕМА, ВОЗ, ICH, ИСО). This guide explains the definition, шаги, тесты, стандарты, и лучшие практики for pharmaceutical packaging validation, and shows how automated equipment (нравиться Jinlu Packing’s machines) is designed to meet these stringent requirements.

Packaging validation in pharma means demonstrating with documented evidence that the packaging process (equipment and materials) reliably produces a package that protects the drug product. На практике, it applies the same rigorous validation concepts used in manufacturing (ИК/Белый/PQ) к начальный, вторичный, and tertiary packaging operations. According to WHO’s GMP guidance, “Packaging processes and equipment need validation/qualification in the same way as any other part of processing within a pharmaceutical facility.”. Другими словами, packaging validation ensures the container-closure system, маркировка, and packaging lines consistently meet quality specifications and regulatory requirements. The goal is to maintain drug strength, purity and stability through packaging, distribution and shelf life, while preventing mix-ups, counterfeits, или повреждение.
Typical deliverables in packaging validation include a Спецификация требований пользователя (УРС) (the design & quality criteria), Installation/Operational/Performance Qualification protocols, and testing records. The validation process covers:
Each level must protect the product. Например, the Australian TGA defines “primary packaging” (the container that immediately covers the goods) and distinguishes вторичная упаковка as the outer box or wrapper. В конечном счете, packaging validation ties back to GMP – ensuring that containers and closures do not alter the drug и “provide adequate protection against external factors” during storage and use.
Pharmaceutical packaging is the final defense for drug quality. Validating packaging processes is vital because:
Packaging validation addresses all packaging stages. Обычно, we distinguish:
Each level requires risk assessment and appropriate testing: например, начальный packaging might involve USP <1207>-style integrity tests, пока вторичный packaging tests might include carton strength and label adhesion, и высшее packaging uses ISTA/ASTM test protocols. All must be documented in the validation plan.
Packaging validation typically follows the equipment/process qualification life cycle, often described as DQ/IQ/OQ/PQ. These stages ensure a new or modified packaging line meets requirements at each stage.
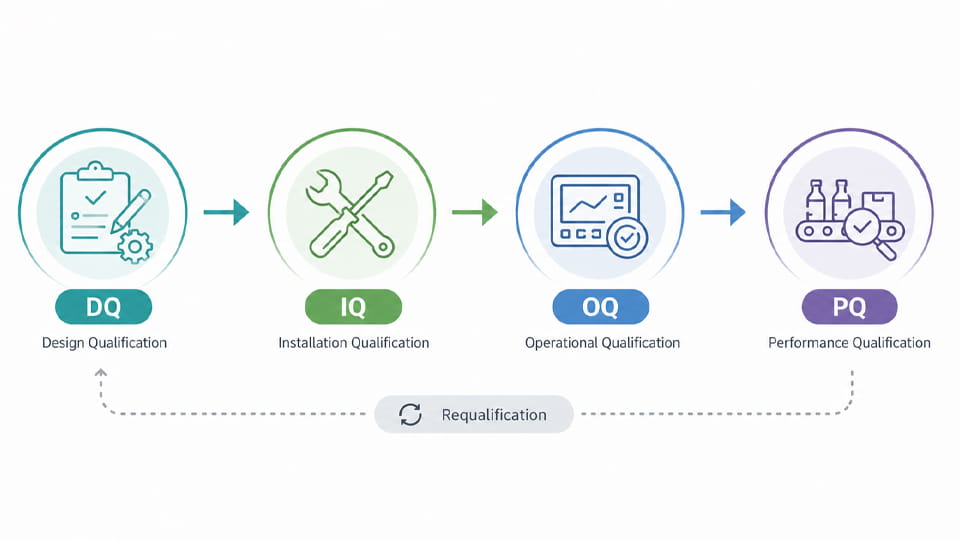
The table below summarizes the DQ–IQ–OQ–PQ stages:
| Этап | Цель | Key Documents | Example Activities/Tests |
| Квалификация дизайнера (ДК) | Verify the packaging system design meets URS/GMP | УРС (requirements spec), design specs (FDS/HDS/SDS), risk assessment | Design review, material compatibility checks, DFMEA |
| Квалификация установки (IQ) | Confirm proper installation of equipment | IQ protocol/checklist, installation log, сертификаты калибровки | Verify utilities, equipment assembly, калибровка |
| Операционная квалификация (ОК) | Verify equipment functions within defined limits | OQ protocol, functional test plans, Сов | Test parameters (скорость, температура, давление); seal checks |
| Квалификация производительности (ПК) | Ensure consistent quality during production runs | PQ protocol, batch records, release criteria | Production runs; sample testing (CCIT, наполняющий вес, визуальный осмотр) |
![]()
As the diagram shows, packaging validation is a life-cycle process from design through performance. Note that requalification may be needed after major changes or periodically.
Several specialized tests are commonly performed during packaging validation:
CCIT is essential for sterile and even non-sterile products. It assesses whether the container closure system (например. vial+stopper+cap, ampoule, blister seal) forms a perfect barrier. Methods include vacuum decay, pressure decay, проникновение красителя, or microbial challenge. As one industry source explains, “Container Closure Integrity Testing (CCIT) is an assay that evaluates the adequacy of container closure systems to maintain a sterile barrier against potential contaminants.”. Regulatory documents (USP <1207>, FDA, EMA Annex 1) emphasize testing closure integrity to ensure no leaks or breaches. Например, USP <1207> provides guidelines on selecting leak test methods to certify sealed packages.
Упаковочные материалы (пластмассы, rubbers, inks, adhesives) must not adversely interact with the drug product. Extractables and leachables studies simulate long-term contact: extractables are chemicals driven out of packaging under harsh conditions, and leachables are those that actually migrate into the product under normal conditions. These tests ensure the packaging does not introduce toxic or stability-affecting impurities. Pharmacopeial and FDA guidance (например. USP <661>, Q3E) outline E/L testing for container closure systems. На практике, one checks that the polymer, покрытия, and label materials meet pharmacopeial standards (например. USP glass type, ISO rubber closure specs). The WHO notes that pharmacopoeial standards for closures (like rubber stoppers) являются “minimum requirements” and emphasizes stability studies to prove suitability.
As per ICH Q1A, stability studies must be done on the drug product in its final packaging configuration. During validation, packaging’s impact on stability is assessed: например, the final packaging is subjected to accelerated aging (high temp/humidity) and real-time conditions to ensure no degradation (например. moisture ingress in blister packs, oxygen permeation in bottles). Stability-indicating assays (chemical and microbial) are performed. Packaging that fails to protect the product under stress invalidates shelf-life claims. Таким образом, validation includes confirming that the chosen packaging meets shelf-life requirements (this overlaps with regulatory stability protocols).
Tertiary packaging tests simulate actual shipping conditions. Standard tests include drop height tests, вибрация (truck/rail simulation), сжатие (stacking weight), and temperature cycling (especially for cold chain). Например, ASTM D4169 (for packaged products) or ISTA protocols may be used. A robust packaging validation plan verifies that cartons, cases and crates protect primary packages during handling: verifying that no breakage, label peeling, or moisture damage occurs under distribution conditions. For cold chain, temperature mapping and validated insulated shippers are tested to maintain required temperature ranges throughout transit.

Pharmaceutical packaging validation must meet multiple regulatory expectations:
В итоге, any packaging line must comply with the national pharmacopeia and regional GMP standards. Compliance checklists typically cite FDA, GMP ЕС, WHO and relevant ISO standards, and often call for fully documented SOPs and validation master plans covering packaging.
Packaging validation poses unique challenges compared to manufacturing processes:
Несмотря на эти проблемы, the underlying principle remains: treat packaging like any other critical process, with thorough qualification and robust QC checks. As the ISPE paper concludes, проверка упаковки “differ(с) little from the validation of processes used for drug manufacture.”.
To overcome challenges and ensure a successful validation, следуйте этим рекомендациям:
By embedding validation thinking at every stage – from design to daily operation – manufacturers can ensure packaging lines deliver products that meet quality and compliance expectations.
The choice and design of packaging equipment heavily influence validation success. High-quality packaging machines are engineered to meet GMP requirements and support easy qualification. Например, Джинлу упаковывает automated machines are built with features that streamline validation:
На практике, a validated packaging line might include: a Jinlu блистерная упаковочная машина linked to a картонажная машина и counting/filling lines. Each equipment must be qualified (ИК/Белый/PQ). Jinlu’s systems are designed to work seamlessly together (например. “highly automated blister cartoning line” до 320 коробок/мин), reducing integration issues during validation. We also offer case studies (например. a complete линия подсчета и розлива, or an automatic cartoning & labeling line) that demonstrate end-to-end validated solutions.

By partnering with Jinlu or similar manufacturers, companies benefit from equipment that is “cGMP compliant”, supports a full validation lifecycle, and comes with after-sales support (ввод в эксплуатацию, обучение) to ensure the line remains validated.
Packaging validation is essential for pharmaceutical quality and compliance. By treating packaging operations as rigorously as core manufacturing steps, companies ensure their products reach patients safely. The process spans definition (УРС, risk assessment), квалификация (DQ/IQ/OQ/PQ), тестирование (CCIT, стабильность, транспорт), and continuous monitoring. It is governed by GMP regulations (FDA, ЕМА, ВОЗ) and industry standards.
Modern automated packaging equipment – such as Jinlu Packing’s machines – play a key role in validation. Designed for precision, hygiene and electronic traceability, they help manufacturers meet validation criteria efficiently. When selecting packaging lines, consider suppliers who provide complete validation documentation and support.
В конечном счете, thorough packaging validation is “the final safeguard for product quality and patient safety”, aligning with both regulatory demands and business goals.
Ready to validate your packaging line? Contact Jinlu Packing’s experts to discuss GMP-compliant packaging solutions or request a quote.
Process validation typically refers to the steps producing the drug substance or bulk drug product (например. смешивание, грануляция, сжатие). Packaging validation specifically applies validation principles to the packaging operations (уплотнение, маркировка, carton filling, и т. д.). Однако, both follow the same DQ/IQ/OQ/PQ lifecycle and GMP requirements. По сути, packaging validation ensures the packaging process (not just the manufacturing process) consistently meets quality requirements.
Key tests include container closure integrity (CCIT) to verify seals, Совместимость материала (extractables/leachables studies), stability tests (drug in final package under stress), and distribution tests (shock, вибрация, temperature for transit). Additional checks cover label accuracy, visual inspection systems, weight/checkweighing, and any contract-specific tests (например. sterile film strength). All tests should have predefined acceptance criteria and be documented.
The timeline depends on complexity (number of formats, оборудование, sites). A single-line IQ/OQ/PQ could range from a few weeks to a couple of months. Factors: the number of OQ parameter tests, required sample sizes in PQ, and time for test execution (например. stability at accelerated conditions takes weeks). Planning parallel activities (like preparing protocols while equipment arrives) can save time.
The primary references are GMP regulations: 21 CFR Part 211 (US FDA), EudraLex Vol. 4 (GMP ЕС, Приложение 1 и 15), and WHO GMP Annex 9. ICH Q7/Q10 provide general validation principles, ICH Q9 covers risk management for packaging decisions, и ИСО 11607-2 applies to sterile pack process validation. Pharmacopeial standards (USP, Ph. Евро.) and local guidelines (например. Chinese NMPA, India Schedule M) also influence packaging requirements. Always align with the target market regulations.
Jinlu’s packaging machines are built for GMP. They provide design documents (УРС, FDS), сертификаты калибровки, and OQ/PQ support. Their equipment (блистерные машины, картотеры, линии заполнения) features precise servo control and hygiene design to meet validation specs. As shown on Jinlu’s site, machines carry cGMP/CE logos and come with technical documentation sets. Jinlu also offers installation and validation support, custom solutions, and training to help implement validated packaging lines. For more info, see Jinlu’s Blister Packing Machine or Cartoning Machine pages.
Ссылки:
1.Q7A Руководство по надлежащей производственной практике для активных фармацевтических ингредиентов -- НАС. Управление по контролю за продуктами и лекарствами
2.Проверка процесса —— Wikipedia
3.ИСО 11607- Package Validation Testing —— ddltesting.com
4.〈1207〉 Package Integrity Evaluation—Sterile Products —— usp.org
5.Container and Closure System Integrity Testing in Lieu of Sterility Testing as a Component of the Stability Protocol for Sterile Product -- НАС. Управление по контролю за продуктами и лекарствами

Мелкий фу, Основатель компании Jinlupacking, приносит 30 многолетний опыт работы в секторе фармацевтического оборудования. Под его руководством, Jinlu превратилась в надежного поставщика, интегрирующего дизайн., производство, и продажи. Петти с радостью делится своими глубокими отраслевыми знаниями, чтобы помочь клиентам разобраться в сложностях фармацевтической упаковки., гарантируя, что они получат не только оборудование, но настоящее комплексное сервисное партнерство, адаптированное к их производственным целям..
У каждого продукта и завода есть свои проблемы и ситуации с упаковкой.. Мы здесь, чтобы помочь с гарантированным качеством машин, индивидуальные решения, и самые беспроблемные услуги.
Авторское право © 2026 JinLuPacking.Все права защищены.. Условия & Условия и политика конфиденциальности
Дружественные ссылки: Богатая упаковка | Производители машин для наполнения капсул