
제약 산업에서, 보호 포장 is crucial for maintaining drug quality and patient safety. It comprises multi-layered systems – primary, 반성, and tertiary packaging – that work together to shield medicines from moisture, 산소, 빛, 오염, and physical damage. 1차 포장 (물집 팩, 바이알, 병, 향 주머니) makes direct contact with the drug; 2차 포장 (상자, 상자, 인서트) groups and labels products; tertiary packaging (cases, 팔레트, stretch wrap) protects bulk shipments. Each layer and material (플라스틱, 유리, 알루미늄 호일, cardboard, insulation liners, 등.) offers specific barrier functions. Advanced features like child-resistant caps, 변조 방지 씰, moisture-barrier films, and serialization further enhance protection and compliance.
These requirements are enforced by global GMP regulations (FDA, WHO, EU) which mandate durable labeling and anti-tampering features. Automation plays a key role: high-speed blister machines, counting systems, 탑승자, 실러, 그리고 라벨러 ensure consistent, GMP-compliant packaging at scale. 이 기사에서는, we explore protective packaging types, 재료, 규제 기준, 기계류, with practical examples and comparisons.
Protective packaging in pharmaceuticals refers to the multi-layered packaging system designed to preserve drug quality throughout its lifecycle. It is not just a container, but an active safeguard. As WHO guidelines emphasize, packaging must protect against all adverse external influences (수분, 빛, 산소, 온도) that can alter a drug’s properties. 실제로, this means using materials and designs that create effective barriers and signals. 예를 들어, 물집 팩 seal individual tablets under an aluminum foil or plastic film, which limits moisture and oxygen exposure. 비슷하게, glass vials and bottles (아래에 표시됨) provide chemically inert, airtight barriers ideal for injectables and sensitive liquids.

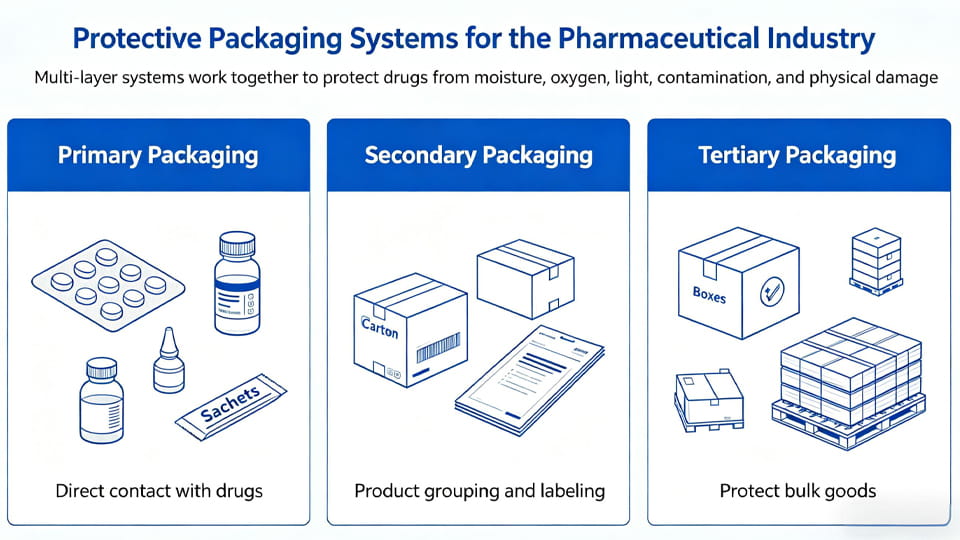
수치: Worker handling primary packaging of tablets (Blister packs ensure that each tablet remains isolated from moisture, 공기, and tampering.).
Pharmaceutical protective packaging also often integrates functional features: moisture/oxygen scavengers, 건조제, light-blocking foils, and radiation shielding for sensitive formulations. Child-resistant caps and tamper-evident seals are examples of safety features that prevent misuse. 요컨대, protective packaging in pharma actively maintains drug stability and safety, acting as both a barrier and communication tool (labels, 지침). This multi-faceted role is essential to prevent degradation, 투약 오류, or contamination that could harm patients and erode trust.
Protective packaging is vital because medicines encounter many hazards before reaching patients. 제조 중, 저장, 및 유통, pharmaceuticals can be exposed to humidity, 산소, 빛, 열, physical shocks, and even microbial contamination. Without robust packaging, active ingredients may degrade (losing potency or forming impurities) or tablets may crumble during transport. 예를 들어, exposure to moisture can hydrolyze drugs like aspirin, while oxygen can oxidize vitamin C, compromising efficacy. Light-sensitive drugs (예를 들어. chlorpromazine) can break down under UV unless shielded by opaque materials.
From a regulatory standpoint, packaging failures are common causes of recalls and safety issues. Poor labeling or packaging defects can lead to mix-ups and dosing errors. 한 포장 전문가가 지적했듯이, protective features directly support patient well-being and compliance. This is why agencies require durable labels (분명한, 내마모성) and anti-tampering measures. Good packaging not only preserves stability and sterility, but also prevents accidental poisonings: child-resistant closures alone have “significantly reduced incidents of accidental poisoning in children”. 본질적으로, protective packaging underpins product integrity, 환자 안전, and legal compliance.
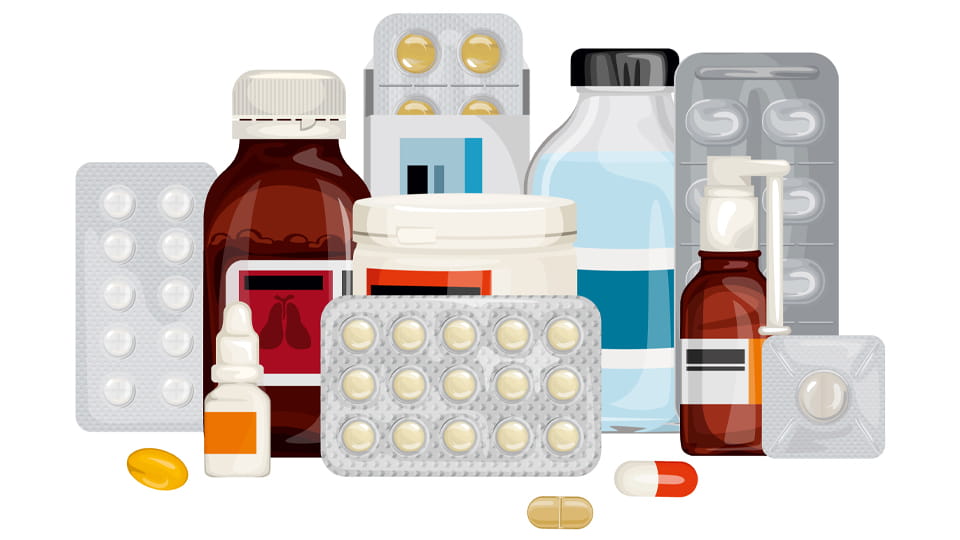
Primary packaging is the first barrier around the drug, contacting the product directly. Its role is to keep the drug stable and sterile until use. ~ 안에 solid dosage forms, blister packs are most common: each tablet or capsule sits in a plastic cavity and is sealed by a foil or film backing. This ensures each dose is protected from air and moisture until popped out. Glass vials and ampoules serve as primary packaging for injectables and serums; they create an airtight, inert environment to keep liquids sterile. 병 및 항아리 (플라스틱 또는 유리) are used for syrups, 서스펜션, 그리고 캡슐. They often include child-resistant caps and tamper-evident seals to protect contents and users. Flexible sachets and medical-grade pouches are primary packs for powders, granules or single-dose items; these lightweight packs offer moisture barrier and convenience.
Each of these primary packs is engineered for safety. 예를 들어, a child-resistant cap on a prescription bottle requires an adult motion to open, preventing accidental pediatric ingestion. Blister foil backing (Alu-Alu or Alu-PVC) is chosen based on barrier needs – Alu-Alu offers near-total light and moisture protection, while PVC/Alu is cost-effective for less-sensitive drugs. 요약, primary packaging is the personal enclosure for each dose, combining material barriers and smart design to preserve the medication’s intended performance.
Secondary packaging protects and groups the primary packages. It includes carton boxes, sleeves, 쟁반, and inserts. Cartons hold blister strips or bottles in place, preventing movement or abrasion during handling. They also carry essential information – dosage instructions, 배치 번호, and expiry dates – that supports patient safety and regulatory compliance. Inside cartons, cardboard inserts or foam separators prevent contact between vials or blisters, adding cushioning. 수축 포장 or over-wraps may bundle multiple units (예를 들어. a retail 10-packs) to deter tampering. 실제로, secondary packaging organizes and annotates the drug, ensuring that each group of doses is protected and traceable through the supply chain.
Tertiary packaging is used for bulk shipping and storage – typically corrugated boxes, cases, 팔레트, and shrink-wrap. While patients never see this layer, it is crucial for global distribution. Robust corrugated cases can withstand stacking pressures and rough handling. Pallets and stretch films secure many cartons together for forklift handling. This outermost armor keeps products intact from the factory to the pharmacy. 요컨대, tertiary packaging ensures that protective layers inside remain undisturbed during long-distance transport.

Different materials provide different barrier properties. The table below summarizes common materials vs. their protective functions:
| 재료 | 장벽 / Protective Function | 일반적인 용도 |
| Plastic Polymers | Excellent moisture barrier; 가벼운 중량, formable | Blister cavities (PVC/알루), 병, 모자, sachet films |
| Cardboard/Paper | Rigidity, cushioning, stacking strength; surface for labels; 재활용 가능 | Secondary cartons, 인서트, labels |
| 알루미늄 호일 | Superior barrier to moisture, 산소, 그리고 빛; heat-sealable | Blister pack backing, foil pouches, 스트립 팩 |
| 유리 | Chemically inert; absolute gas/moisture barrier; sterilizable | 바이알, 앰플, bottles for injectables and sensitive liquids |
| Insulating Materials | Thermal barrier to maintain cold chain temperatures | Insulated liners, refrigerated cartons for vaccines/biologics |
| Specialty Coatings | UV-blocking or antimicrobial coatings for extra protection | Light-sensitive drug packaging, sterile applications |
Each material is chosen based on the drug’s needs. 예를 들어, PVC-Alu blister film offers flexibility and moisture protection for tablets, while Alu-Alu foil (pure aluminum) provides the highest level of barrier for very sensitive products. Glass is favored for biologics because it won’t interact with the drug. Modern composite films and engineered polymers (PVDC, EVOH) are also used to achieve tailor-made barrier performance. The key is selecting the right material so that the packaging material itself never undermines the drug’s safety.
Modern protective packaging goes beyond inert barriers. Functional design adds user safety and supply-chain features. 주요 기능은 포함됩니다:
Innovative solutions also appear: eg. cold-seal blister technology or recyclable packaging formats. Such innovations aim to balance protection with sustainability. But the bottom line remains: the protective system must reliably shield the drug in real-world conditions while facilitating correct use.
제약 포장 is governed by strict GMP and pharmacopoeial requirements. As WHO advises, packaging must “protect against all adverse external influences (수분, 빛, 산소, 온도)” and meet quality standards throughout shelf life. 사실은, the quality of a drug is linked to its packaging quality – materials must be non-reactive and proven suitable via stability studies.
실제로, this means applying GMP to packaging processes just like to drug manufacturing. 예를 들어, ISO 15378 defines GMP specifically for primary packaging material suppliers. The EU and FDA require that packaging materials and container-closure systems are validated and controlled. Chapter 5 of EU GMP explicitly states that suppliers of packaging materials deserve “as much attention as that given to suppliers of starting materials”. Comprehensive documentation and testing (extractables/leachables, material certificates) are expected to ensure safety.
Printed information on packages is also regulated. EU GMP mandates that text and images on cartons and labels be “clear, lightfast, and abrasion-resistant”, preventing recalls due to illegible instructions. 뿐만 아니라, global regulations like the EU Falsified Medicines Directive require tamper-evident features on packaging and serialization (track-and-trace) of unit-dose packs. FDA similarly demands evidence of child-resistant or tamper-resistant closures where applicable. 요컨대, pharmaceutical packaging must not only physically protect the drug, but also comply with detailed quality and safety standards at every layer.
Modern pharma packaging relies on specialized machinery at each stage. Below is a mapping of common machine types to packaging functions, with examples of Jinlu Packing equipment:
| 기계 유형 | Protective Function | Example Jinlu Solution |
| 물집 포장 기계 | Forms sealed primary packs (tablets/caps in foil cavities). Creates the first protective barrier around each dose. | Jinlu DPP-270Max / DPP-180Pro high-speed blister lines (알루미늄-PVC, 알루-알루). |
| 병 충전 & Sealing Line | Fills liquids or counts solids into bottles; applies child-resistant caps and seals. Protects liquids from leaks and contamination. | Jinlu automatic bottle filling & 캡핑 기계 (integrated lines with induction sealing). |
| Tablet/Capsule Counting Machine | Accurately counts doses into bottles or blisters (ensures correct quantity). Critical for patient dosage and GMP compliance. | Jinlu JLPCP-600 series tablet/capsule counting machines (최대 800 ppm). |
| 포장 기계 | Encases primary packs into 보조 상자 with labels (batch no., 지침). Prevents movement and provides tamper-evidence when sealed. | Jinlu automated cartoners (horizontal/vertical) capable of 15,600 상자/시간. |
| 라벨링 머신 | Applies product, 일괄, and serial labels on bottles/cartons. Ensures traceability and regulatory info. | Jinlu automatic labelers (bottle and carton labelers). |
| Induction Sealing Machine | Applies foil liners to bottle mouths. Adds leak-proof, 수분- and tamper-evident seal on primary containers. | Jinlu induction sealing units (for PC/PE foil). |
| 케이스 패커 / Palletizer | Groups cartons into tertiary cases or pallet loads. Secures shipments with stretch-wrap and straps. | Jinlu case packers and palletizers for bulk packaging. |
Manufacturers like Jinlu Packing offer integrated packaging lines combining these machines. 예를 들어, an automatic blister line can be synchronized with a cartoner and case packer to form a complete primary-to-tertiary packaging solution. Such automation ensures consistent sealing, minimizes human error, and fully supports GMP production. 특히, Jinlu’s blister and cartoning machines meet cGMP standards for pharma. Their blister units run from 4,800 에게 11,200 물집/시간, while cartoners can box up to 15,600 units/hour.
[jl_youtube src=”https://www.youtube.com/embed/1Bb_J6rluac”]
동영상: An automated pharmaceutical packaging line, illustrating protective packaging processes (물집 충전, carton packaging, 등.) 행동 중 (Jinlu Packing facility).
Selecting the optimal packaging involves considering the drug’s sensitivity and regulatory needs. Key factors include the drug form (solid vs. 액체), shelf-life goals, and transport conditions. 예를 들어, highly moisture-sensitive tablets may need Alu-Alu 물집 팩 그리고 건조제; vaccines require validated cold-chain packaging. A good approach is to conduct stability studies with candidate materials to confirm barrier performance.
Machine selection is equally important. High-speed lines improve efficiency but require reliable quality control systems (예를 들어. 검사 카메라, 체중 검사). Partnering with an experienced packaging equipment supplier can ensure the line design suits the product. Jinlu Packing provides customization and support for pharma lines; 예를 들어, they offer special feeders (떨리는, 브러시) to handle delicate tablets, and can help integrate serialization and vision systems for traceability.
실제로, many companies use a combination: 예를 들어, a blister packer to create primary blisters, followed by a tablet counter and bottle filler for some dosage forms, then a cartoning machine and case packer for secondary/tertiary steps. By aligning packaging machinery with product requirements, manufacturers achieve both efficiency and compliance.
Protective packaging is far more than a container in 의약품 제조 — it is a critical system that safeguards drug stability, 안전, and regulatory compliance throughout the product lifecycle.
From production to patient use, medicines face risks such as moisture, oxygen exposure, 빛, 오염, and physical damage. Well-designed protective packaging acts as a barrier against these factors, helping maintain drug efficacy and shelf life while ensuring patient safety. Industry research consistently shows that packaging materials and sealing integrity directly influence pharmaceutical stability and product quality.
For pharmaceutical manufacturers and packaging engineers, success comes from aligning materials, packaging design, and automation equipment into one integrated protection strategy. Companies that optimize protective packaging not only reduce product loss but also improve compliance readiness and operational efficiency.
Ready to Upgrade Your Protective Packaging System?
If you are planning a new pharmaceutical packaging line or improving existing protection performance, choosing the right packaging solution is essential.
Jinlu Packing supports pharmaceutical and nutraceutical manufacturers with reliable protective 포장 기계 and customized automated packaging solutions tailored to real production needs.
👉 Contact our team today to discuss your project or request a customized solution.
It is a layered packaging system designed to shield medicines from external threats (수분, 빛, 산소, 오염, impact) and to ensure patient safety. This includes primary packs (물집, 바이알, 병), secondary packs (carton boxes with labels), and tertiary shipping containers, all working together to keep drugs stable and usable until administration.
There are three levels: 주요한 (direct contact with drug, 예를 들어. 물집 팩, 병, 바이알), 반성 (grouping units into cartons with instructions and batch IDs), and tertiary (bulk transport crates, 팔레트, stretch wrap). Each layer serves a distinct role – primary for isolation, secondary for information and organization, tertiary for shipment integrity.
Common materials include plastics (PVC, 애완 동물) for blister cavities and bottles, aluminum foil for high-barrier blister backs, glass for injectables (바이알, 앰플), cardboard for cartons and inserts, and insulating liners for cold-chain. Each offers specific barriers: 예를 들어. aluminum foil blocks moisture/light, glass provides inert containment, and certain plastics (PVDC, EVOH) add extra oxygen barriers.
Modern machines automate precise filling and sealing. 예를 들어, an automatic blister machine forms and heat-seals each tablet into a foil-backed cavity, creating a reliable barrier. A bottle filling line meters liquid and caps it with an induction-sealed, child-resistant closure. Counting machines verify dosage counts, and cartoners wrap primary packs into tamper-sealed boxes. Using these machines ensures consistent quality and GMP compliance across every package.
Pharmaceutical packaging must meet stringent GMP rules. WHO and pharmacopeias require packaging materials not react with the drug and protect it from moisture, 빛, 산소, 그리고 오염. Labeling must be durable and clear. Regulations like the US and EU mandates demand child-resistant closures for many medications and tamper-evident seals to prevent counterfeits. Packaging lines must also implement controls (supplier qualification, testing for extractables/leachables) to ensure safety throughout shelf life.
참고자료:
1.Dosage Form Drug Manufacturers cGMPs (10/93) – FDA Inspection & 규정 준수.
2.TRS 902 – Annex 9: 의약품 포장에 관한 지침 - WHO.
3.ISO 15378 – Primary Packaging Materials for Medicinal Products (Quality Management Standard).

사소한 푸, 진루패킹 창업자, 가져오다 30 제약 기계 부문에 대한 다년간의 전문 지식. 그의 리더십 아래, Jinlu는 디자인을 통합하는 신뢰할 수 있는 공급업체로 성장했습니다., 생산, 그리고 판매. Petty는 고객이 제약 포장의 복잡성을 헤쳐나갈 수 있도록 깊은 산업 지식을 공유하는 데 열정을 갖고 있습니다., 장비뿐만 아니라 제품도 받을 수 있도록 보장, 생산 목표에 맞춘 진정한 원스톱 서비스 파트너십.



