右を選択する 医薬品包装機 製品の安全性を確保するために重要です, 規制のコンプライアンス, 効率的な生産. 世界の医薬品包装市場は活況を呈しており、ある予測ではそれを超えると予測されています $160 10億まで 2026. 同時に, 規制当局は厳格な品質管理を要求しています: 例えば. FDA の CGMP (21 CFR パート 210 ~ 211) 製造に厳しい要件を課す, 処理, そして薬の梱包. 実際に, ほぼ 半分 医薬品のリコール パッケージングまたはラベル付けの間違いが含まれる, 賭け金を強調する. このガイドでは, 製品フォーマットなど、重要な考慮事項をすべて説明します。, 容量, コンプライアンス, オートメーション, ベンダーサポート, など – お客様のニーズに合った適切な医薬品包装機器を自信を持って選択できます. マシンタイプをカバーします (ブリスターマシン, カプセル充填剤, 箱詰め業者, 等), 自動システムと半自動システムを比較する, 専門家のヒントも含まれています, 比較表, 選択手順のフローチャート, よくある質問, 実用的な推奨事項.
適切なマシンの選択が重要な理由
医薬品包装 通常の消費者向けパッケージよりもはるかに重要です. それ 医薬品の品質を守る, 投与量の正確性を保証します, そして患者の安全を守ります. 例えば, WHO のガイドラインでは、包装は医薬品を湿気から保護する必要があると強調しています, ライト, 酸素, 保存期間を通じてその他の悪影響が及ぶ可能性があります。. 同じく, FDA の CGMP 規則では、「方法」を義務付けています。, 設備, 加工および梱包における「管理および管理」は、医薬品の安全性と正しい成分を保証する必要があります. 実務的には, 不適切な梱包は原因となる可能性があります 汚染, 間違ったラベル付け, 投与量の間違い, または装置の誤動作 – 患者に損害を与える場合も. 確かに, リコールデータの分析により、 まで 50% 医薬品リコールの割合はパッケージまたはラベルの間違いによるものです. それは一見小さな見落としを意味します (間違ったバーコード, 破損した水疱, 錠剤の数え間違い) 多額の費用がかかるリコールや規制上の警告を引き起こす可能性があります.
安全を超えて, 適切な機械は生産性を向上させ、コストを削減します. 最新の医薬品ラインには充填が統合されています, 封印, 計数とラベル付けを高速自動システムに導入. 出力ニーズと自動化レベルに合った機器を選択すると、スループットが大幅に向上し、労力が削減されます。. 逆に, 選択を誤ると頻繁に渋滞が発生する可能性があります, 一貫性のない塗りつぶし, ダウンタイム – 効率を損なう. そして、製薬工場は頻繁に稼働しているため、 24/7, 予定外のメンテナンスは悲惨な結果を招く可能性があります. こういった理由から, 業界の専門家はバランスの取れた選択を重視します: 「医薬品包装機器を選択するには、GMP 準拠能力を慎重に評価する必要があります」, 検証の準備状況, 長期的な運用信頼性を実現します。」. 要約すれば, 適切なマシンが確実に 品質とコンプライアンス (欠陥/リコールの回避) 最適化しながら 生産能力とコスト.

医薬品包装機の種類
「医薬品の包装」は多くの製品と形式をカバーします. 機械が陥るのは、 一次包装 (薬物と接触する最初の層) そして 二次・三次包装 (外箱, バンドル, パレット). 代表的なマシンには次のものがあります。:
- 充填および分注機: のために 液体, クリーム, または粉末 バイアル/ボトル/小袋入り. 例: 液体充填剤, 粉末充填機, シロップ充填剤. あなたの製品が 液体の薬, 液体用に設計された機械を選択する (ペリスタルティックまたはピストンフィラー). のために 粉末・顆粒 (例えば. 袋詰め中のバルク錠剤), 容積測定またはオーガーフィラーが理想的です.
- カプセル/タブレット機器: のために カプセルまたは錠剤, キーマシンは カプセル充填機, 打錠機, そして 計数/充填機. 最新のカプセル充填機は配置を自動化できる, 分離, 充填, そしてカプセルのロック. 錠剤プレスは粉末を錠剤に圧縮します. 計数機 (光学カウンター) ボトルまたはパウチに固形分を与えて計数する. JinLuPacking が指摘するように, で 固形分 「すべては正確な計数から始まります」 – 高速マルチチャンネルセンサーにより、各ボトル/パウチの計数が正確に正確であることを確認します.
- ブリスター包装機: ブリスターパック入りの錠剤・カプセルの場合. ホイルの下に錠剤を密封する熱成形トレイ. ブリスターマシン として来てください 平板 (インデックス付き) または ロータリー連続 種類, さまざまな形式で (PVC ベースまたは冷間成形フォイル). 単位用量の包装に最適です.
- 箱詰め/箱詰め機: 二次パッキン. 箱詰め業者 カートンを折りたたんで密封する, ブリスターパックやボトルと患者用リーフレットを挿入することが多い. 彼らはそうすることができます 水平 (サイドロード) または 垂直 (トップロード) 箱詰め業者は箱の組み立てを自動化します, コンテンツの読み込み, 密封 – 手作業を軽減し、確実に箱を確実に閉めることができます。 (GMPトレーサビリティにとって重要). 現代の箱詰め業者は、厳しい規制を満たすために、ステンレス鋼の衛生的なデザインと改ざん防止シールを使用しています。.
- ラベル作成者と検査者: ラベル付けマシン バッチコードを含む印刷済みラベルをボトル/カートンに貼り付ける, 有効期限, とバーコード. 検査システム (カメラビジョン, 金属探知機, X線) 充填精度をチェックする, 異物, ラベルの存在, コードの可読性. コンプライアンスのために, シリアル化をサポートする機器を探す (追跡可能なバーコード) そしてエラーの拒否.
- 統合包装ライン: 実際に, 製薬工場でよく使われるのは 完全な行 上記のマシンを組み合わせて. 例えば, タブレットからカートンへのラインにはタブレットプレス機が含まれる場合があります, 乾燥剤インサルター, カプセルカウンター, ボトルアンスクランブラー, 充填ユニット, キャッピングマシン, 誘導シーラー, ラベリング, 箱詰め業者, そして最終検査コンベア. これらのエンドツーエンド システムは、ラインとして事前に組み立てて検証することができます。, 資格認定とフットプリントを簡素化します. .
要するに, 剤形と包装形式に適合するマシンを選択してください. あるガイドがアドバイスするように、, 「ブリスター包装機は錠剤に最適です」, 一方、横型の製袋・充填・密封システムは高処理量のパウチングに使用されます。」. 製品を理解する (錠剤, カプセル, 液体, 粉) そして希望のパッケージ (水膨れ, ボトル, 小袋, 既成ポーチ, 等) マシン選びの第一歩です.

考慮すべき重要な要素
特定のマシンまたはベンダーを評価する場合, これらの重要な要素を念頭に置いてください:
- 製品 & パッケージ形式の互換性: 機器がお客様の製品の形状と容器を正確に処理できることを確認します. 剤形と容器の種類 案件: 錠剤, カプセル, 液体, 粉末にはそれぞれ独自の取り扱いニーズがあります. 例えば, 計数可能な固体は計数システムまたはブリスターシステムを使用します, 一方、液体には密閉性の高い液体充填剤が必要です. こちらもチェック サイズと形状: 一部の機械は複数の充填重量または容器サイズを処理できます, 他の人は献身的です. 複数の SKU を計画する場合は柔軟性が重要です. 多くの場合、マシンの説明には互換性のある形式が指定されています; 可能であればサンプル製品の試用版で確認する.
- 生産速度 & 容量: マシンのスループットを現在および将来の需要に合わせます. 機械には「定格速度」がある (例えば. ボトル/分またはブリスター/分) そして実践的なアウトプット (切り替えを考慮して). オーバースペック (非常に高速) お金とスペースを無駄にする; スペック不足がボトルネックを引き起こす. 多くのメーカー (ジンルのように) 見積速度: 例えば. 一部のカプセルラインでは 1 時間あたり数十万個のカプセルを製造できます, 計数機 >99.98% ~6,000 ボトル/時間の精度, およびカートナーまで 450 カートン/分. 現在の生産量だけでなく成長も考慮 – JinLuPacking は、「成長の余地を残しながら現在の需要に適合する」容量を選択するようアドバイスしています. 量の増加が予想される場合は、モジュール式マシンまたはマルチレーンのオプションを探してください。.
- 正確さ & 品質管理: 製薬業界では高精度は譲れません. 充填および計数装置は、次のことを保証する必要があります。 余分な錠剤や不足している錠剤はありません, 正しい投与量, 確実に密封されたパッケージ. トップラインは、すべてのコンテナで「1 つも余分ではなく、1 つも欠けていない」ことを目指しています. 各マシンの評価 精度仕様: 例えば. カウントフィラーが主張するかもしれない 99.98% 正確さ. 厳しい公差を達成するには、マルチチャンネル光学センサーまたはサーボ駆動の投与をお勧めします。. 組み込みの高品質機能もチェックしてください: 例えば. カウントを確認するためのビジョンシステム, 漏れ検出, 重量チェッカー. マシンは統合する必要があります 工程内制御 エラーをキャッチする (バッチの拒否, アラート) 安定した品質を確保するために.
- GMP & 規制の遵守: 製薬機械は厳しい衛生基準と文書基準を満たさなければなりません. 探す cGMP準拠の設計: すべての製品接触部品 ステンレス鋼 (SUS316L) またはその他の製薬グレードの材料, 滑らかな表面 (デッドレッグや隙間がないこと), 掃除のため簡単に分解できます. 表面は腐食/汚染に耐える必要があります. マシンには次の機能が必要です 安全ガード, 密閉された制御パネル, 可能であれば工具を使わずに切り替え可能. 同様に重要なのはソフトウェアのコンプライアンスです: 現代の医薬品ラインは多くの場合 FDA の管轄下にあります 21 CFRパート 11 (電子記録) および EU GMP 付属書 11 (コンピュータ化されたシステム). 機器がサポートしていることを確認することをお勧めします。 電子記録, 監査証跡, およびロールベースのアクセス, 包括的な検証パッケージが付属しています (デザインドキュメント, FAT/SATプロトコル, IQ/OQ/PQ テンプレート). 要するに, マシンはそうでなければなりません 検証準備完了. 輸出する場合は、CE マーキングまたはその他の現地認証を取得した機械を優先します。. 梱包ガイドラインに留意してください (誰が, FDA, 欧州連合) あらゆるストレスをクリーンに保ちながら製品の完全性を維持, 検証済みのプロセス.
- 柔軟性 & 切り替え時間: 医薬品ラインでは多くの製品が稼働することがよくあります. マシンを探す 素早い切り替え機能. 機能にはサーボ駆動の調整可能なヘッドが含まれる場合があります, 設定の「レシピ」を記憶, 金型やガイドの工具不要の交換. JinLuPacking は「工具不要」を強調します, 複数のSKUを処理するための「レシピストレージによる素早い切り替え」. 数分でフォーマットを切り替えられるマシン (vs. 時間) ダウンタイムの削減と OEE の増加によって成果が得られます. 半自動機械は調整が簡単ですが、オペレーターが必要です; 全自動機械は通常、適切に設計されていれば工具交換が速くなります。. レイアウトの柔軟性(垂直フィードと水平フィード)も考慮してください。, 車線, 等, さまざまなパッケージ形状に対応するため.
- 自動化レベル: どちらかを決定します 半自動 (いくつかの手動手順) そして 全自動 システム. 自動包装ラインは労働力と人的ミスを最小限に抑えます, 一貫性を達成する 24/7 スループット. 例えば, 自動カートナー直立, オペレーターの入力を最小限に抑えてカートンに積み込んで密封します, 一方、半自動カートナーは手動でカートンを積み込む必要があります. 完全自動の計数/充填ラインには、振動フィーダーと錠剤用のピック アンド プレース アームが統合されています, 手作業によるカウントの排除. トレードオフ: 自動機械はコストが高く、設置が複雑です. 半自動機械はコストは低いですが、作業員が拘束され、動作速度が遅くなります。. 以下の表で比較します:
| 特徴 |
自動機 |
半自動機械 |
| スループット |
非常に高い (継続的な, 24/7) |
適度 (バッチまたは断続的) |
| 労働要件 |
低い (監視する少人数の乗組員) |
より高い (給餌/ハンドリングのオペレーター) |
| 柔軟性 |
中くらい (専用フォーマット; 音量を上げると速くなります) |
高い (手動処理によりさまざまなフォーマットが可能) |
| 切り替え時間 |
もっと長くてもよい (複雑なセットアップ) |
通常は短い (より単純な仕組み) |
| 初期費用 |
より高い (複雑なコントロール & ロボット工学) |
より低い |
| メンテナンス |
複雑な (訓練を受けた技術者が必要です) |
よりシンプルに (ユーザーが修理できる部品) |
要するに, ~のために自動機械を使用する 大容量, 安定した生産. バッチの実行が小さいか頻度が少ない場合, 予算的には半自動でも十分かもしれない. (例えば, 半自動ブリスターマシンは、オペレーターが手動で錠剤を装填している間のみブリスターをヒートシールします。) どのレベルを選択しても, コントロールが最新のものであることを確認する (PLC/表示器, 安全インターロック) ユーザーインターフェイスが直感的であること.
- 衛生 & 洗浄性: 製薬機械は、 掃除と滅菌が簡単 相互汚染を防ぐために. すべての接触部品が洗浄のためにアクセスできることを確認してください, 隅に粉や液体がたまりません. 多くの製薬機械は 工具不要のクイックリリースパーツ 掃除用. マシンがサポートしているかどうかを検討してください CIP (定位置クリーン) または拭き掃除サイクル. 設計では次のことを許可する必要があります ドライクリーニング (例えば, 粉末を入れるための交換可能なパーツ). 粉末が含まれる場合はフィルターと換気も評価します. 製薬環境では厳格な洗浄プロトコルが必要です, したがって、毎日/毎週のメンテナンスルーチンを計画してください. (ヒントとして: HMC のアドバイスをお読みください。製薬ラインには「残留物を除去し、バッチ間の相互汚染を防ぐための厳格な洗浄プロトコル」が必要です。)
- データの整合性 & トレーサビリティ: 機械は完全なバッチトレーサビリティを容易にする必要があります. 探すべき機能は次のとおりです シリアル化されたバーコーディング (単位用量パック用), データロギング プロセスパラメータの, 工場のMES/ERPシステムとの統合. 最上位マシンはすべての重要なパラメータの監査証跡を提供します (温度, 重み, カウント). 連載用 (米国の DSCSA や EU の FMD など、多くの市場で必要とされています), ラインが 1D/2D コードの視覚検査に対応できることを確認する, 悪いコードを拒否する, 親子データを集約する (例えば。, 個々のブリスターパックをカートンにリンクする). 基本的に, この機器は、FDA および EU の追跡義務を満たすのに役立ちます。. 最近のマシンには OEE が含まれることがよくあります (全体的な機器の有効性) ダッシュボードの監視も.
- 総所有コスト (TCO): 表示価格の向こう側に目を向ける. 考慮する エネルギー消費 (発熱体, 真空ポンプ, モーター), 消耗品費 (膜, ホイル, 粉末, シールなどのスペアパーツ), そして サービス要件. 非常に安価なマシンはダウンタイムが長かったり、非標準の部品を使用している可能性があります. 逆に, 高品質の機械はより多くのエネルギーを使用しますが、廃棄物は少なくなります. 要因としては以下が挙げられます。: メンテナンスの手間 (メンテナンスの頻度と難しさ), スペアパーツのリードタイム (輸入機械にとって重要), および資金調達/ROI. Jinlu ブログのブリスター マシンの注意事項, 「高品質のマシンは初期費用が高くなりますが、無駄やダウンタイムを削減できます。」. 要約すれば, を計算します ライフサイクルコスト: 購入価格 + 運営費 + メンテナンス. サプライヤーは多くの場合、このための TCO ツールまたは計算ツールを提供します。.
- サプライヤーの経験 & サポート: ついに, 製薬経験のある評判の良いメーカーを選択する. 包装設備は長期的な投資です; あなたはパートナーが欲しいです, ただのベンダーではない. サプライヤーの実績を確認する: あなたの地域/業界に参考となる植物はありますか? 自社の機械は製薬会社の顧客向けに検証されていますか? 実証済みのGMP専門知識を持つサプライヤーを優先する, 包括的な検証サポート, 規制された業界での確かな実績. アフターサービスも評価: 試運転サポートを提供していますか (脂肪/飽和/IQ/OQ), スペアパーツの在庫, リモート診断, そしてトレーニング? 優れたサプライヤーがレイアウトの最適化をサポートします, 規制監査を支援する, 緊急の故障にも対応します. 夜間の営業を避ける – 業界の認定を探す (ISO, ce), 保証, そしてグローバルネットワーク.
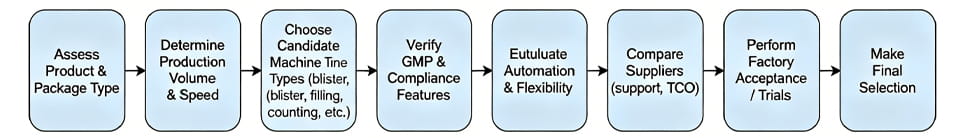
以下は、 選考プロセスをまとめたフローチャート:
自動 vs. 半自動包装: 比較
オプションの範囲を決めるとき, 全自動ラインと半自動ラインを比較するのに役立ちます. 供給を統合した自動ライン, 形にする, 充填, 1 つの連続プロセスでシーリングを行う, 一方、半自動ラインでは手動介入が必要です (例えば. ボトルまたはカートンを手で積み込む). 一般的に:
- 自動 システムが提供する 非常に高いスループット (多くの場合、~にとって不可欠な >100 カートン/分, または大きなカプセルバッチ). 彼らは、 最小限の労力 (1– 監視するオペレーター 2 名), ヒューマンエラーを減らす. また、通常は完全なデータ ログも自動的に収集されます。. 欠点は 資本コストが高い そして複雑さ. メンテナンスには訓練を受けた技術者が必要です.
- 半自動 機械は 低コスト そしてよりシンプルな. 製品や紙パックを供給するために労働者が必要になる場合があります, それらを〜に適したものにする 小規模から中規模のラン. 切り替えが早くなる傾向にある (動作軸が少なくなる), そしてトレーニングはより簡単になります. しかし, 手作業による手順により、人件費とばらつきが増加します. 半自動ラインは全自動ラインと同じ速度に達することはほとんどありません.
主要な属性の簡単な比較は次のとおりです。:
| 特徴 |
自動ライン |
半自動ライン |
| スループット |
非常に高い (100+ 単位/分) |
適度 (オペレータの速度に応じて) |
| 労働 |
低い (1–2名, ほとんど見てる) |
より高い (部品を供給する数人のオペレーター) |
| 柔軟性 |
中くらい (特定の形式に最適化された) |
高い (オペレーターはその場で適応できる) |
| 切り替え |
エンジニアリングが必要な場合があります (より多くの部品/ツール) |
多くの場合、より速くなります (交換する部品が少なくなる) |
| 初期費用 |
高い (PLC, ロボット工学, センサー) |
より低い (基本的なアクチュエータとコンベア) |
| メンテナンス |
集中的な (専門的な技術スキル) |
より簡単に (一般的な機械のメンテナンス) |
実際に, 多くの製薬製品は両方を組み合わせています: 例えば. 半自動カートナーに供給する自動充填ステーション, またはその逆. キーは一致することです ボリュームに対する適切なレベルの自動化. 規模を拡大する予定がある場合、または 24/7 走る, 完全な自動化は通常効果をもたらします. パイロット バッチまたは予算制約の場合, 半自動のベンチトップまたは卓上マシンで十分かもしれません.

避けるべきよくある落とし穴
- コンプライアンスの無視: コストを節約するためにGMP機能を決して軽視しないでください. 準拠していないマシンは監査失敗またはリコールを引き起こす.
- 「最も安い」オプションを購入する: 入札額が非常に低い場合、精度やサポートが不足する可能性があります. 長期的なROIを考慮する, 前払い料金だけではなく.
- 将来のニーズを無視する: 買わないでください ちょうど今日の 出力; 成長や新製品の余地を残す.
- 工場での受け入れテストをスキップする: 必ずサンプルを使用して工場で機械をテストしてください (脂肪) 出荷前に. 同じく, サイトの承認を実行する (土) チームと協力して仕様を満たしていることを確認してください.
- 公共施設を見渡す & 統合: 施設要件を確認する (力, 空気, 水) マシンがフロアプランに適合していることを確認してください. また、既存のコンベアや制御システムとスムーズに統合できるようにします。.

適切なサプライヤーを選択する方法
適切な機器メーカーと提携することは、機器自体の選択と同じくらい重要です. 探す:
- 業界の専門知識: を専門とするサプライヤー 製薬機器 FDA/EMA/WHO の基準と検証プロトコルの最新情報が得られます. レイアウトや品質管理についてアドバイスすることができます. あるガイドが述べているように、, 「選択したマシンは剤形と一致する必要があります, 包装形態, 規制要件, と生産能力」, だから経験が重要だ.
- 検証サポート: ベンダーが完全なドキュメントを提供していることを確認する: 工場/サイトの受け入れテスト計画, IQ/OQ/PQプロトコル, リスク評価, およびソフトウェア検証パッケージ. 理想的には、SAT の実行も支援します。.
- アフターサービス: 保証条件と会社のスペアパーツやサービスの取り扱い方法を確認する. 現地オフィスまたは代理店はありますか? リモートサポートは利用可能ですか? 部品を迅速に発送したり、技術者を派遣したりできるベンダーは、ダウンタイムを最小限に抑えます。.
- 評判 & 参考文献: 同様の設置の参考資料や事例を尋ねる. 技術サポートが充実している会社を選ぶ, グローバルサービスチーム, 検証済みの製薬プロジェクト, 実証済みの機械の信頼性.
- カスタマイズ能力: 制作には独自のニーズがあるかもしれません (特殊な材料, クリーンルーム仕様, 等). 優れたサプライヤーは機械をカスタマイズします (例えば. 特別な部屋, 自動化の追加, ユーザーインターフェース言語) あなたの要件に合わせて. 彼らは, 例えば, スペースとプロセスのニーズに合わせたターンキーラインを提供.
これらの点についてサプライヤーを慎重に精査することで、, プロジェクトのリスクを軽減します. 覚えて: 「適切な機器パートナーは、品質チームとエンジニアリングチームの延長になります。」.
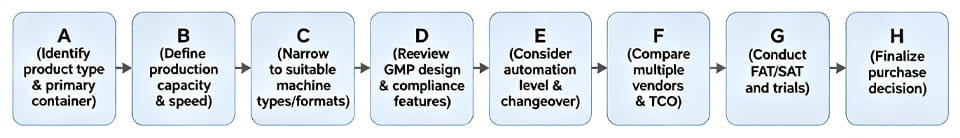
選定フローチャート
わかりやすくするために, これは簡単です 選択手順のフローチャート:
次のステップ
適切なマシンの選択は複雑ですが、やりがいのあるプロセスです. 詳しく書くことをお勧めします ユーザー要件仕様 (URS) あなたのチームのために, 上記の必須機能をすべてリストアップ. 次に、信頼できるサプライヤーから見積もりとデータシートを求めます。. ご購入の前に, をリクエストする 工場受け入れテスト そして, もし可能なら, 実際の製品を使用したサイトトライアル.
で JinLu包装機械, 当社はエンドツーエンドの医薬品包装ラインを専門としています。. 当社の製品 (カプセル充填剤, ブリスターパッカー, 計数機, 箱詰め業者, 等) GMP準拠と精度を考慮して設計されています. 製品ページをご覧になることをお勧めします (見る 内部リンク 下に) カスタマイズされたソリューションについては当社のエンジニアにお問い合わせください. 自動箱詰めラインが必要かどうか, 半自動ブリスターマシン, または完全に統合されたボトルライン, 選択と検証のプロセスをご案内します.
医薬品包装機に関するよくある質問
「医薬品包装機」とは一体何なのか??
製薬業界で医薬品を梱包するために使用される自動装置です。. これには、液体をバイアルに充填する機械が含まれる場合があります。, ボトルに入る錠剤を数える, トレイの中の膨らんだ錠剤, 製品を箱に詰める, 等々. 本質的には, 封じ込めに役立つあらゆる機械, ラベル, シール, または、バンドル医薬品は医薬品包装機器の一部です.
これらの機械にとって GMP 規格がそれほど重要な理由?
優れた製造業 (GMP) 標準 (FDA 21CFR, EU GMP, WHOのガイダンス) 製品が一貫して高品質であることを保証する. 準拠した機械は衛生的な設計になっています (ステンレス鋼316L, 掃除可能な表面), 検証済みのコントロール (正確な, 追跡可能な操作), そして適切な文書化. 準拠していない機器は、リコールや規制当局への言及につながる可能性のある汚染や製造エラーのリスクを伴います.
これらの機械の価格はいくらですか?
コストはマシンのタイプと複雑さによって大きく異なります. シンプルな半自動ベンチトップ錠剤カウンターは数千ドルになる可能性があります, 一方、高速自動ブリスター ラインや統合ボトル ラインには数万、さらには数十万の費用がかかる場合があります。. 総所有コストを常に考慮する (上を参照). 要件を定義したら、複数のベンダーから見積もりを取得するのが最善です.
錠剤に最適な包装機はどれですか?
それはフォーマットによって異なります. ボトルや袋に入った錠剤の数を数えるのに, 自動計数/充填機が最適です. 1回分包装用, ブリスターマシンが一般的です. ボトルにバルク包装する場合, 自動ボトル充填およびキャッピングライン (上流にタブレットカウンターあり) 使用されています. 一部の企業では、錠剤の個別用量包装にスティックパックまたはサシェの機械も使用しています。. 各形式の長所/短所を評価する (料金, 保護, ユーザーの好み) 機械を選ぶ前に.
私の製品は湿気に敏感です. 何を考慮すればよいでしょうか?
湿気に敏感な錠剤またはカプセルの場合, コールドフォームが必要な場合があります (アルアル) ブリスターマシンまたは専用の乾燥剤システム. 機械のシールが医薬品グレードのものであることを確認してください, 統合された乾燥または窒素不活性オプションを検討してください. 梱包材自体も (フォイル対. PVCフィルム) も同様に重要です.
1台のマシンを複数の製品に使用できますか?
多くのマシンはある程度調整可能です, しかし、大きく異なる製品間の切り替えは困難な場合があります. さまざまなフォーマットのレシピ/設定を備えた機器を選択してください. 工具不要, クイックチェンジツールにより、同じマシン上で複数の SKU を処理できるようになります。. しかし, 多種多様な製品を想定している場合, モジュラーマシンまたは複数の専用ラインの方が効率的である可能性があります.
参考文献:
1.医薬品の包装に関するガイドライン(別館 9) - 誰が.
2.現在の適正製造基準要件に関する質問と回答 | 装置 - 私たち. 食品医薬品局.
3.GMPパッケージング 2026: 要件, ガイドライン, コントロール, 一次包装と二次包装 – pharmuni.com.
4.世界的な包装規制と規格 – GMPナビゲーター.












