Tablet deduster with metal detector primarily used in the intermediate stage of tablet compressing, between the presse à comprimés et emballages pharmaceutiques.
The dedustering system separates unqualified tablet, while a metal detection system identifies and removes tablets containing metal impurities.







Fully enclosed anti-interference, ensuring metal detection precision of 0.2mm.

Accurately identify and remove excessive metal impurities in pharmaceuticals.

The tablets are smooth and have no burrs, and the transparent cover is easy to observe.

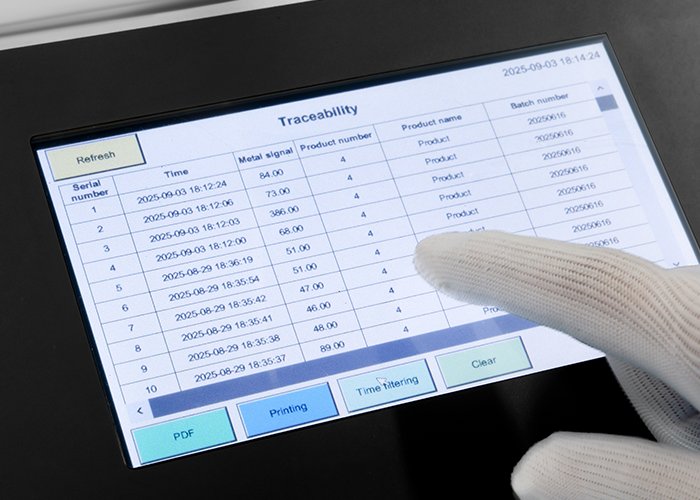
HMI displays all machine data, making operation simpler and more convenient.
| Modèle | JL-DMD |
| Capacité | 500,000 pièces/h |
| Sensitivity | Fe 0,2 mm, non-Fe 0.3mm, SUS 0,4 mm |
| Convient pour | Comprimé, Capsule, Gélules |
Situé à Canton, JL est un fournisseur de machines de conditionnement pharmaceutique intégrant l'ingénierie, fabrication et vente.
Notre équipe se concentre sur le développement du traitement pharmaceutique & des machines d'emballage qui allient fonctionnalité, qualité, et la rentabilité, en plus de fournir à nos clients un service à guichet unique sans tracas.
Nos avantages








Our customer service team and our own engineering team are always on call to solve your urgent needs.
The Tablet deduster with metal detector is produced in strict accordance with 6S workshop manufacturing standards, and the machine complies with CE and cGMP standards.
Jinlupacking fournit des manuels et des vidéos complets pour une utilisation quotidienne, entretien et nettoyage.
A automatic dépoussiéreur de comprimés with metal detector is a critical quality control device after the tablet pressing process. It is primarily used in the intermediate stage of tablet compressing machine production, located between the tablet press and pharmaceutical packaging machine. Its core value lies in its integrated design, enabling dual quality control: a screening system separates unqualified tablets (such as broken, de forme irrégulière, or weight-incorrect tablets), while a metal detection system identifies and removes tablets containing metal impurities. This integrated design not only reduces equipment footprint throughout the production process but, plus important encore, avoids the risk of secondary contamination during tablet transport across multiple machines.
En production réelle, ce détecteur de métaux and tablet deduster machine handles tablets ranging in diameter from 3mm to 25mm, meeting the inspection requirements for tablets of various shapes, including round and irregular shapes. Typical applications include final quality inspection of oral solid dosage forms, end-to-end quality tracking of high-risk drugs (such as sustained-release and controlled-release tablets), and specialized testing of special dosage forms sensitive to metal impurities (such as tablets containing magnetically sensitive ingredients). According to Thermo Scientific’s application data, this type of equipment can reduce the risk of metal contamination in pharmaceuticals by over 99.9%, significantly reducing the risk of drug recalls.
From a production efficiency perspective, current models have a maximum processing capacity of up to 1 million tablets per hour (based on 8mm diameter tablets), while the metal detection module can process up to 500,000 comprimés par heure, perfectly matching the production pace of high speed rotary tablet press machines and preventing it from becoming an efficiency bottleneck on the production line.
A tablet deduster machine with metal detection consists of two core modules: a screening system and a metal detection system, which work together through sophisticated mechanical design and electronic control. Emballage Jinlu’s metal detection tablet deduster machine utilizes a top-spinning design that allows tablets to naturally rotate during the lifting process, improving screening efficiency and removing dust from the tablet surface.
The metal detection system operates based on the working principle of electromagnetic induction. When tablets containing metal impurities pass through the magnetic field, they disrupt the magnetic field balance, generating an induced voltage difference between the receiving coils on both sides. When the system detects a voltage difference exceeding the set value, it triggers rejection. Actuellement, there are two mainstream rejection methods: air-blowing and push-rod. The air-blowing method uses high-pressure airflow to instantly blow impure tablets away from the main conveyor channel and is suitable for lightweight or small tablets. The push-rod or flip-rod method uses a mechanical lever to push rejected tablets into a waste channel or drop them into a waste collection device and is suitable for heavier or odd-shaped tablets.v
The key performance parameters of an automatic metal detector and high speed tablet deduster machine include the polishing capacity and efficiency of the screening process, as well as the impurity detection sensitivity of the metal detection system. The screening system is suitable for tablets ranging from 3mm to 25mm. Using a 316L stainless steel screen, it can maintain a tablet breakage rate below 0.1% at a processing rate of 500,000 comprimés par heure.
The performance parameters of a metal detection system are more complex, requiring differentiating detection sensitivities based on different metal types. According to industry standards, high-quality models should be able to detect iron particles larger than 0.3mm, non-ferrous metals (such as copper and aluminum) larger than 0.4mm, and stainless steel particles larger than 0.5mm. Emballage Jinlu’s metal detectors have improved their detection limit for iron particles to 0.2mm by optimizing coil layout and signal processing algorithms. This accuracy is sufficient to capture tiny metal debris generated during production, such as iron chips from tablet press wear.
Dynamic stability is another key metric for high-speed metal inspection and screening equipment, referring to its ability to maintain detection accuracy while operating at full capacity. This metric is typically measured by signal fluctuation amplitude, which should be within ±2% for high-quality equipment.
Production adaptability parameters are equally important, including the equipment’s processing speed adjustment range, tablet thickness adaptability, and environmental tolerance. Modern machines typically offer adjustable processing speeds ranging from 300,000 à 1,000,000 tablets per hour to accommodate varying batch production needs. Environmental requirements include a temperature range of -10°C to 40°C and a humidity range of 30% à 85%, which is particularly important in tropical regions or high-humidity production environments.
In pharmaceutical equipment design, compliance is as important as performance. Tablet sorting and metal detector machines with metal detection must meet multiple international standards, including FDA (NOUS. Administration des aliments et des médicaments) material certification, Ema (European Medicines Agency) quality regulations, et GMPc (Bonnes pratiques de fabrication actuelles) requirements. These standards not only govern the selection of equipment materials but also set specific requirements for structural design, enregistrement de données, and cleaning validation.
Components that come into direct contact with nutraceutical and pharmaceutical products must be pharmaceutical-grade materials. 316L stainless steel is preferred for its excellent corrosion resistance and low leachable properties. It withstands common cleaning and disinfecting agents (such as ethanol and peracetic acid) without corrosion or leachables. The equipment’s structural design fully embodies the cGMP principle of « cleanability. » Emballage Jinlu’s tablet deduster with metal detection features a quick-release design, allowing both the sieve body and outer cover to be quickly removed. All drug-contact surfaces are clear, ensuring thorough cleaning. This design of jinlu packing’s capsule tablet deduster with metal inspection not only effectively prevents cross-contamination but also significantly reduces cleaning validation time. En outre, the complete isolation of the working chamber and drive chamber prevents mechanical impurities such as lubricants from entering drug-contact areas, structurally eliminating contamination risks.
Data traceability is a core compliance requirement for modern pharmaceutical equipment. FDA 21 Partie CFR 11 regulations impose strict standards on electronic data management, including data integrity, confidentiality, et traçabilité. High-end models, such as the DMD, automatically record all inspection data, including the size, location, and rejection time of metal impurities. This data is encrypted and stored with audit trails, fully meeting regulatory data management requirements.
Chaque produit et chaque usine ont leurs propres défis et situations en matière d'emballage.. Nous sommes là pour vous aider avec des machines de qualité garantie, solutions personnalisées, et les services les plus simples.
Inscrivez-vous pour recevoir le lancement de notre nouvelle machine et nos informations.
Droits d'auteur © 2026 JinLuPacking.Tous droits réservés. Termes & Conditions et politique de confidentialité
Liens amicaux: Emballage riche | Fabricants de machines de remplissage de capsules