
ফার্মাসিউটিক্যাল শিল্পে, প্যাকেজিং বৈধতা is a critical subset of প্রক্রিয়া বৈধতা focused on ensuring that packaging equipment and processes consistently protect drug product quality. It involves a documented program of qualification (ইনস্টলেশন, Operational, কর্মক্ষমতা) and testing (যেমন. container-closure integrity, স্থিতিশীলতা, পরিবহন) to prove that packaging systems perform as intended under GMP conditions. Effective packaging validation safeguards patient safety (by preventing contamination, degradation or mix-ups) and ensures regulatory compliance (এফডিএ, ইএমএ, WHO, ICH, আইএসও). This guide explains the definition, পদক্ষেপ, পরীক্ষা, মান, এবং সেরা অনুশীলন for pharmaceutical packaging validation, and shows how automated equipment (পছন্দ Jinlu Packing’s machines) is designed to meet these stringent requirements.

Packaging validation in pharma means demonstrating with documented evidence that the packaging process (equipment and materials) reliably produces a package that protects the drug product. অনুশীলনে, it applies the same rigorous validation concepts used in manufacturing (IR/WH/PQ) থেকে প্রাথমিক, মাধ্যমিক, and tertiary packaging অপারেশন. According to WHO’s GMP guidance, “Packaging processes and equipment need validation/qualification in the same way as any other part of processing within a pharmaceutical facility.”. অন্য কথায়, packaging validation ensures the container-closure system, লেবেলিং, and packaging lines consistently meet quality specifications and regulatory requirements. The goal is to maintain drug strength, purity and stability through packaging, distribution and shelf life, while preventing mix-ups, counterfeits, বা ক্ষতি.
Typical deliverables in packaging validation include a ব্যবহারকারীর প্রয়োজনীয়তা স্পেসিফিকেশন (ইউআরএস) (the design & quality criteria), Installation/Operational/Performance Qualification protocols, and testing records. The validation process covers:
Each level must protect the product. উদাহরণস্বরূপ, the Australian TGA defines “primary packaging” (the container that immediately covers the goods) and distinguishes সেকেন্ডারি প্যাকেজিং as the outer box or wrapper. শেষ পর্যন্ত, packaging validation ties back to GMP – ensuring that containers and closures do not alter the drug এবং “provide adequate protection against external factors” during storage and use.
Pharmaceutical packaging is the final defense for drug quality. Validating packaging processes is vital because:
Packaging validation addresses all packaging stages. সাধারণত, we distinguish:
Each level requires risk assessment and appropriate testing: উদাহরণস্বরূপ, প্রাথমিক packaging might involve USP <1207>-style integrity tests, যখন মাধ্যমিক packaging tests might include carton strength and label adhesion, এবং তৃতীয় packaging uses ISTA/ASTM test protocols. All must be documented in the validation plan.
Packaging validation typically follows the equipment/process qualification life cycle, often described as DQ/IQ/OQ/PQ. These stages ensure a new or modified packaging line meets requirements at each stage.
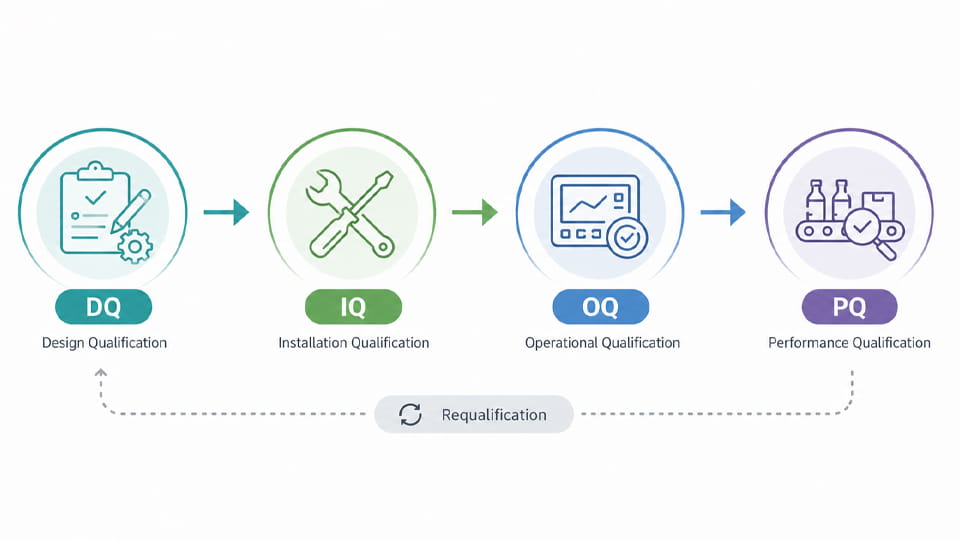
The table below summarizes the DQ–IQ–OQ–PQ stages:
| মঞ্চ | উদ্দেশ্য | Key Documents | Example Activities/Tests |
| ডিজাইনের যোগ্যতা (ডিকিউ) | Verify the packaging system design meets URS/GMP | ইউআরএস (requirements spec), design specs (FDS/HDS/SDS), risk assessment | Design review, material compatibility checks, DFMEA |
| ইনস্টলেশন যোগ্যতা (আইকিউ) | Confirm proper installation of equipment | IQ protocol/checklist, installation log, ক্রমাঙ্কন শংসাপত্র | Verify utilities, equipment assembly, ক্রমাঙ্কন |
| অপারেশনাল যোগ্যতা (OQ) | Verify equipment functions within defined limits | OQ protocol, functional test plans, Sops | Test parameters (গতি, তাপমাত্রা, চাপ); seal checks |
| কর্মক্ষমতা যোগ্যতা (পিকিউ) | Ensure consistent quality during production runs | PQ protocol, batch records, release criteria | Production runs; sample testing (CCIT, fill weight, চাক্ষুষ পরিদর্শন) |
![]()
As the diagram shows, packaging validation is a life-cycle process from design through performance. Note that requalification may be needed after major changes or periodically.
Several specialized tests are commonly performed during packaging validation:
CCIT is essential for sterile and even non-sterile products. It assesses whether the container closure system (যেমন. vial+stopper+cap, ampoule, blister seal) forms a perfect barrier. Methods include vacuum decay, pressure decay, ছোপানো প্রবেশ, or microbial challenge. As one industry source explains, “Container Closure Integrity Testing (CCIT) is an assay that evaluates the adequacy of container closure systems to maintain a sterile barrier against potential contaminants.”. Regulatory documents (ইউএসপি <1207>, এফডিএ, EMA Annex 1) emphasize testing closure integrity to ensure no leaks or breaches. উদাহরণস্বরূপ, ইউএসপি <1207> provides guidelines on selecting leak test methods to certify sealed packages.
প্যাকেজিং উপকরণ (প্লাস্টিক, rubbers, inks, adhesives) must not adversely interact with the drug product. Extractables and leachables studies simulate long-term contact: extractables are chemicals driven out of packaging under harsh conditions, and leachables are those that actually migrate into the product under normal conditions. These tests ensure the packaging does not introduce toxic or stability-affecting impurities. Pharmacopeial and FDA guidance (যেমন. ইউএসপি <661>, Q3E) outline E/L testing for container closure systems. অনুশীলনে, one checks that the polymer, আবরণ, and label materials meet pharmacopeial standards (যেমন. USP glass type, ISO rubber closure specs). The WHO notes that pharmacopoeial standards for closures (like rubber stoppers) হয় “minimum requirements” and emphasizes stability studies to prove suitability.
As per ICH Q1A, stability studies must be done on the drug product in its final packaging configuration. During validation, packaging’s impact on stability is assessed: উদাহরণস্বরূপ, the final packaging is subjected to accelerated aging (high temp/humidity) and real-time conditions to ensure no degradation (যেমন. moisture ingress in blister packs, oxygen permeation in bottles). Stability-indicating assays (chemical and microbial) are performed. Packaging that fails to protect the product under stress invalidates shelf-life claims. এইভাবে, validation includes confirming that the chosen packaging meets shelf-life requirements (this overlaps with regulatory stability protocols).
Tertiary packaging tests simulate actual shipping conditions. Standard tests include drop height tests, কম্পন (truck/rail simulation), কম্প্রেশন (stacking weight), and temperature cycling (especially for cold chain). উদাহরণস্বরূপ, ASTM D4169 (for packaged products) or ISTA protocols may be used. A robust packaging validation plan verifies that cartons, cases and crates protect primary packages during handling: verifying that no breakage, label peeling, or moisture damage occurs under distribution conditions. For cold chain, temperature mapping and validated insulated shippers are tested to maintain required temperature ranges throughout transit.

Pharmaceutical packaging validation must meet multiple regulatory expectations:
সংক্ষেপে, any packaging line must comply with the national pharmacopeia and regional GMP standards. Compliance checklists typically cite FDA, ইইউ জিএমপি, WHO and relevant ISO standards, and often call for fully documented SOPs and validation master plans covering packaging.
Packaging validation poses unique challenges compared to manufacturing processes:
এসব চ্যালেঞ্জ সত্ত্বেও, the underlying principle remains: treat packaging like any other critical process, with thorough qualification and robust QC checks. As the ISPE paper concludes, প্যাকেজিং বৈধতা “differ(এস) little from the validation of processes used for drug manufacture.”.
To overcome challenges and ensure a successful validation, এই সেরা অভ্যাস অনুসরণ করুন:
By embedding validation thinking at every stage – from design to daily operation – manufacturers can ensure packaging lines deliver products that meet quality and compliance expectations.
The choice and design of packaging equipment heavily influence validation success. High-quality packaging machines are engineered to meet GMP requirements and support easy qualification. উদাহরণস্বরূপ, জিনলু প্যাকিংয়ের automated machines are built with features that streamline validation:
অনুশীলনে, a validated packaging line might include: a Jinlu ফোস্কা প্যাকিং মেশিন linked to a কার্টোনিং মেশিন এবং counting/filling lines. Each equipment must be qualified (IR/WH/PQ). Jinlu’s systems are designed to work seamlessly together (যেমন. “highly automated blister cartoning line” আপ 320 কার্টন/মিনিট), reducing integration issues during validation. We also offer case studies (যেমন. a complete গণনা এবং বোতলের লাইন, or an automatic cartoning & labeling line) that demonstrate end-to-end validated solutions.

By partnering with Jinlu or similar manufacturers, companies benefit from equipment that is “cGMP compliant”, supports a full validation lifecycle, and comes with after-sales support (কমিশনিং, প্রশিক্ষণ) to ensure the line remains validated.
Packaging validation is essential for pharmaceutical quality and compliance. By treating packaging operations as rigorously as core manufacturing steps, companies ensure their products reach patients safely. The process spans definition (ইউআরএস, risk assessment), যোগ্যতা (DQ/IQ/OQ/PQ), পরীক্ষা (CCIT, স্থিতিশীলতা, পরিবহন), and continuous monitoring. It is governed by GMP regulations (এফডিএ, ইএমএ, WHO) and industry standards.
Modern automated packaging equipment – such as Jinlu Packing’s machines – play a key role in validation. Designed for precision, hygiene and electronic traceability, they help manufacturers meet validation criteria efficiently. When selecting packaging lines, consider suppliers who provide complete validation documentation and support.
শেষ পর্যন্ত, thorough packaging validation is “the final safeguard for product quality and patient safety”, aligning with both regulatory demands and business goals.
Ready to validate your packaging line? Contact Jinlu Packing’s experts to discuss GMP-compliant packaging solutions or request a quote.
Process validation typically refers to the steps producing the drug substance or bulk drug product (যেমন. মিশ্রণ, দানাদার, কম্প্রেশন). Packaging validation specifically applies validation principles to the packaging operations (sealing, লেবেলিং, carton filling, ইত্যাদি). তবে, both follow the same DQ/IQ/OQ/PQ lifecycle and GMP requirements. অগত্যা, packaging validation ensures the packaging process (not just the manufacturing process) consistently meets quality requirements.
Key tests include container closure integrity (CCIT) to verify seals, উপাদান সামঞ্জস্য (extractables/leachables studies), stability tests (drug in final package under stress), and distribution tests (shock, কম্পন, temperature for transit). Additional checks cover label accuracy, visual inspection systems, weight/checkweighing, and any contract-specific tests (যেমন. sterile film strength). All tests should have predefined acceptance criteria and be documented.
The timeline depends on complexity (number of formats, সরঞ্জাম, sites). A single-line IQ/OQ/PQ could range from a few weeks to a couple of months. Factors: the number of OQ parameter tests, required sample sizes in PQ, and time for test execution (যেমন. stability at accelerated conditions takes weeks). Planning parallel activities (like preparing protocols while equipment arrives) can save time.
The primary references are GMP regulations: 21 সিএফআর অংশ 211 (US FDA), EudraLex Vol. 4 (ইইউ জিএমপি, অ্যানেক্স 1 এবং 15), and WHO GMP Annex 9. ICH Q7/Q10 provide general validation principles, ICH Q9 covers risk management for packaging decisions, এবং আইএসও 11607-2 applies to sterile pack process validation. Pharmacopeial standards (ইউএসপি, Ph. ইউর) and local guidelines (যেমন. Chinese NMPA, India Schedule M) also influence packaging requirements. Always align with the target market regulations.
Jinlu’s packaging machines are built for GMP. They provide design documents (ইউআরএস, FDS), ক্রমাঙ্কন শংসাপত্র, and OQ/PQ support. Their equipment (ফোস্কা মেশিন, কার্টোনার, ভরাট লাইন) features precise servo control and hygiene design to meet validation specs. As shown on Jinlu’s site, machines carry cGMP/CE logos and come with technical documentation sets. Jinlu also offers installation and validation support, custom solutions, and training to help implement validated packaging lines. For more info, see Jinlu’s Blister Packing Machine or Cartoning Machine pages.
তথ্যসূত্র:
1.Q7A সক্রিয় ফার্মাসিউটিক্যাল উপাদানগুলির জন্য ভাল উত্পাদন অনুশীলন নির্দেশিকা —— U.S. খাদ্য ও ওষুধ প্রশাসন
2.প্রক্রিয়া বৈধতা —— উইকিপিডিয়া
3.আইএসও 11607- Package Validation Testing —— ddltesting.com
4.〈1207〉 Package Integrity Evaluation—Sterile Products —— usp.org
5.Container and Closure System Integrity Testing in Lieu of Sterility Testing as a Component of the Stability Protocol for Sterile Product —— U.S. খাদ্য ও ওষুধ প্রশাসন

পেটি ফু, জিনলুপ্যাকিং এর প্রতিষ্ঠাতা, নিয়ে আসে 30 ফার্মাসিউটিক্যাল যন্ত্রপাতি সেক্টরে দক্ষতার বছর. তার নেতৃত্বে, জিনলু একটি বিশ্বস্ত সরবরাহকারী একীভূত ডিজাইনে পরিণত হয়েছে, উত্পাদন, এবং বিক্রয়. পেটি ক্লায়েন্টদের ফার্মা প্যাকেজিংয়ের জটিলতাগুলি নেভিগেট করতে সহায়তা করার জন্য তার গভীর শিল্প জ্ঞান ভাগ করে নেওয়ার বিষয়ে উত্সাহী, নিশ্চিত করা যে তারা কেবল সরঞ্জামই পাবে না, কিন্তু একটি সত্যিকারের ওয়ান-স্টপ সার্ভিস পার্টনারশিপ তাদের উৎপাদন লক্ষ্য অনুসারে তৈরি.
প্রতিটি পণ্য এবং উদ্ভিদের নিজস্ব প্যাকেজিং চ্যালেঞ্জ এবং পরিস্থিতি রয়েছে. আমরা এখানে মানের গ্যারান্টিযুক্ত মেশিনগুলির সাথে সাহায্য করতে এসেছি, কাস্টমাইজড সমাধান, এবং সবচেয়ে ঝামেলামুক্ত পরিষেবা.
কপিরাইট © 2026 JinLuPacking.সর্বস্বত্ব সংরক্ষিত. শর্তাদি & শর্তাবলী এবং গোপনীয়তা নীতি
বন্ধুত্বপূর্ণ লিঙ্ক: সমৃদ্ধ প্যাকিং | ক্যাপসুল ফিলিং মেশিন নির্মাতারা